Z. Zaidi ( Department of Dermatology, Dow Medical College, Karachi. )
Z Wahid ( Department of Dermatology, Dow Medical College, Karachi. )
R. Cochinwala ( Department of Dermatology, Civil Hospital and Institute of Skin Diseases. Karachi. )
M.Soomro ( Department of Dermatology, Civil Hospital and Institute of Skin Diseases. Karachi. )
A. Qureishi ( Department of Dermatology, Civil Hospital and Institute of Skin Diseases. Karachi. )
November 2002, Volume 52, Issue 11
Original Article
Abstract
Objective: To correlate the density of the yeast Malassezia with the clinical severity of seborrhoeic dermatitis.
Method: Fifty patients and twenty control subjects were selected for the study. The patients were evaluated both clinically for the severity of seborrhoeic dermatitis and microscopically for the presence of the yeast Malassezia.
Results: Out of 50 patients Malassezia was present in 41 patients (82%). On microscopic evaluation it was found that patients with mild seborrhoeic dermatiis had a density of 2+ (more than 5 but less than 10 yeast cells per high power field (hpf). Patients with moderate seborrhoeic dermatitis had a density of 3÷ (more than 10 but less than 20 yeast cells per hpt) and patients with severe seborrhoeic dermatitis had a density of 4+ (more than 20 yeast cells per hpf). Of the 20 normal subjects only 8 (40%) had Malassezia and they had a density of 1 + (5 or fewer yeast cells per hpf). The results show a strong correlation of the yeast Malassezia to the severity of seborrhoeic dermatitis (p value <0.05).
Conclusion: Malassezia increases in proportion with the severity of seborrhoeic dermatitis; an antifungal agent should therefore be used in the treatment of seborrhoeic dermatitis (JPMA 52:504;2002).
Introduction
Seborrhoeic dermatitis is a common papulosquamous disorder with a worldwide distribution. Its etiology is poorly understood, but the disease is strongly associated with the skin commensal yeast of genus Malassezia (formerly Pityrosporum1-5. The normal microflora of the scalp are Pityrosporum, aerobic cocci and Corynebacterium acne. High density of the sweat glands, rapid epidermal turnover and large sebaceous glands are responsible for the high proportion of microorganisms in the scalp6.
The number of Corynebacterium acne decreases in seborrhoeic dermatitis due to the increased blood flow as a result of inflammation, coiynebacterium being a strict anaerobic organism, The aerobic cocci do not significantly increase, as the scaling becomes more obvious. The only organism that significantly increases in seborrhoeic dermatitis is the yeast Pityrosporum6. The aim of this study was to correlate the severity of seborrhoeic dermatitis with the density of the yeast Malassezia (formerly Pityrosporum).
Patients and Method
Fifty patients with seborrhoeic dermatitis of the scalp and twenty control subjects were selected for study. The study was conducted at the department of dermatology Civil Hospital and at the Institute of Skin Diseases (ISD) Karachi, Thirty patients were selected from the Civil Hospital and twenty from ISD. In order to maintain a uniformity of results, the same doctor at each hospital examined the patients. The pathologist at ISD conducted the entire microscopic evaluation.
Patients selected were those who complained of itching and scaling of the scalp typical of seborrhoeic dermatitis preferably with seborrhoeic dermatitis at another site. Those selected for study were the patients without topical or systemic therapy for seborrhoeic dermatitis 4 weeks prior to study. Patients of psoriasis and those with a family history of psoriasis were excluded from the study and so were patients with dermatitis requiring antifungal or steroid therapy. The patients were examined both clinically and microscopically 4 days after a bland shampoo. Most of the people belonged to the lower socio-economic group. For control twenty subjects were selected from the paramedical staff of the department of dermatology Civil Hospital and ISD, Karachi. Most of the people belonged to the lower socio-economic group. The subjects selected were those, who had no systemic or dermatological disorder and did not complain of itching or scaling of the scalp. A scraping of the scalp was taken from them for microscopic evaluation 4 days after a bland shampoo.
The patients were grouped as normal (non-dandruff), dandruff (non-inflammatory seborrhoeic dermatitis) and those with inflammatory seborrhoeic dermatitis. Both dandruff and inflammatory seborrhoeic dermatitis are regarded as parts of a spectrum of the same disease3,6. Some patients therefore had dry flaky scales; others had the more classical yellowish red greasy scales with underlying eiythema.
The patients and controls were assessed both clinically and microscopically. Clinically the patients were examined on a 0-3 scale, With 0= clear, 1= mild scaling, 2= moderate scaling and 3= severe scaling. Clinically the type of scales (dry or greasy), underlying inflammation and the area of the scalp and body involved also assessed the severity.
