Ahmad Khalil Ahmed Al Sadi ( Catheterization Laboratory, King Abdallah University Hospital, Irbid, Jordan. )
Abdallah Fandi Yousef Omeish ( Department of Interventional Cardiology, King Abdallah University Hospital, Irbid, Jordan. )
Ibtisam Moawiah Al-Zaru ( Faculty of Nursing, Jordan University of Science and Technology, King Abdallah University Hospital, Irbid, Jordan. )
August 2010, Volume 60, Issue 8
Original Article
Abstract
Objective: To identify the timing and predictors of haematoma development after manual compression of femoral arteriotomies.
Methods: The study involved 239 consecutive patients undergoing cardiac catheterization at King Abdullah University Hospital, Irbid, Jordan. Size and timing of haematoma development were recorded. A total of 33 prespecified variables were evaluated by univariate analysis to identify predictors of haematoma development. These predictors were again analyzed using multivariate logistic regression model.
Results: Femoral haematomas developed in thirty nine patients leading to an overall frequency of 16.3 %. More than two-thirds (74.4%) of the haematomas occurred later in the bed unit. Only 8 among 33 studied variables were found to be significant predictors of haematoma development upon multivariate analysis. These predictors are low haematocrit level before the procedure, advanced age, wider waist circumference, high systolic blood pressure, multiple artery puncture, longer ACT at the end of the procedure, anticoagulant treatment before procedure, and Glycoprotein IIB/IIIa inhibitors use during the procedure.
Conclusions: Identifying predictors of haematoma development at femoral access sites and trying to correct or modify them when possible, can have a major impact on reducing the frequency of this serious complication. If an assessment indicates that a patient is at high risk, then several cautious management strategies should be implemented in this setting prior to performing cardiac catheterization (JPMA 60:620; 2010).
Introduction
Transfemoral sheath insertion is a routine component of both diagnostic as well as invasive cardiac catheterization procedures,1 and is the most common vascular access site for this sake.2,3 Haematoma development (HD) proved to be one of the most common vascular complications encountered after femoral artery puncture and its safe management remains always a serious concern for the operating cardiologist as well as the catheterization laboratory (cath lab) staff members for it constitutes a serious cause of morbidity and mortality in patients undergoing cardiac catheterizations.4
Manual compression over the puncture site is a proven and widely used method for obtaining haemostasis in most patients undergoing diagnostic cardiac catheterizations and percutaneous interventions (PCI) after their activated clotting time (ACT) normalizes (return to <150 to 180 seconds). Thrombus usually forms at the access site within minutes after placing the fingers 1 to 2 cm above the femoral puncture site and continuously exerting pressure for 10 minutes or more to achieve haemostasis.3 This is followed by 4 to 6 hours of complete bed rest and then gradual ambulation.5 Nowadays, sheath removal after diagnostic and invasive cardiac procedures has predominantly become a routine part of nursing practice, although in some academic institutions is still restricted to cardiology fellows serving the interventional or regular cardiology programmes.1,6-8
Although vascular closure devices (VCDs) which were first developed in the mid-1990s, are more and more used to achieve faster and more secure haemostasis of the femoral puncture site, their use is not without complications9 and closure failures with these devices still take place.10 In addition, their high cost has the potential to substantially impact cath labs and hospital budgets especially in developing countries , where their use is almost negligible. Here, manual compression offers a much cheaper way to deal with arteriotomies in the presence of cheap man-power. By and large, manually compressing 30 arteriotomies may cost 20 (< 1 dollar for each) U.S. dollars per day whereas it may cost more than 6000-9000 $ if VCDs are to be used our aim in this study was to identify the timing and predictors of HD after manual compression of femoral arteriotomies performed during cardiac catheterizations. Understanding these factors in our setting could have a major impact on reducing the frequency of this serious complication.
Methods
A prospective descriptive correlational design with comparative procedures for subgroups (with or without haematoma) were used to examine the relationship of several predetermined variables to haematoma development in patients undergoing manual compression to their puncture sites after diagnostic or interventional cardiac procedures performed via percutaneous transfemoral arterial approach. Data were collected during a one month period between the 28th of October 2007 and the 28th of November 2007, at King Abdullah University Hospital (KAUH), a 450 bed health care center that is affiliated to Jordan University of Science and Technology (JUST). KAUH is located in the North of Jordan and is the largest hospital providing both secondary and tertiary health care to the population living in this region, sub-serving an estimated population of 1.5-2 million. The staff of cardiologists working in Cardiac Catheterization Unit comprises of four consultants and five senior specialists.
