Nasreen Kirmani ( PMRC Research Centre, Jinnah Postgraduate Medical Centre, Karachi. )
Talat J. Hassan ( PMRC Research Centre, Jinnah Postgraduate Medical Centre, Karachi. )
Sadiqua N. Jafarey ( Department of Obstetrics and Gynaecology, Jinnah Postgraduate Medical Centre, Karachi. )
Salim Hafiz ( Department of Urology, Civil Hospital and Dow Medical College, Karachi. )
February 1991, Volume 41, Issue 2
Original Article
ABSTRACT
One hundred pregnant women In third trimester were screened for the carriage of group B streptococci (GBS), in a prospective study. Rectal and urogenital samples were collected, 25% women were positive for non group B beta haemolytic streptococci. Twelve (48%) were positive at single site and 13(52%) at more than one site. Beta haemolytic streptococci isolated were non-groupables In 19(76%) and groupables in 6(24%). Among groupables, Group D streptococci (GDS) were the commonest 3(12%) followed by Group 02(8%) and Group F 1(4%). Group B streptococci were not isolated in our study population (JPMA 41: 42, 1991).
INTRODUCTION
The importance of group B streptococci (GBS) as a pathogen for both the newborn and mother is well known1-4. Approximately 5-30% of females carry GBS in the vagina or rectum5-7 and 2-5% in urine. 8-9 GBS causes neonatal infections like septicaemia, meningitis, pneumonitis1,2,10,11 and puerReral infections like amnionitis, endometritis and sepsis. 1-4,12. It is a potential pathogen for urina? tract during pregnancy and known to cause primary1,9,12-14 as well as secondary urinary tract infections15,16. In terms of intranatal and perinatal infection risk, screening for GBS in the third trimester or at the time of delivery is important. A prospective study was planned to determine the carriage rate of GBS, frequency of GBS bacteriuria and antibiotic sensitivity pattern of GBS, in order to choose the right antibiotic to prevent potential lethal consequences of the organism to mothers and their babies.
SUBJECTS AND METHOD
One hundred pregnant women in the third trimester of pregnancy attending antenatal clinic of Jinnah Postgraduate Medical Centre, Karachi were registered irrespective of age and parity. Diabetic patients and those taking antibiotics were excluded. Complete history and physical examination were recorded. High vaginal swabs, rectal swabs, and mid stream urine specimens were collected in sterile containers. Samples were also collected for blood grouping. Urine specimens were screened for basic biochemical tests by multistix and cultured on blood, MacConkey’s and Islam’s agar plates by using standard method17 for quantitative analysis of bacteria,. High vaginal swabs and rectal swabs were inoculated on blood agar and Islam’s agar plates and incubated aerobically and anaerobically in 5% C0_ (in candle jar) at 37°C. Growth observed after every 24,48 and 72 hrs of incubation for beta haemolytic and non haemolytic colonies of streptococci on Mood agar and orange-red pigmented colonies of GBS on Islam’s agar plates. Colonies isolated were Gram stained, serologically grouped by Slidex Strep to kit (Biomeriux 6290 Charbonnicresles Bains, France) and isolates tested for bacitracin sensitivity.
RESULTS
The demographic characteristics of hundred pregnant women were as follows: all were from low socioeconomic group, living in Karachi. Sixty six belonged to Sindh (Karachi), 25 were from Punjab and 9 from NWFP. Their age range was 17-40 years, with a mean of 265 years. Twenty seven women were primigravidas and 73 multigravidas. Thirty four had blood group ‘B’ , twenty nine blood group ‘A’, twenty two blood group ‘D’ and fifteen blood group ‘AB. Beta haemolytic streptococci (non group B) were isolated from rectal and urogenital tract in 25% of pregnant females. Of twenty five females, twelve (48%) were positive at single site-either, vagina, rectum or urine and thirteen (52%) at more than one site (vagina-rectum, vagina-urine or vagina-rectum-urine) as shown in Table 1.
