Farrukh Azmi ( Department of Immunology and Microbiology, Public Health Division, National Institute of Health, Islamabad. )
Mubashir A. Khan ( Central Research Centre, Pakistan Medical Research Council, National Institute of Health, Islamabad. )
Saeed Tariq ( Department of Immunology and Microbiology, Public Health Division, National Institute of Health, Islamabad. )
Jamshaid Iqbal ( Department of Immunology and Microbiology, Public Health Division, National Institute of Health, Islamabad. )
Abdur Rab ( Department of Immunology and Microbiology, Public Health Division, National Institute of Health, Islamabad. )
Abdul Ghafoor ( Department of Immunology and Microbiology, Public Health Division, National Institute of Health, Islamabad. )
July 1987, Volume 37, Issue 7
Original Article
Abstract
Two hundred and fourteen human faecal specimens were examined for detecting the presence of rotavirus by direct electron microscopy, antibody capture electron microscopy and ELISA. Twenty nine samples were positive with one of the three methods. Thirtyeight percent were positive with all three methods, 72.4% with ELISA, 62% with antibody capture electron microscopy and 55% with direct electron microscopy. ELISA was found to be the most sensitive method in our experience (JPMA 37: 172, 1987).
INTRODUCTION
Rotavirus is recognized as a major cause of nonbacterial diarrhoea particularly in young children1,2,3 It has also been implicated as an aetiological agent of diarrhoea in adults4.
Since this virus cannot be easily cultivated5, lab oratory diagno sis of rotavirus gastro enteritis is based on the direct detection of virus particles in clinical specimen by Electron Microscopy (EM)6, Enzyme Linked Immunosorbent Assay (ELISA) 7,8,Radioimmunoassay (RLA) 9 and Latex Agglutintion10
The present study was conducted to compare direct EM, ELISA and antibody capture EM techniques for the detection of rotavirus in the faeces of children with acute diarrhoea.
MATERIALS AND METHODS
Clinical Specimens: During October 1985 to April 1986, 214 samples were collected at local hospitals from diarrhoeic children under three years of age. For EM and antibody capture technique, specimens were processed on the same day and for ELISA, frozen at-20°C. All the specimen were analysed using the following techniques.
Clarification of Faecal Samples: A 20% suspension of faeces was made in PBS, centrifuged at 1 ,000g for 10 minutes and then at 10,000 g for 20 minutes.
Direct Electron Microscopy: One drop of clarified faecal sample was added to one drop of 2% Phosphotungstic Acid (PTA) on glass slide, a carbon coated grid was placed on the drop with coated surface toward the drop and left for 30 seconds, excess fluid removed by dabbing with a strip of blotting paper. The grid was examined with the Jeol JEM-100 Cxl 1 electron microscope for ten minutes.
Antibody Capture Electron Microscopy (ACEM): Bovine rotavirus antiserum (Welicome Diagnostics-England) was diluted 1:100 in PBS. Carbon coated grids were adsorbed with the antiserum for one hour at 37°C in a moist chamber. Excess fluid was blotted off and grids washed with PBS.~ Faecal suspension was incubated on the coated grids for one hour at 4°C, excess fluid was blotted off and grids washed with PBS x 3. The grids were stained with 2% PTA and examined with the Jeol JEM—100 Cxll electron microscope.
EUSA W.H.O. rotavirus ELISA kit was used according to the manufacturers’ instructions.
RESULTS
Results obtained by the three techniques used in this study are shown in Table.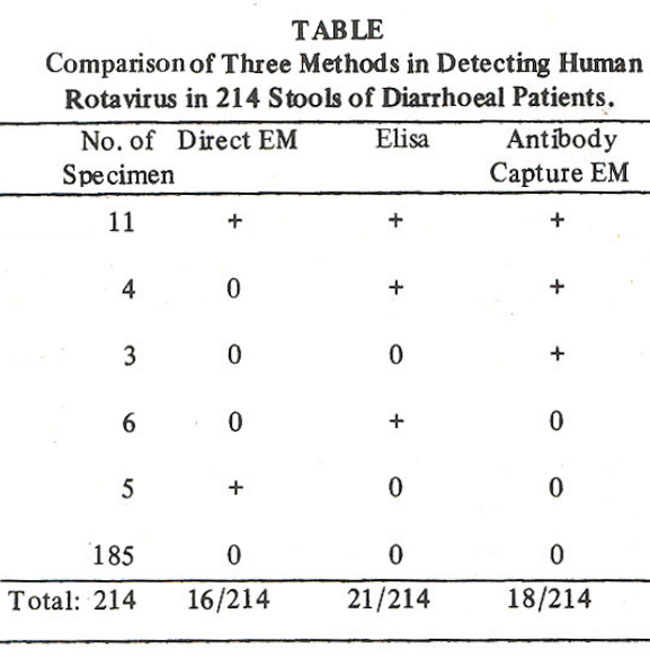
185 faecal specimens were negative by all three methods. Sixteen were positive by EM and 18 by ACEM. Using ELISA procedure, viral antigens were detected in 21 samples. The relative sensitivity of the different methods was 55% for EM (16/29), 62% for antibody capture (18/29) and 72% for ELISA (21/29). Six specimens were positive by
ELISA only (negative by other methods), five by EM alone. Nine cases were negative by ELISA, l3by EM and 11 byACEM.