Inclusion criteria were adult patients (more than 18 years old) undergoing diagnostic or interventional cardiac procedures performed via percutaneous transfemoral arterial approach using 6 French sheath and manual compression after sheath removal. Two hundred and fifty-two consecutive patients were initially included. Thirteen patients were later excluded due to the absence of complete data on the record form; insufficient time to obtain patient consent before the procedure; conversion of the case to urgent surgery; undergoing acute cardiac catheterization; and receiving fibrinolytic therapy 48 hours before the procedure. A total of 239 patients were included in statistical analysis, which is larger than the appropriate number of sample size calculated by Cohen formula11 (using a power level of 0.08, an alpha level of 0.05, a moderate effect size as an R2 of 0.13, and number of independent variables of 35 were selected. Given this, the appropriate sample size would be 227).
Data were collected and relevant information was recorded on a data collection form after its approval was granted by the appropriate Internal Review Boards (IRB). The investigation conforms to the principles outlined in the Declaration of Helsinki.
We adopted the data collection form developed by Andersen et al. with some modification.4 The original instrument was edited according to available medications and other different conditions at our setting. The permission for such modification was obtained from the corresponding author. The edited instrument was reviewed by one consultant cardiologist, one senior cardiovascular nurse, one senior nurse researcher, and one statistician.
After the end of the cardiac procedure, patients\' groins were prospectively examined and followed up by a research nurse or cardiac catheterization nurse for the development of haematomas. HD was assessed immediately after haemostasis was established (baseline), 6-8 hours and 12-16 hours post-haemostasis. After the removal of the femoral sheaths and using current hospital protocols, groin dressings were secured using elastic tapes with bi-directional stretch.
The edges of the dressing were elevated to make the assessment at 6-8 hours. If no problems were identified, the edges of the dressing were resealed. At 12-16 hours, the dressing was completely removed and the area reassessed. A "small adhesive bandage" was then applied to the puncture site.
Developing haematomas were assessed by two experienced nurses (one from the cardiac catheterization unit and the other from the coronary care unit), at least with 5 years of experience in the cardiac field. The registered nurse (RN) palpated the femoral arterial puncture site for swelling and observed the puncture site for bruising. The edges of the haematoma were outlined with a marking pen and the widest dimension was measured to the nearest centimeter.
For the purpose of the study, haematoma was defined as follows: an accumulation of blood at skin level with bruising or swelling in the area of the artery punctures during the period of sheath insertion through the 12-16 hours following sheath removal. (1) Small haematoma: haematoma measuring 2 to < 5 cm in diameter; (2) Large haematoma: haematoma measuring equal or greater than 5cm in diameter, while significant femoral haematoma was defined as >10 cm in diameter that required blood transfusion, surgery, or prolonged hospital stay. Blood transfusion was defined as the administration of whole blood or packed red blood cells within 7 days of cardiac cath.4
Thirty three prespecified variables to predict HD were chosen and analyzed using univariate analysis. Variables which proved to be significant were arranged in 4 models and were again analyzed using multivariate logistic regression using Omnibus and Wald statistical tests. The different variables tested are shown in Table-3.
Statistical Analysis was performed using SPSS statistical analysis programme (SPSS Inc, Chicago, Ill- version 13.0). Univariate analysis was done using c2 tests for categorical variables and t tests for continuous variables. Logistic regression was finally performed to predict the relation of haematoma development to several identified independent predictors that were significant through univariate analysis. A probability level <0.05 signifies statistical significance.
Results
Patient-related characteristics are summarized in Table-1.