For microscopic evaluation the scales were removed with a blunt scalpel, mounted in 20% potassium hydroxide. The specimen was left for one hour to soften the keratinized cells and then examined under a microscope. The scales were examined under both low and high power. While examining the specimen it is important to ensure that the material has softened adequately and that the intensity of light passing through was not too strong. The presence of short coarse mycelium together with spherical thick walled yeast confirmed the presence of Malassezia. The appearance of the fungal elements observed on direct examination is so characteristic of Malassezia that culture is not necessary for the diagnosis of this infection. These were graded as follows:
1+ - Total of 5 or fewer yeast cells per high power field (hpf)
2+ - More than 5 but less than 10 yeast cells /hpf
3+ -More than 10 but less than 20 yeast cells Ihpf
4+ - More than 20 yeast cells Ihpf
Results
Of 50 patients examined there were 26 males and 24 females. The age ranged from 13 to 48 years, with a mean of 32.4 years. Most of the patients examined, also had seborrhoeic dermatitis in other areas of the body, one patient had erythroderma. Dry scales were present in 34 patients and 16 patients had yellowish greasy scales. Three patients had systemic disease; these were pylonephritis, epilepsy and asthma.
Clinical evaluation: Twenty six patients had mild seborrhoeic dermatitis, 16 had moderate and 8 patients had severe seborrhoeic dermatitis.
Microscopic evaluation: Dry scales were generally seen in mild and moderate seborrhoeic dermatitis; they were present in 34 patients clinically and 26 patients microscopically, greasy scales generally found in moderate and severe seborrhoeic dermatitis, were present in 16 patients clinically and 15 patients microscopically (p value <0.05) (Table 1).
Microscopic grading: Nine patients were negative for Malassezia, in 2 patients it was 1+ (less than 5 yeast cells per high power field (hpf), in 17 patients of mild seborrhoeic dermatitis (SD) it was 2+ in 14 patients of moderate SD, the grading was 3+ and in 8 patients with severe SD the grading was 4+ (Table 2).
Of the 20 normal subjects, there were 12 females and 8 males whose ages ranged from 18 to 50 years with a mean of 31 .6 years. Malassezia was present in only 8 cases (40%). The grading was 1+ (Table 3).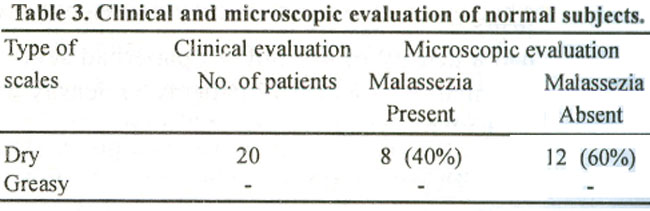
On microscopic evaluation Malassezia were significantly higher (p value <0.05) in greasy scales as compared to dry scales.
SD: Seborrhoeic dermatitis Mod: moderate-ye: negative for Malassezia, 1+: 5 or fewer yeast cells per high power field (hpf), 2+: more than 5 but less than 10 yeast cells per hpf, 3+: more than 10 but less than 20 yeast cells per hpf, 4+: more than 20 yeast cells per hpf.
Comparing the controls with the study subjects it was observed that only 40% of the former had SD with greasy scales (Table 4).
Discussion
There is a strong association of the yeast Malassezia and the severity of seborrhoeic dermatitis, but the results vary from study to study. This is partly due to the lack of a universal method of obtaining the scales. The amount of scales present at the time of examination depends upon the rapidity at which the scales are formed after their removal as by shampooing. The scales can be collected from the scalp either by the help of a blunt scalpel or obtained by the glass cylinder. Obtaining the scales by the method of scrubbing and aspiration washes the yeast from within the follicles. Pityrosporum within the follicles do not activate the complement, whereas those on the surface are associated with epidermal proliferation6. We used the simple scraping method of collecting the scales because of the simplicity of the procedure and its reducibility. The scales were collected 4 days after shampooing the scalp. In our study Malassezia were present in 40% of normal subjects, 76% in dandruff and 94% in inflammatory seborrhoeic dermatitis, This shows a strong correlation between the association of the yeast Malassezia and the severity of seborrhoeic dermatitis.
In a study by Kenneth et al5, Pityrosporum were present in 46% of normal subjects, 74% in dandruff and 82% in inflammatory seborrhoeic dermatitis. In a study of 100 patients by Mandelene et at7, they found 53 cases of mild seborrhoeic dermatitis, with a Pityrosporum density of 1+1 hpf in 46 patients and 7 patients had a density of 2+1 hpf. 32 cases had moderate seborrhoeic dermatitis, of which 14 had a density of 2+1 hpf, 15 patients had a density of 3+1 hpf and 3 had a density of 4+1 hpf, 15 patients had severe seborrhoeic dermatitis of which 13 patients had a density of 4+ Ihpf and 2 patients had a density of 2+/ hpf.
