Gul Nayyar ( Department of Paediatrics (Medicine), National Institute of Child Health, Jinnah Postgraduate Medical Centre, Karachi-35. )
Afroze Ramzan ( Department of Paediatrics (Medicine), National Institute of Child Health, Jinnah Postgraduate Medical Centre, Karachi-35. )
Mushtaq A. Khan ( Children Hospital, Islamabad Medical Complex, Islamabad. )
A. Ghaffar Billoo ( Department of Paediatrics (Medicine), National Institute of Child Health, Jinnah Postgraduate Medical Centre, Karachi-35. )
July 1987, Volume 37, Issue 7
Original Article
Abstract
The acceptability and the efficacy of flavoured vs non flavoured ORS was tested in 112 dehydrated children. The two solutions were equally effective in correcting the initial dehydration but non-flavoured ORS was a better and safer solution for the maintenance therapy. (JPMA 37: 1 67, 1987).
INTRODUCTION
Oral Rehydration Salt or ORS (WHO formula) is universally recommended for dehydration due to diarrhoea. ORS is widely accepted as a most effective, cheap and easily administered therapy for mild to moderately dehydrated cases of infantile diarrhoea1,2 ORS (WHO formula) is non-flavoured, addition of commonly used flavours might increase acceptability by the children and help achieve early rehydration or it may lead to excess consumption and over-hydration. Acceptability/efficacy of flavoured vs non-flavoured ORS should therefore be tested. This clinical trial was done to compare the acceptability of flavoured vs non-flavoured ORS (WHO formula) in mild to moderately dehydrated cases of infantile diarrhoea during initial rehydration and maintenance therapy.
MATERIAL AND METHODS
A prospective study was done at the National Institute of Child Health Karachi in December, 1985-March, 1986. One hundred twelve children between 1-24 months of age having diarrhoea with mild to moderate dehydration were included in this study. A detailed history was taken, clinical examination done and degree of dehydration and nutritional status assessed and recorded on a specially designed proforma. Blood samples for electrolyte and urea were taken on admission and after 24 hours. Patients were divided into two groups, A and B. The first patient that attended the Oral Rehydration Therapy Unit with diarrhoea every day was alternately assigned to group A or group B.
Group A: ORS: (WHO formula).
Group B: Flavoured ORS: (WHO formula) flavouring agent (lemon ÷ lime). (“ORASAL.F” Wilson’s Pharmaceuticals was used in the study).
Amount of ORS required for rehydration was calculated by multiplying the admission weight in kilograms by percentage of dehydration based on the clinical assessment3 (This amount was expected to be taken by the infant within 4 hours). Both groups were offered the respective ORS as specified above, ad-libitum and/or every 10 minutes. If either the non flavoured or flavoured ORS was refused on three consecutive offerings the alternate ORS was offered. Treatment was explained to the mothers, who administered ORS with cup and spoon, under supervision. Breast feeding was continued or 1/2 strength formula milk feeding was started after initial rehydration depending upon the type of feeding the baby was already taking. Patients were constantly supervised and periodically re-assessed. Complete intake and output charts were maintained and final assessment done after 24 hours progress of rehydration therapy was assessed after one hour, four hours and twenty four hours, for the following:
The presence of, and change in the signs of dehydration.
Whether the rehydration fluid was being given successfully and in adequate amounts.
CRITERIA USED FOR ASSESSMENT OF ACCEPT ABILITY
Good: Acceptance was considered good if 70-100% of the required amount was ingested during the initial rehydration period of 4 hours.
Average: Acceptance was consklered average if fluid intake was 50-70% of the calculated amount during initial vrehydration period of 4 hours.
Bad: Refusal to drink the solution inthree consecutive offerings at 10 minute intervals, or vomiting in two successive intakes, or 50% intake in first 4 hours, or 25% intake within the Ist hour.
Any complication and signs of over-hydra tion were recorded.