Femoral haematomas developed in thirty nine patients among the 239 patients who constituted the study population leading to an overall frequency of 16.3 %. More than two-thirds (74.4%) of the haematomas occurred later in the bed unit while 20.5% developed earlier before leaving the cath lab. Only two patients (5%) developed femoral arterial haematomas during transfer from the cath lab to the floor or intensive care unit. Among haematomas developing in the bed unit, 59% (17 patients) developed after removal of the sheath, four of whom after ambulation. Meanwhile, most of the haematomas (80%) that developed in the cath lab did so before removal of the sheath. The sizes of haematomas were small (59%), large (36%) and huge>10cm (5%). Most of large (86%) and all huge haematomas occurred outside the cath lab. The details of haematoma characteristics can be seen in Table-2.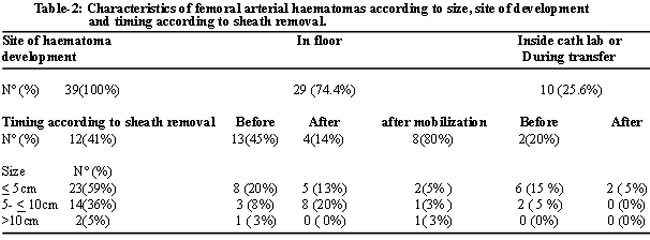
Among the 33 prespecified variables that were analyzed using univariate analysis, only 19 proved to be statistically significant. These 19 significant variables were then divided into a total of 4 models which were analyzed using multivariate logistic regression to test their ability to predict HD. The different variables tested can be seen in Table-3. 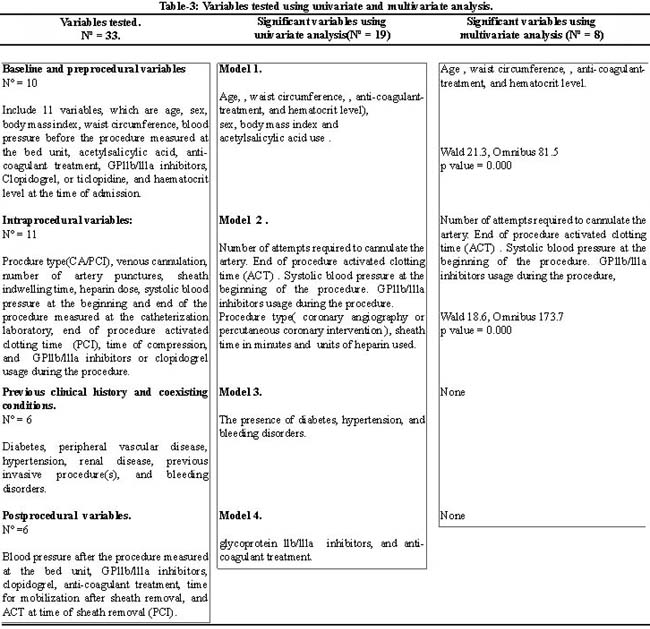
Model 1 tested seven baseline and preprocedural variables (Table-3). The model was restricted to those variables that made a significant contribution to the prediction of HD by univariate analysis. In this model, the model Omnibus tests of model coefficients was significant (c2=81.563, P=.000). Coefficient and Wald statistics for this model was also significant ( p= 0.000) .The prediction accuracy of this model was 5.8% greater than that of the null model, and the difference was statistically significant. Four variables from this model a) age, b) waist circumference, c) anticoagulation or low molecular weight heparin before procedure and d) haematocrit before procedure, were found to be predictive of HD.
Model 2 tested a set of seven intraprocedural variables (Table 3) . The predictive accuracy of this model was 10.7% greater than that of the null model, and the difference was statistically significant. The model Omnibus tests of model coefficients was significant (c2=173.771, P=0.000. Coefficient and Wald statistics for this model was also significant (p= 0.000). Four variables from this model were predictive of haematoma development upon multivariate analysis, these were high systolic blood pressure, number of attempts required to cannulate the artery, glycoprotein GPllb/llla inhibitors use during the procedure, and procedure activated clotting time.
Model 3 tested a set of three variables involving previous clinical history and coexisting conditions including the presence of diabetes, hypertension, and bleeding disorders. Bleeding disorders were defined as preexisting thrombocytopenia, anaemia, and von Willebrand disease.
Meanwhile, model 4 tested a set of two postprocedural variables . These variables were use of postprocedural glycoprotein llb/llla inhibitors, and anti-coagulant treatment. Models 3 and 4 did not reach statistical significance upon multivariate analysis; that is, none of these variables were predictive of HD.
Discussion
The scale for evaluating haematoma formation was originally developed by Christenson et al. in 1976.12 In 1995, it was modified by Hogan-Miller et al by adding measurement of the haematoma in centimeters.13 Variations of this instrument have been used in several studies1,12,14 although these researchers did not discuss the validity or reliability of these tools.
