Khadija Nuzhat Humayun ( Department of Pediatrics and Child Health, Aga Khan University, Karachi, Pakistan. )
Taimur Saleem ( Medical College, Aga Khan University, Karachi, Pakistan. )
Umair Khalid ( Medical College, Aga Khan University, Karachi, Pakistan. )
Fyezah Jehan ( Department of Pediatrics and Child Health, Aga Khan University, Karachi, Pakistan. )
Sajid Soofi ( Department of Pediatrics and Child Health, Aga Khan University, Karachi, Pakistan. )
June 2010, Volume 60, Issue 6
Case Reports
Abstract
Existing standard treatment options for visceral leishmaniasis are less than optimal. We report here the use of oral miltefosine in the treatment of two paediatric cases of visceral leishmaniasis at a tertiary care hospital in Karachi, Pakistan. One patient came from Balochistan while the second patient was from Northern Pakistan. Both presented with a prolonged history of fever, massive hepatosplenomegaly, anaemia and thrombocytopenia. Visceral leishmaniasis was diagnosed with bone marrow studies. Amphotericin B was first started in the first patient; however severe hypokalaemia and allergic reaction occurred. Oral miltefosine was then administered. The child showed clinical improvement with regards to signs of leishmania infection but succumbed to a nosocomial infection during the hospital stay. In the second patient, miltefosine was started in the first instance. He showed remarkable clinical improvement. At 2 months follow-up, the child showed adequate weight gain along with successful resolution of hepatosplenomegaly and fever. Miltefosine has the potential to be considered a first line therapy for visceral leishmaniasis in developing countries; however larger studies are warranted to validate the trends observed in this small case series.
Introduction
Visceral leishmaniasis (VL) or \'Kalazar\' is a serious parasitic infection of the reticuloendothelial system. Unless promptly managed, the disease can result in significant morbidity and mortality especially in the endemic areas.1 Drug resistance has become a major impediment in the effective treatment of VL. Miltefosine has produced remarkable results in the last few years and is now rapidly emerging as an efficacious and cost effective agent in the treatment of VL. However, there are no reports of the successful use of miltefosine for the treatment of VL in Pakistan. Here, we report the first instance of the treatment of two cases of VL with oral miltefosine in Pakistan.
Case One:
An 8 years old boy from Balochistan province of Pakistan presented with 11 months history of intermittent high grade fever, night sweats, severe anorexia, vomiting and weight loss. He had been empirically treated for malaria and enteric fever multiple times with no response to any of these therapies. On examination, he was severely malnourished, cachectic and extremely pale. His abdomen was distended with massive hepatosplenomegaly (Figure-1).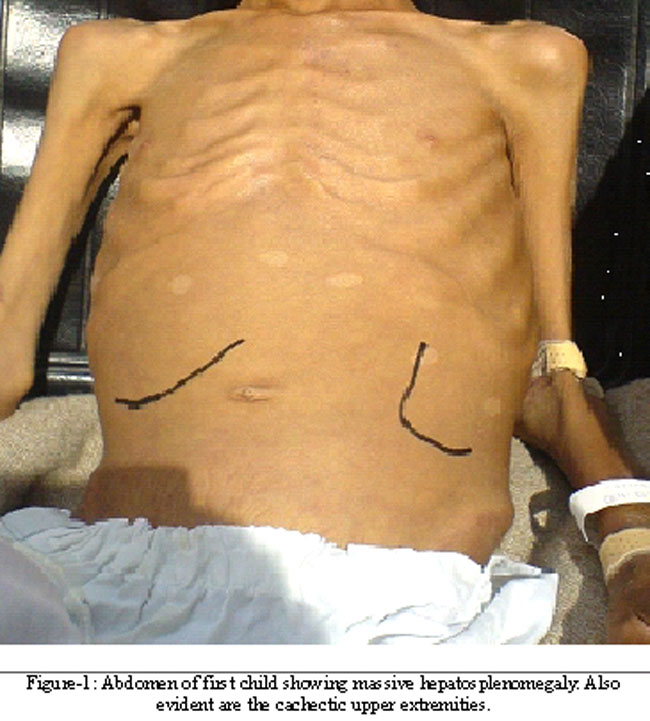
