Mobammad Abdur Rab ( National Institute of Health, Islamabad. )
Jamshaid Iqbal ( National Institute of Health, Islamabad. )
Farrukh Hameed Azmi ( National Institute of Health, Islamabad. )
Mohammad Arif Munir ( National Institute of Health, Islamabad. )
Mohammad Saleem ( Armed Forces Institute of Pathology, Rawalpindi. )
September 1989, Volume 39, Issue 9
Original Article
Abstract
Between January’85 and August’87, 22 cases of VL were seen at National Institute of Health, Islamabad. Three (14.6%) came from the previously known endemic region of Gilgit, 15 (68.1%) from different localities in Azad Jammu and Kashmir (AJ&K), and 4(1 7.3%) from neighbouring foci in NWFP and Punjab. Mean age of the patients was 4.2 years, (Range, 10 months to 57 years) median 2.5 years and mode 2 years. High levels of Leishmania antibodies were detected by Indirect Immunofluorescent Antibody Technique (IFAT) in all cases. Leishmania were isolated from bone marrow aspirates of 2 patients and isoenzyme characterization performed in one of these, the organism was typed as Leishmania infantum sensu stricto. Sera from 289 children residing in 5 endemic localities in AJ&K was tested for Leishmania specific antibodies by IFAT and low levels of these antibodies were detected in 15.4% of the cases (JPMA 39: 225, 1989).
INTRODUCTION
Human visceral leishmaniasis was first reported in Pakistan from the northern areas in the late fifties and early sixties1,2, the disease was thought to be restricted to foci in Gilgit and Skurdu. Since the advent of this decade, sporadic cases began to occur not only in different foci of AJ&K but also in some extreme north eastern areas of Punjab as well as nearby localities of NWFP3. The disease is acquired through the bite of infected phlebotomine sand flies. Amastigotes multiply within mononuclear phagocytes throughout the reticuloendothelial system including spleen liver, lymphnodes and bone marrow. Clinically, the disease is marked by intermittent fever, anaemia, lymphadenopathy and hepatosplenomegaly. Progressive weight loss, cachexia and malaise, followed by bacterial superinfections and can sometimes result in death. In general once hepatosplenomegaly occurs host defence mechanisms are not able to prevent further parasitic multiplication4. Patients. with VL during the acute stage have high levels of globulins and produce Leishmania-specific antibodies. Following successful treatment, these ant tibody levels decline and a gradual, cell mediated, delayed type hypersensitivity response to leishma nial antigen is elicited which manifests over several months, and resistance to infection usualiy develops5. Bone marrow aspiration has been used extensively for the diagnosis of VL. This is an invasive procedure and may lead to serious complications if performed by individuals not fully trained in this technique. IFAT, a relatively more sensitive procedure, is now widely used for the diagnosis and seroepidemiological studies of VL6. This pilot study was undertaken to detect leishmania antibody levels in patients and determine baseline antibody levels in endemic populations in AJ&K using IFAT.
MATERIALS AND METHODS
a) Selection of Localities:
Five villages in AJ&K (Total population, 2600) were selected for seroepidemiological survey. From these villages atleast 7 active cases had been reported in the past two year period. These villages are located in the vicinity of 3 major towns in AJ&K i.e. Rawalakot, Dirkot and Chikar. The houses in these villages are generally scattered along the hills in this region.
b) Collection of Sera for IFAT:
Sera was obtained from 22 patients admitted in different hospitals of Rawalpindi and Islamabad. .Sera samples were also collected from 289 children (under 12 years) residing in 5 localitjes mentioned above. Of these 289, venous blood was drawn from 90. From the remaining 189 children, 100 microliter blood samples were drawn by finger pricks and dried on No. 4 Whatman filter paper. These were appropriately numbered and stored at -20C Serum was eluted in 1 ml Phosphate buffered saline (PBS, pH 7.4) to give a starting dilution of 1 in 20.
c) Preparation of Antigen:
Leishmania was isolated from a case of VL and an invitro culture was maintainS in NNN medium. Promastigotes were harvested from this culture and after three washings in PBS, stored overnight at 4°C in 1% formol PBS. This was washed three times again with PBS and diluted to contain 30 to 40 parasites per high power field. One drop of this antigen was placed on each spot of a 12 well multispot slide (flow Laboratories). The antigen slides were air dried at room temperature, properly wrapped in aluminum foil and placed in self sealed polytbene bags, with a few crystals of silica gel. The slides were stored at -70°C till use.
d) Conjugate:
Commercially available antihuman IgG fluorescence isothiocyanate (FITC) conjugate obtained from Sigma, was used at a working dilution of 1:20 in PBS. At this dilution no fluorescence was observed in the negative control.