Of various serologic groups of beta haemolytic streptococci (BHS) isolated,6(24%) were groupable, of these3(12%) belonged to BHS group D, 2(8%) to group G and 1(4%) to group F, group B streptococci were not isolated in this study. The remaining 19(76%) of BHS were ungroupable. Relationship between carriage of J3HS (non-Group B) and age, ethnicity, gravidity and blood groups is shown in Table - II.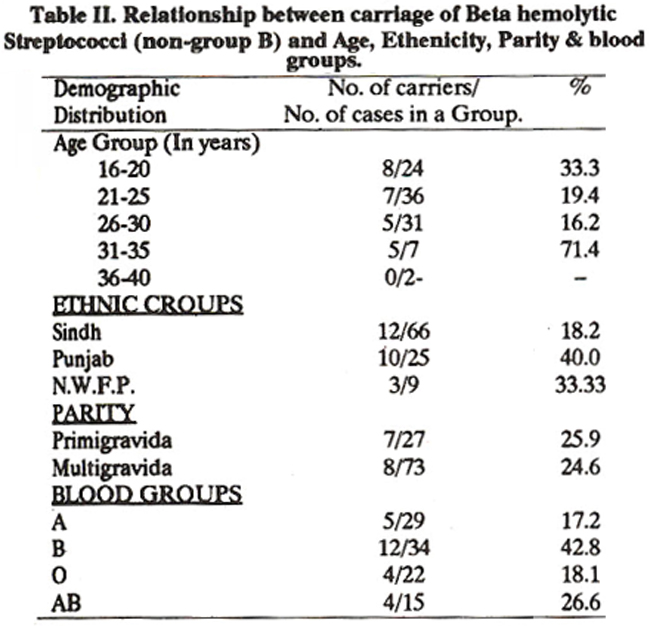
Differences found between carriage of BHS (non-Group B)and age, gravidity and blood group were not significant. Only significant difference was found in ethnic groups. Twelve (18%) out of sixty six women from Sindh (Karachi), ten (40%) of twenty-five women from Punjab, and three (33%) out of nine women of NWFP living in Karachi were the carriers of BHS (non-Group B).
DISCUSSION
This preliminary study shows that Group B streptococci are not present in the third trimester of pregnancy in our study population although 25% of pregnant women were found to be the carrier of beta haemolytic streptococci (non-Group B). The carriage rate of GBS reported from West is 5- 25%5,6 and from Peshawar 31% which seems to be very high. Reported differences may be due to culture techniques, variations in time of gestation at which cultures were obtained, 13,14 eating habits, seasonal 15 and geographical factors,5 genetic factors or ethnicity, age, parity15,16 and blood groups18 distribution. The role of ethnicity on the carriage of GBSIBHS cannot be ruled out as some individuals have natural Immunity against certain microorganisms, e.g. Mexican women have a lower and blacks living in United States higher carriage rates of GBS16. In our study carriage of GBS was nil but carriage rates of BHS (non-Group B) differs in different ethnic groups; women of Sindh (Karachi) have lower and women of Punjab and NWFP living in Karachi have higher carriage rate of BHS (non-Group B). This may be due to small number of cases in the later two ethnic groups. Age, parity and blood groups distribution of the individuals play an important role in carriage of BHS. Increasing age and parity are associated with lower rate in carriage15-16 as shown in this study. In order that the significance of the differences’ be more clear equal and larger number of cases should be studied. Individuals with blood group B are at a higher risk of colonization by GBS17 and BHS non-group B as indicated in this study. Beta haemolytic streptococcus has received little attention as a pathogen of the urinary tract although it is a potential pathogen during pregnancy. Carriage rate of 14% BHS non-Group B reported in our study was unllke2-5% of BHS Group B reported8,9 by others which may be due to misuse of antibiotics. Investigations have shown5,15,16 that anal or vaginal carriage of BHS Group B are possible cause of bacteriuria, and patients with diabetes mellitus are at a greater risk of infection by this organisma
REFERENCES
1. Eickoff, T. C., Klein, J. 0., Daly, A. K., Ingall, D. and Finland, M. Neonatal sepsis and other infections due to group B betahemolytic streptococci. N. Engi. J. Med., 1964; 271:1221.