Laboratory investigations showed anaemia (haemoglobin=6 g/dl) and thrombocytopenia (platelets=60,000/L). Bone marrow smear showed Leishmania Donovani bodies (Figure-2)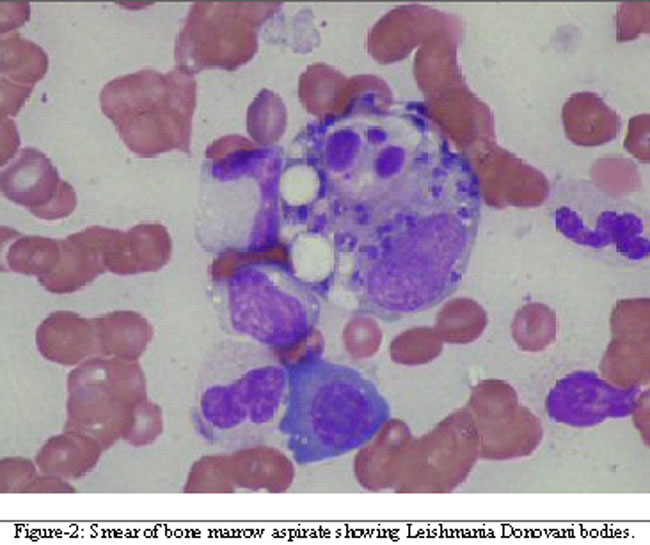
and a diagnosis of VL was made. The patient was initially treated with amphoterecin B. However, the incidence of adverse effects such as severe hypokalaemia, thrombocytopenia and allergic reactions dictated the discontinuation of this drug. Miltefosine was specially arranged for this patient through a request to the manufacturer as it was not available locally at that time. He was started on a dose of 2.5mg/kg/day and showed successful response to therapy.
His body temperature was the first parameter to normalize followed by improvement in appetite and weight gain within 1 week of treatment. The child continued to show clinical improvement with respect to signs and symptoms of VL but unfortunately contracted a nosocomial infection during the hospital stay and succumbed to it.
Case Two:
A one year 8 months old boy from northern Pakistan presented with a 6 month history of high grade intermittent fever, progressive abdominal distension, anorexia and weight loss. During his treatment at two other cities of Pakistan, he had received multiple courses of antibiotics and antituberculous therapy without success. On examination, his height and weight were below the 5th percentile for age. Abdominal examination revealed massive hepatosplenomegaly. His laboratory investigations showed anaemia (haemoglobin= 6.9g/dl), thrombocytopenia (platelets=80,000/L) and a raised erythrocyte sedimentation rate of 60 mm/hr. A bone marrow trephine was done which helped clinch the diagnosis of VL by demonstration of Leishmania Donovani bodies. This patient was treated in the first instance with oral miltefosine at a dose of 2.5mg/kg/day. He was discharged on 5th day of therapy to complete a total of 28 days of treatment on an outpatient basis. Subsequent follow-ups at 2 weeks and 2 months showed adequate weight gain along with successful resolution of hepatosplenomegaly and fever.
Discussion
Protozoal organisms of Leishmania Donovani complex, the causative agent of VL, are acquired through sandfly bites. Untreated cases have a high mortality and morbidity. An estimated 500,000 cases of VL occur annually with 90% of the cases occurring in five countries including India, Bangladesh, Nepal, Sudan and Brazil. The paediatric population is especially affected by this disease.1 In Pakistan, the disease is endemic in Northern Pakistan and Azad Jammu and Kashmir.2 Important confounding diseases include tuberculosis, enteric fever and malaria because of their high prevalence in areas where VL is endemic.