e) Test Procedure:
The antigen slides were brought to room temperature before use. 25 micro liter of test serum was applied to each antigen spot and allowed to react for 30 minutes at room temperature inamoist chamber. The slides were washed 3 times in PBS through serial changes of 10 minutes each. Antihuman IgG FITC conjugate was then applied and incubated for 30 minutes at room temperature in moist chamber. Washing procedure was repeated. The slides were mounted in a preparation of 90% glycerol in 03M Carbonate/Bicarbonate buffer (pH 9.0) and covered with coverslips. Both positive and negative control sera were used each time the test was performed.
f) Fluorescence Microscopy
Slides were examined with an Olympus fluorescent BH2 microscope using a dark field condenser and 400 X magnification. Light source was 12 volts, 50-60HZ halogen lamp.
g) Isoenzyme Characterization:
Culture on NNN medium was obtained from bone marrow biopsy of a two year old male patient admitted in a local hospital. Isoenzyme characterization on this sample was performed at aWHO recognized center, Instituto Superiore Di Santa in Rome, Italy.
RESULTS
Serology of Patients
All confirmed cases of VL demonstrated very high titers of Leishmania antibodies (Figure 1).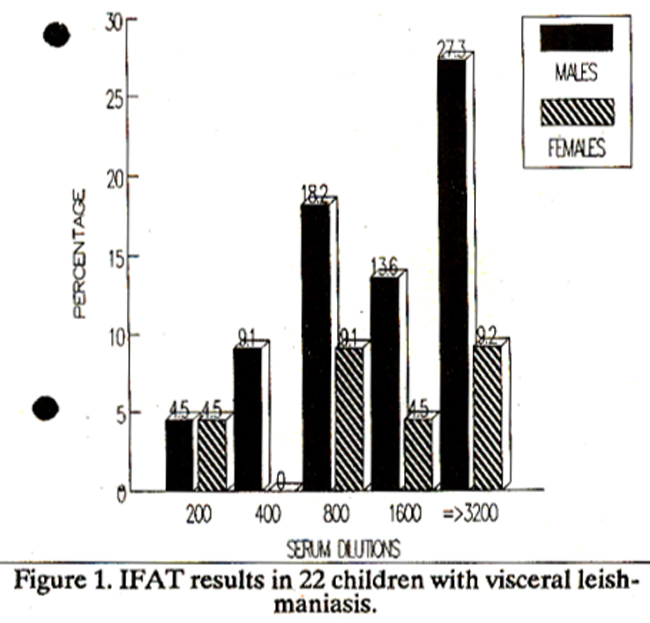
9.1% were positive at titers of 1:400, 27.2% at 1:800, 182% at 1:1600 and 36.4% had titers of 1:3200 or more. Serology of Children in Endemic Areas Of the 289 children examined (Figure 2),
none showed clinical evidence of disease. IFAT results did not reveal detectable antibodies to Leishmania in 84.6%. 14.1% were positive at serum dilutions of 20, and 13% were positive at dilutions of 40 (Figure 3).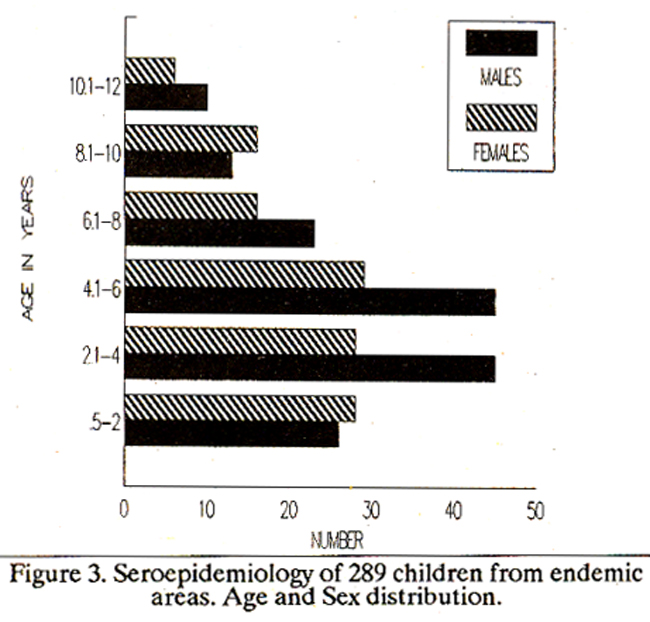
Sera from 40 control children living in nonendemic regions were all negative for leishmania antibodies.
Isoenzyme Characterization
Strain characterization on one isolate, identified the parasite strain as Leishmania infantum sensu stricto. Reference stocks used for identification included L. infantum s. st., L.infantum NH variant Linfantum NH MDH ASAT variant and L.donovani. Enzyme patterns were examined for enzymes such as PGM, PGJ, GOT, G6PD, P6GD, ME, MDH, LDH, MPI and NH.