The frequency of haematoma in our series was 16.3% [< 5cm (9.6 %) , 5-10cm (5.9 %), and > 10cm (0.8%)] which corresponds with reports from similar previous studies.4,15
Large haematomas, in 14 (5.9%) patients were the cause of severe hypotension and requirement for blood transfusion. Hospitalization was extended by 3 days.
Twenty three patients developed small haematomas, of whom 17 (74%) patients were hospitalized overnight for observation. Although these complications did not result in any subsequent vascular compromise, they did account for delayed hospitalization.
Interestingly, 41% of the haematomas that were reported later in bed units developed before sheath removal. These findings mandate more emphasis on patient\'s education, to teach them how to report immediately, any suspicious symptoms for haematoma formation which includes any swelling, oozing, hotness, numbness, or pain at or around the puncture site.
In addition, arranging teaching cessions for ward nurses regarding the haematoma detection, and management may also have a role in decreasing the frequency of HD. Increasing the number of ward nurses could also be recommended, but this could increase the economic burden.
On the other hand, only one quarter of the haematomas developed earlier in the cath lab, and especially before sheath removal, suggested that these are related to patient as well as procedural characteristics. As a strict cath lab policy, new nurses should not practice manual compression of the access site.
In the present cohort, 79.5% of the 39 patients who developed haematomas, had a haematocrit level below 0.40, a finding consistent with a previous study.2 In addition, older age was also predictive of HD in our series as shown in several previous studies.16-19
Waist circumference an indicator for abdominal obesity,20 was a significant predictor for haematoma by logistic regression. Our results were in keeping with many previous studies indicating that obesity confers increased risk for vascular access complications.3,17,21 Obese patients present many challenges such as difficulty in palpating the pulse and gaining femoral arterial access, and may be difficult to compress because of the depth of the artery to achieve postprocedural haemostasis. This can also cause a delay in the recognition of poor haemostasis. Obese patients should be very carefully monitored, because haematomas may not be detected until blood loss becomes significant.21
Our results revealed that systolic blood pressure (SBP) above or equal to 160 mmHg measured at the beginning of the invasive cardiac procedure is a significant predictor for HD. This is in keeping with previously reported studies.17,22,23 Haematoma development in our study was related to multiple artery punctures, which is consistent with many previous studies.4,18
In the current study, preprocedural treatment with low molecular weight heparin (LMWH) and intraprocedural use of glycoprotein IIb/IIIa inhibitors were considered as predictors for HD, similar to findings of Andersen et al.4 Better dosing of these agents and adjunctive heparin is recommended.24,25 Longer activated clotting time (ACT) values at the end of the interventional procedure was also a significant predictor of HD which has been reported in other studies.1,7,21
Timing and predictors of HD could help in developing and application of risk-reducing strategies that could limit the morbidity and the increased expenses associated with this adverse outcome.
High systolic blood pressure, advanced patient age, low hematocrit levels, high waist circumference, preprocedural usage of anti-coagulant-treatment and intraprocedural GPllb/llla inhibitors use and end of procedure activated clotting time (ACT) are important factors for admission assessment. It may be appropriate to design a screening tool or scoring system that would assist in the identification of patients with these risk factors before performing the transfemoral percutaneous arterial puncture in the cardiac catheterization lab.
Acknowledgments
We would like to acknowledge Dr. Amjad Al-Naser for his assistance in statistical analysis. We would like to acknowledge and express our appreciation to our nurse colleagues in the cardiac catheterization laboratory at KAUH, for their support in data collection.
References
1.Benson LM, Wunderly D, Perry B, Kabboord J, Wenk T, Birdsall B, et al. Determining best practice: comparison of three methods of femoral sheath removal after cardiac interventional procedures. Heart Lung 2005; 34: 115-21.
2.Hamner JB, Dubois EJ, Rice TP. Predictors of complications associated with closure devices after transfemoral percutaneous coronary procedures. Crit Care Nurse 2005; 25: 30-7.
3.Berry C, Kelly J, Cobbe SM, Eteiba H. Comparison of femoral bleeding complications after coronary angiography versus percutaneous coronary intervention. Am J Cardiol 2004; 94: 361-3.
4.Andersen K, Bregendahl M, Kaestel H, Skriver M, Ravkilde J. Haematoma after coronary angiography and percutaneous coronary intervention via the femoral artery frequency and risk factors. Eur J Cardiovasc Nurs 2005; 4: 123-7.