For years, the standard treatment for VL consisted of daily injections of pentavalent antimony compounds for 28 days. However, this therapy is fraught with many pitfalls such as reduced efficacy due to drug resistance, difficulties of administration and increasing frequency and severity of adverse events such as acute pancreatitis, myalgias, arthralgias and arrhythmias. Alternatives such as amphotericin B and pentamidine also have their fair share of shortcomings. Pentamidine has been virtually abandoned because of an unfavorable constellation of serious toxicity, inconvenient scheduling of administration, and no efficacy advantage. This had led physicians to rely predominantly on amphotericin B and liposomal amphotericin B for the treatment of VL. However, increased frequency of adverse reactions such as electrolyte derangements and anaphylaxis, necessity of hospital stay for IV injections, problems with availability and greater cost preclude its use in endemic areas; many of which form part of the developing world.1,3,4
Developed initially as an anti-neoplastic agent, miltefosine is currently the only oral treatment available for VL. Multiple trials have shown that oral miltefosine is an overall highly effective treatment for VL both in adults and in children. In Pakistan, miltefosine is a newly registered pharmaceutical compound. To date, only one non-randomized comparative clinical trial has been conducted in Pakistan to compare the efficacy of miltefosine and pentavalent antimony compounds in patients with cutaneous Leishmaniasis.4
For developing countries, miltefosine may prove to be more advantageous in the treatment of VL. In the face of increasing costs incurred from health care and associated paraphernalia, affordability is a major constraint in the treatment of many ailments. Standard agents used in the treatment of VL may not be costly on their own but due consideration needs to be given to associated expenses incurred. Although the total cost of amphotericin B for a 20-day treatment regimen in a 15 kg child is around US $50 (1 US $ = 83.3 Pakistani rupees), other added costs that must be considered include hospital stay for administration of the medication, possible need for pre-medication with antihistamines and steroids, frequent laboratory testing to check renal function, platelets and electrolytes, administration of fluids and high risk of nosocomial infections. Hence, the total expenditure is multiplied manifold. In contrast, a 28-day cost of miltefosine is currently around US $145; however this can be administered on an outpatient basis without the above mentioned adjunct expenses. This is an extremely important consideration for the paediatric patient population in many developing countries where health care expenditure on children may not, unfortunately, be a priority. In addition, the child is not unnecessarily exposed to the risk of nosocomial infections. From this perspective, miltefosine may provide a cost effective alternative to currently standard agents.
Although few studies exist on this compound, miltefosine is generally well-tolerated with minimal and largely reversible side effects such as gastrointestinal intolerance. Vomiting and diarrhoea generally lasts only one to two days. A few patients have also had reversible nephrotoxicity and hepatotoxicity with this drug but this is mild-to-moderate. Miltefosine may also increase the risk of other infections and should be used with caution in immunosuppressed individuals. Because of its potential to cause teratogenicity and abortion, miltefosine should not be given to women of child-bearing age unless pregnancy has been excluded and rigorous contraceptive precautions are taken during treatment and for 8 weeks after the completion of treatment.1,5-8
We turned to miltefosine in the first case because of the adverse effects experienced with amphotericine B while our second patient was treated with oral miltefosine in the first instance. One of our patients succumbed to a nosocomial infection in the latter part of his hospital stay. Although acquisition of this severe infection may well be attributable to the generalized ill health of the child resulting from the prolonged illness for 11 months; we can\'t entirely rule out this event as an adverse effect of miltefosine.
However, we were able to ascertain a gradual response to miltefosine in the treatment of VL in him as judged by improvement in signs and symptoms of leishmaniasis. We didn\'t encounter any adverse effects of miltefosine during the treatment of VL in the second patient.
Conflict of Interest:
The authors declare no conflict of interest.
References
1.Bhattacharya SK, Jha TK, Sundar S, Thakur CP, Engel J, Sindermann H, et al. Efficacy and tolerability of miltefosine for childhood visceral leishmaniasis in India. Clin Infect Dis 2004; 38: 217-21.
2.Altaf C, Ahmed P, Ashraf T, Anwar M, Ahmed I. Childhood visceral leishmaniasis in Muzaffarabad, Azad Jammu and Kashmir: Frequency and response to treatment in 61 cases. J Pak Med Assoc 2005; 55: 475-8.
3.Sundar S, Rai M, Chakravarty J, Agarwal D, Agrawal N, Vaillant M, et al. New treatment approach in Indian visceral leishmaniasis: single-dose liposomal amphotericin B followed by short-course oral miltefosine. Clin Infect Dis 2008; 47: 1000-6.
4.Rahman SB, Bari AU, Mumtaz N. Miltefosine in cutaneous Leishmaniasis. J Coll Physicians Surg Pak 2007; 17: 132-5.
5.Vanlerberghe V, Diap G, Guerin PJ, Meheus F, Gerstl S, Van der Stuyft P, et al. Drug policy for visceral leishmaniasis: a cost-effectiveness analysis. Trop Med Int Health 2007; 12: 274-83.
6.Herwaldt BL. Miltefosine - the long awaited therapy for visceral leishmaniasis? N Engl J Med 1999; 341: 1840-2.
7.Berman JD. Human leishmaniasis: clinical, diagnostic, and chemotherapeutic developments in the last 10 years. Clin Infect Dis 1997; 24: 684-703.
8.Sundar S, Jha TK, Thakur CP, Engel J, Sindermann H, Fischer C, et al. Oral miltefosine for Indian visceral leishmaniasis. N Engl J Med 2002; 347: 1739-46.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