RESULTS
One hundred and twelve children were included in the study, 50% (56) assigned to each group. The average age of group A was 8.59 months and that of group B 8.78 months (Table-I).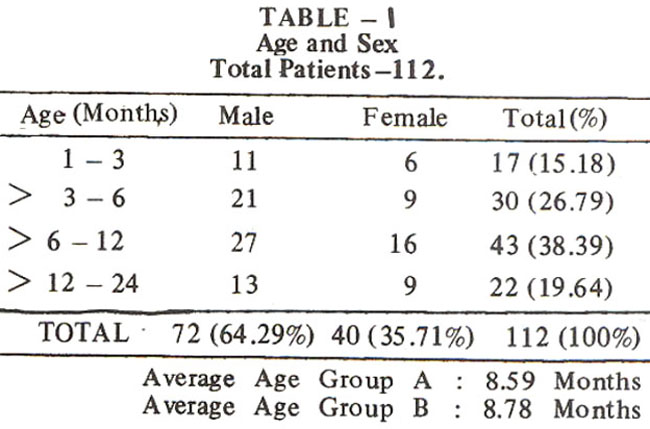
Results show that average time taken for rehydration was 3.34 hours for group A as against 3.09 for group B, which difference is negligible (Table II).
Almost 20% of the total group B patients developed generalized oedema or puffmess of face as compared to only 3.5% of group A (Table III, IV).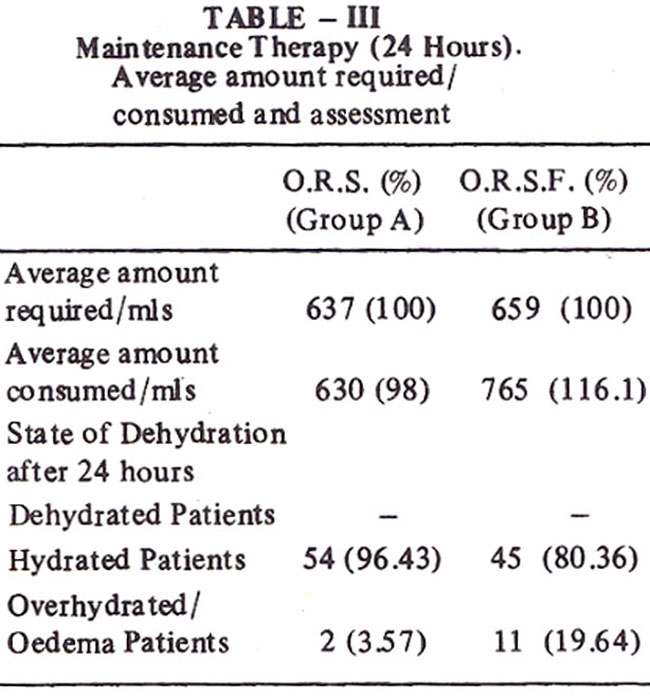
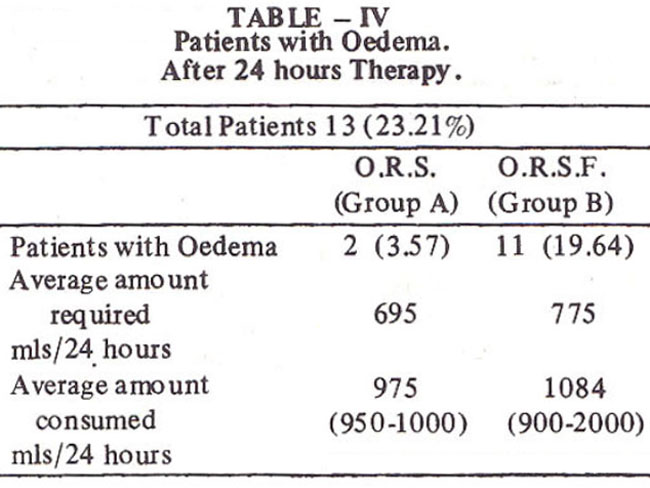
Serum sodium levels revealed that as many 21 .21% of flavoured ORS group B patients developed hyponatraemia
As regards transfer of patients from one group to the other, 4 patients from each group had to be transferred onto the other solution and they accepted the other fluid well excepting for two patients who did not like either of the two solutions.