5.Allie DE, Hebert CJ, Walker CM, Caputo RP. Manual compression may not be Benign. Endovasc Today 2003; 4: 42-6.
6.Petula SM, Hudacek S. The FemoStop compression device: utilization guidelines for the critical care nurse. Dimens Crit Care Nurs 1995; 14: 259-64.
7.Liew R, Lidder S, Gorman E, Gray M, Deaner A, Knight C. Very low complication rates with a manual, nurse-led protocol for femoral sheath removal following coronary angiography. Eur J Cardiovasc Nurs 2007; 6: 303-7.
8.Schiks I, Schoonhoven L, Verheugt F, Aengevaeren W, van Achterberg T. Performance evaluation of arterial femoral sheath removal by registered nurses after PCI. Eur J Cardiovasc Nurs 2007; 6: 172-7.
9.Nikolsky E, Mehran R, Halkin A, Aymong ED, Mintz GS, Lasic Z, et al. Vascular complications associated with arteriotomy closure devices in patients undergoing percutaneous coronary procedures: a meta-analysis. J Am Coll Cardiol 2004; 44: 1200-9.
10.Hermiller JB, Simonton C, Hinohara T,Lee D, Cannon L, Mooney M, et al. The StarClose Vascular Closure System: interventional results from the CLIP study. Catheter Cardiovasc Interv 2006; 68: 677-83.
11.Munro BH. Statistical methods for health care research. 5th ed. New York: Lippincott Williams and Wilkins 2005; 261.
12.Christenson R, Staab EV, Burko H, Foster J. Pressure dressings and postarteriographic care of the femoral site. Radiology 1976; 119: 97-9.
13.Hogan-Miller E, Rustad D, Sendelbach S, Goldenberg I. Effects of three methods of femoral site immobilization on bleeding and comfort after coronary angiogram. Am J Critic Care 1995; 4: 143-8.
14.Barkman A, Lunse CP. The effect of early ambulation on patient comfort and delayed bleeding after cardiac angiogram: a pilot study. Heart Lung 1994; 23: 112-7.
15.Schickel SI, Adkisso P, Miracle V, Cronin SN. Achieving femoral artery haemostasis after cardiac catheterization: a comparison of methods. Am J Critic Care 1999; 8: 406-9.
16.Piper WD, Malenka DJ, Ryan TJ Jr, Shubrooks SJ Jr, O\'Connor GT, Robb JF, et al. Predicting vascular complications in percutaneous coronary interventions. Am Heart J 2003; 145: 1022-9.
17.Waksman R, King SB 3rd, Douglas JS, Shen Y, Ewing H, Mueller L, et al. Predictors of groin complications after balloon and new-device coronary intervention. Am J Cardiol 1995; 75: 886-9.
18.Hillegass WB, Brott BC, Chapman GD, Phillips HR, Stack RS, Tcheng JE, et al. Relationship between activated clotting time during percutaneous intervention and subsequent bleeding complications. Am Heart J 2002; 144: 501-7.
19.Bailes BK. Perioperative care of the elderly surgical patient. AORN J 2000; 72: 186-207.
20.Health Canada, Canadian Guidelines for Body Weight Classification in Adults, 2003. Office of Nutrition Policy and Promotion Health Canada. Ottawa, Ontario.
21.No author listed. Use of a monoclonal antibody directed against the platelet glycoprotein IIb/IIIa receptor in high-risk coronary angioplasty. The EPIC Investigators. N Engl J Med 1994; 330: 956-61.
22.May O, Schlosser H, Skytte L. A high blood pressure predicts bleeding complications and a longer hospital stay after elective coronary angiography using the femoral approach. J Interv Cardiol 2009; 22: 175-8.
23.Satler L. Reducing complications of femoral access. Catheter Cardiovasc Interv 2008; 71: 524-5.
24.Dougherty KG, Gaos CM, Bush HS, Leachman DR, Ferguson JJ. Activated clotting times and activated partial thromboplastin times in patients undergoing coronary angioplasty who receive bolus doses of heparin. Cathet Cardiovasc Diagn 1992; 26: 260-3.
25.Topol EJ. Toward a new frontier in myocardial reperfusion therapy: emerging platelet preeminence. Circulation 1998; 97: 211-8.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