Daniel Grossier8 et al have also shown the relationship between Pityrosporum and seborrhoeic dermatitis in 10 patients with HIV disease, he found 0-1 yeast cells Ihpf in normal subjects, 1-3 yeast cells in mild cases of seborrhoeic dermatitis and 3-10 yeast cells/hpf in inflammatory seborrhoeic dermatitis. In a study by Roia7 et al, Pityrosporum were present in 75% of normal subjects and 96.4% in patients with seborrhoeic dermatitis.
Ketoconazole has been used to treat seborrhoeic dermatitis in a number of studies2-5, the good response indicates the relationship of the yeast to the etiology of seborrhoeic dermatitis. As the yeast cells cause increased epidermal proliferation, oral retinoids10-11, narrow band UVB therapy12, tacacitol cream13, cinnamic acid and related compounds14, topical metronidazole15, have been used in the treatment of seborrhoeic dermatitis. Treatment should no longer be with steroids alone but also with a suitable anti-yeast agent. The dose, application regimens, possible development of resistant organisms and cutaneous hypersensitivity should be considered while treating seborrhoeic dermatitis4,5.
References
1.Parry EM, Sharpe RG. Seborrhoeic dermatitis is not caused by an altered immune response to Malassezia yeast. Br. J. Dermatol.,1998; 139: 254.63.
2.Skimmer BR, Noah WP, Taylor MR. et at. Double blind treatment of seborrhoeic dermatitis with 2% ketokonazole cream. J. Am. Acad. Dermatol., 1985; 12: 852-56.
3.Carr MM, Pyrce MD, lye AF. Treatment of seborrhoeic dermatitis with ketokonazole: 1. Response of seborrhoeic dermatitis of the scalp to topical ketokonazole. Br. J. Dermatol., 1987; 116:213.16.
4.Green AC, Farr M.P Shuster S. Treatment of seborrhoeic dermatitis with ketokonazole: 11. Response of seborrhoeic dermatitis of the jace, scalp and trunk to topical ketokonazole. Br. J. Dermatol., 1987; 116: 217-21.
5.Shuster S. The etiology of dandruff and the mode of action of therapeutic agents. Br J of Dermatol. 1984; 111: 235-242.
6.McGinley JK, Leyden J.J. Marples RR. et at. Quantitative microbiology of the scalp in non.dandruff, dandruff and seborrhoeic dermatitis. J. Invest. Dermatol., 1975; 64: 401-5.
7.Heng YCM, Henderson LC, Barker CD, et at. Correlation of Pityrosporum density with the clinical severity of seborrhoeic dermatitis as assessed by a simplified technique. J. Am. Acad. Dermatol., 1990; 23: 82-86.
8.Grossier D, Button JE, Lebwohl M. Association of Pityrosporum orbiculare (Malassezia furfur) with seborrhoeic dermatitis on patients with acquired immunodeficiency syndrome (AIDS). J. Am. Acad. Dermatol., 1989; 20:770-73.
9.Roia FC, Vanderwyk RW. Resident microflora of the human scalp and its relationship to dandruff. J. Soc. Cosmec. Chem., 1969; 20:113-34.
10.Orfanos CE. Zouboulis CC. Oral retinoids in the treatment of seborrhoea and acne. Dermatology, 1998;196:140-47.
11.Burton LJ. Holden AC. Eczema Lichenification and Prungo. In: Rook A., Wilkinson DS, Ebling FJG, Champion RH, Burton LJ Textbook of Dermatology. 6th Edition. Blackwell Scientific Publication. 1998, pp. 638-43.
12.Pirkhammer D, Seeber A, Honigsmann HH, et al. Narrow band (TL-01) phototherapy is an effective and safe treatment option for patients with severe seborrhoeic dermatitis. Br, .J Dermatol., 2000;143: 964-68.
13.Nakayama J. Four cases of sebopsonasis or seborrhoeic dennatitis of the face and scalp successfully treated with 1 a-24 (R) dehydroxycholecalciferol. (tacalcitol) cream. Euro. J. Dermatol.. 2000;10:528-32.
14.Baroni A, Rossa R. New strategies in dandruff treatment: growth control of Pityrosporum ovalis. Dermatology, 2000; 201:332-36.
15.Prasad D, Pandhi R, Negi SK, et at. Topical metronidazole in seborrhoeic dermatitis -a double blind study. Dermatology, 2001; 202:35-37.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