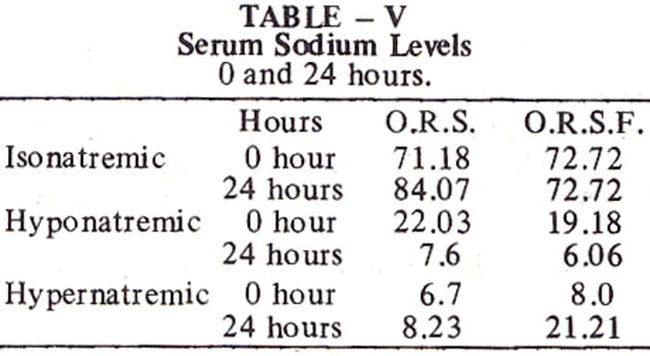
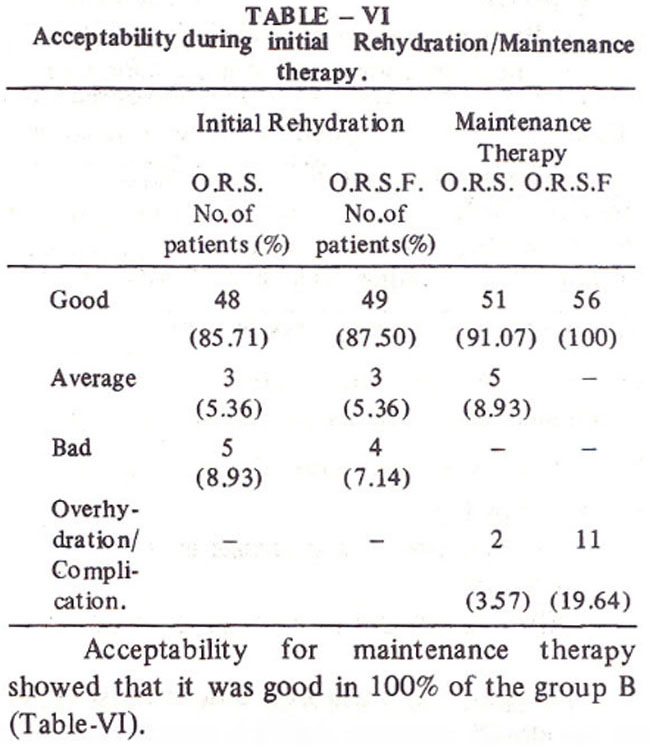
Table VI shows that as far as acceptability for initial rehydration was concerned, it was good in almost 86% of group A and 87% of group B. No signs of overhydration during initial rehydration were detected.
DISCUSSION AND CONCLUSIONS
Results of our study are in contrast to the study done by Carraza et al. which showed unflavoured ORT solution was significantly more acceptable than the flavoured solution. In their study cherry flavour ORT solution was used, while lemon and lime flavoured ORT solution (ORASOL-F by Wilson’s Pharmaceuticals) was used in our study.
Our study revealed that during initial rehydration, both the solutions had equally good acceptability and no significant preference was shown for either of the two solutions. However, during maintenance therapy, a marked preference was evident for the flavoured solution, which was consumed in much larger quantities.
The probable explanation appears to be that in dehydrated children thirst is the principal factor regulating intake of fluids and taste matters little;dehydrated children take any fluid.
However, during maintenance therapy, thirst having quenched, taste becomes a decisive factor. Accordingly flavoured solution was preferred and consumed in larger quantities with the disadvantage that there was greater incidence of overhydration and hypernatraemia in group B using the flavoured solution.
As we are advocating ORS as home therapy and use of flavoured ORS necessitates careful regulation of intake to obviate chances of overhydration and hypernatremia, mothers should be clearly advised to use ORS to replace only ongoing losses during maintenance phase and to give plain water and other fluid for normal daily requirements. Special emphasis should be laid upon continuing feeding during diarrhoea.
Conclusion to be drawn from our study appears to be that whereas either of the two solutions may be used during initial rehydration, non-flavoured ORS (WHO formula) definitely emerges as a better and safer solution during maintenance therapy.
REFERENCES
1. Pierce, N.F. and Hirschhorn, N. Oral fluid-8 simple weapon against dehydration in a diarrhoea. WHO Chron., 1977; 31 :87
2. Rohde, J.E., et al. Therapeutic interventions in diarrhoea. Food Nutr. Bull. (United Nations Unive.), 1981; 3: 34.
3. A manual for treatment of acute diarrhoea. WHO/CDD/SCR/80.2, 1980.
4. Carraza, F.R., et al. Comparative trial of the acceptability of two oral rehydration solutions in children with acute diarrhoea. Foiha Med.(Brazil), 1978;77:611.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