DISCUSSION
Direct EM, ELISA and ACEM techniques are all useful in the detection of rotaviruses from diarrhoea cases. Direct EM of human stools is a well established test for the detection of rotavirus11 and though least sensitive, is a method for rapid detection of faecal viruses. Sensitivity of the EM may be related to many factors like size of the virus, purity and concentration of faecal extract, wetting properties of the grid used, time elapsed for searching the virus and skill of the operator. The biggest advantage of the EM is that it can detect the other viruses. However, it is not suitable for routine examination of large number of the specimens. In this study 16/214 cases were found positive by EM. One hundred ninety eight specimens were negative by this method for one of the reasons stated above. Five samples were negative by ELISA and ACEM, but were positive by direct EM. This could be due to these viruses being antigenically distinct from classical rota virus; they could be para rotavirus Similar results have been reported by others12,13,14. Inspite of its limitations, direct EM technique remains a valuable screening test.
We found ELISA to be the most sensitive method; it revealed 72% positive faecal specimens. Similar findings were reported by other workers15,16,17 Six samples were positive with ELISA alone, which could be false positive results as we did not perform confirmatory ELISA, or the rotãvirus particles were broken and could only be detected by ELISA12,13,14 ELISA has an edge over EM as it can be used for screening a large number of faecal specimens. ELISA can also be used in laboratories where EM is not available.
ACEM revealed the presence of rotavirus in 18/214 positive specimen, 3/214 samples were positive only with antibody capture EM presumably because of the viral particles being presant in very low number which were trapped by antibodies only. The serological trapping of virus was simple and rapid and required less examination time18. We did not find antibody capture technique as sensitive as ELISA and this may need further evaluation.
In conclusion, our comparison of three methods for detection of human rotavirus has shown ELISA as the most sensitive technique under our conditions.
ACKNOWLEDGEMENT
The authors are grateful to Mr. Ikram ul Haq for his assistance.
REFERENCES
1. Blacklow, N.R. and Cukor, G. Viral gastroenteritis. N. Engi. J. Med., 1981; 304 : 397.
2, Ithan, M.M.A., Khan, M.A., Burney, M.I. and Ghafoor, A. Aetiology of infantile gastroenteritis. Pakistani. Med. Res., 1982;21 :40.
3. Steinhoff, M.C. Rotavirus ; the first five years. J. Pediatr., 1980;96:611.
4. Echeverria, P., Blacklow, N.R., Cukor, G.G., Vibulbandhitkit, S., Changchawalit, S. and Boonthai, P. Rotavirus as a cause of severe gastroenteritis in adults. J. Clin. Microbiol., 1983; 18:663.
5. ward, R.L., Knowlton, D.R. and Pierce, M.J. Efficiency of human rotavirus propagation in cell culture. J. Clin. Microbiol., 1984; 19: 748.
6. Riepenhoff-Talty, M., Barrett, H.J., Spada, B.A. and Ogra, P.L. Negative staining and immune electron microscopy as technique for rapid diagnosis of viral agents. Ann. N.Y. A cad. Sci., 1983;420:391.
7. Bradburne, Enzyme-linked immunosorbent assay for the detection of human rotavirus in stool. J.Virol. Methods, 1983;7:65.
8. Chernesky, M., Castriciano, S., Mahony, J. and Delong, D. Examination of Rotazyme. II Enzyme Immunoassay for the diagnosis of rotavirus gastroenteritis. J. Clin Microbiol., 1985; 22: 462.
9. Middleton, P. J., Hokiaway, M.D., Petric, M., Syzmanski, T. and Tam, J.S. Soil! phase radioimmunoassay for the detection of rotavirus, Infect. Immun., 1977; 16: 439.
10. Doern, G.V., Herrmann, J.E., Henderson, P., Stobbs-Warlo, D., Perron, D.M. and Blacklow, N,R. Detection of rotavkus with a new polyclonal antibody enzyme immunoassay (Rotazyme II) and a commercial latex agglutination test (Rotalex); comparison with a monoclonal antibody enzyme iminunoassay. J. Cim. Microbiol., 1986; 23:226.
11. Cheung, E.Y., Hnatko, S.I Gunning, H. and Wilson, J. Comparison of Rotazyme and direct electron microscopy for detection of rotavirus in human stools. J. Clin. Microbiol., 1982; 16:562.
12. Brandt, C.D., Kim, H.W., Rodriguez, W.J., Arrobio, J.O., Jaifries, B.C., Stallings, E.P., Lewis, C., Miles, A.J., Chanock, RM., Kapikian, A.Z. and Parrott, R.H. Pediatric viral gastroenteritis during eight years of study. J. CJin. Microbiol., 1983;18: 71.
13. Nicnlas, J.C,, Cohen, J., Fortier, B., Lourenco, M.H. and Brioout, F. Isolation of a human pararotavirus. Virology, 1983; 124:181.
14. Rodger, S.M., Bishop, R.F. and Holmes, I.H. Detection of a rotavirus-like agent associated with diarrhoea in an infant. J. Clin. Microbiol., 1982;16 :724.
15. Brandt, CD., Kim, H.W., Rodriguez, WJ., Thomas, L., Yolken, R.H., Arrobio, J.O., Kapikian,A.Z., Parrott, R.H. and Chanock, R.M. Comparison of direct electron microscopy, immune electron microscopy, and rotavirus enzymelinked Immunosorbent assay for detection of gastroenteritis viruses in thildren. J. Clin. MicrobioL,1981;13 :976.
16. Hovi, T., Vaisanen, Ukkonen, P. and Von Bonsdorff C.H. Solid-phase enzyme immunoassay for rotavirus antigen; faecal protease activity as a reason for false-negative results. J. Virol. Methods, 1982;5 :45.
17. Morinet, F., Ferchal, F., Colimon, R. and Perol, Y. Comparison of six methods for detecting human rotavirus in stools. Eur. J. Clin. Microbiol., 1984:3:136.
18. Nicolaieff, A,, Obert, G. and van Regenmortal, M.H. Detection of rotavirus by serological trapping on antibodycoated electron microscope grids. J. Clin Microbiol., 1980; 12: 101.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