2. Hood, M., Janney, A. and Dameron, G. Beta haemolytic streptococci Group B associated with problems of the perinatal period. Am. J. Obstet. Gynecol., 1961; 82: 809.
3. Bobitt, J. R. and Ledger, W. J. Obstetric observations in eleven cases of neonatal sepsis due to group B beta haemolytic streptococcus. Obstet. Gynecol., 1976; 47:439.
4. Anthony, B. F. and Concepcion, N. F. Group B Streptococcus in a general hospital. J. Infect. Dis., 1975; 132: 561.
5. Badri, M. S., Zawanch, S., Cruz, A. C., Mantilla, G., Baer, H., Spellagly, W. N. and Ayoub, E. M. Rectal colonization with Group B streptococcus; relation to vaginal colonization of pregnant women. J. Infect. Dis., 1977; 135: 308.
6. Baker, C. J. and Barrett, F.F. Transmission of group B streptococci among parturient women and their neonate. J. Pediatr., 1973; 83:919.
7. Akhtar,T., Zai, S. and Khatoon, J. Group B. Streptococcal carnage rate of pregnant women and new born infants. Pakistan J. Med. Res., 1984; 23: 12.
8. Wood, B. 0. and Dillon, H. C. Jr. A prospective study of group B streptococcal bacteriuria in pregnancy. Aim J. Obstet. Gynecol., 1981; 140: 515.
9. Mead, P. J. and Harris, R. E. The incidence of group B beta haemolytic streptococcus in antepartum urinaiy tract infection. Obstet. Gynecol., 1978; 51: 412.
10. Hey, D. J., Hall, R. T., Burly, V. F. and Thurn, A. N. Neonatal infection caused by group B streptococci. Am. J. Obstet. Gynecol., 1973; 116:43.
11. Barton, L L, Feigin, R. D. and Lins, R. Group B beta haemolytic streptococcal meningitis in infant. J. Pediatr., 1973; 82: 719.
12. Becroft, D. M. 0., Farmen, K., Mason, G. H., Morris, M. C. and Stewart, J. G. Perinatal infections by group B beta haemolytic streptococci. Br. J. Obstet. Gynaecol., 1976; 83: 960.
13. Baker, C.J. Early-onset of group B streptococcal disease. i. Pediatr., 1978; 93: 124.
14. Aber, R. C., Allen, N., Howell, J. T., Wilkenson, H. W. and Facklam, It It Nosocomial transmission of group B streptococci. Paediatries, 1976; 58: 346.
15. Anthpny, B. F. Carriage of group B streptococci during pregnancy, a pazzler. 3. Infect. Dis., 1982; 145: 789.
16. Anthony, B. F., Okada, D. M. and Hobel, C. J. Epidemiology of group B streptococcus; longitudinal observations, during pregnan. cy. J. Infect. Dis., 1978; 137: 24.
17. Frankel, S., Reitman, S. and Sonnenwirth, A. C. Gradwohis clinical laboratory methods and diagnosis. 6th ed. Louis, Mosby, 1963, p.512.
18. Regan, 3. A., Chao, S. and James, L. S. Maternal ABO blood group type B: A risk factor in the development of neonatal group B streptococcal disease. Paediatrics, 1978; 62: 504.
19. Freedman, It M., Ingram, D. L., Gross, I., Ehrenkranz, R. A., Warshaw, J.B., Baltimore, R.S. A half century of neonatal sepsis at Yale. Am. J. Dis. Child., 1981; 135: 140.
20. Siegal, J. D. and McCraken, G. M. Jr. Group D streptococcal infections. J. Paediat., 1978; 93: 542.
21. Easmon, C.S.F. Epidemiology of group B streptococcal infection. J. Med. Microbiol., 1984; 19: 139.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