DISCUSSION
IFAT has been used to study leishmania antibodies in patients suffering from YL. All patients exhibited high levels of such antibodies, except two children, in whom the levels were rather low (Positive at dilution of 200). Both these patients had re ceived a course of Pentavalent Antimony before their serum sample was tested. Successful treatment of VL with pentavalent antimonials results in a rapid decline in leishmania specific antibodies and this is used as an indicator to monitor and assess the response to treatment in patients7,8. Titers over 1:256 are considered to be diagnostic for VL9, and in our study, 91% of the patients presenting with clinical disease had antibody levels higher than 1:400 (Figure 1). Of the 22 patients in the study, the disease was diagnosed both by a positive bone marrow aspiration and high levels of leishmania antibodies detected by tEAT in 18. IFAT alone was used successfully for diagnosis in the remaining four, and two of these patients had IFAT antibody levels of 1:800 while the other two had titers in excess of 1:3200. Clinical studies in this region show evidence that the disease is of the Mediterranean type, affecting most commonly young children. The disease is manifested by prolonged fever, anaemia, hepatosplenomegaly, generalised lymphadenopathy, weakness, wasting and emaciation. Laboratory investigations in patients revealed pancytopenia and markedly increased levels of gammaglobulins. Leishmania isolate obtained from bone marrow of a two year old male child admitted in a local hospital was typed as Leishmania infantum ss on isoenzyme characterization. Seven cases occurred in five villages of AJ&K. A randomly selected sample of 289 children under 12 years of age, from these villages revealed low levels of Leishmania antibodies. Of these, 14.1% had antibody titers of 1:20 and only 13% had levels of 1:40, while 84.6% of the children were negative for leishmania specific antibodies. Sera obtained from 40 control children from non endemic area were also negative. In one house hold where a six year old female patient had recently expired of VL, none of her family members including 3 sisters (age range 2-10 years) showed any clinical evidence nor had history suggestive of past disease. They were also negative on serology. In another village a 5 year old girl who had suffered from the disease two years before, and had since fully recovered demonstrated an antibody level of 1:20 in her serum. Careful foflowup of the children with low levels is therefore essential. Asymptomatic and subclinical forms of VL are known to occur. 10 In a prospective study in Brazil11, of the 86 children with leishmania antibodies 23.3% remained asymptomatic, 17.4% developed classical VL within weeks of index serology. The remaining 59.3% children showed subclinical disease manifested by mild constitutional symptoms malaise, diarrhoea, poor work-play tolerance and intermittent hepatomegaly. Of those showing subclinical disease 25% progressed to classical VL, and the remaining resolved theft illness after a prolonged period. As Geimsa stained smears of bone marrow aspirates are usually negative for leishmania in the absence of classic Kala-azar, infection rates in populations can be determined by sensitive serological techniques such as IFAT, EUSA, RIA12 and by delayed hypersensitivity skin testing13,14. Identification of vector in this region has not yet been achieved. Although a large variety of phlebotomine sandflies are found in Pakistan15, the Ones that have been identified in the northern areas include Ph.chinensis, Ph. sergenti, Ph.kendeliki burnei and Ph.major. Promastigotes have not been found in any of the species so far. Dogs and other wild carnivores such as foxes, jackals and wolves are primarilybelieved to be the reservoir of disease in most endemic areas16. The prevalence of human infection cannot be directly correlated with that in dogs. A number of domestic dogs were examined for evidence of clinical disease such as cutaneous lesions, onychogyrophosis, keratoconjunctivitis, Hgidity of posterior limbs etc. None were found tobe affected. Sera from 4 dogs tested for leishmania antibodies by bemagglutination, yielded negative results. Recently however, seilsitive and more reliable techniques such as IFAT17 have been successfully employed in the detection of antibodies in the reservoir host. In conclusion, therefore it is observed that sporadic cases of VL are occurring in the northern parts of the country, most commonly affecting young children. The disease was first reported from the northern areas about three decades ago and was thought to be restricted to foci in that region. However, since the advent of this decade, cases of visceral leishmaniasis began to be reported from different foci in Azad Jammu and Kashmir. Even more alarming is the fact that the disease has also been reported from regions of the North West Frontier Province as well as Punjab that neighbour AJ&K. There is thus conclusive evidence that the disease is gradually spreading southwards in the country. In order to limit this disease the exact epidemiological pattern needs to be defmed and knowledge regarding both vector as well as the reservoir host is essential before comprehensive control efforts can be undertaken.
ACKNOWLEDGEMENTS
The authors are indebted to Drs. Gramiccia, M. and Gradoni,L. of the Instituto Superiore Di Santa, in Rome, Italy, for isoenzyme characterization of a Leishmania isolate. Support of Drs K Abbas and Z. Kundi from Polyclinic and Holy Family hospitals at Islamabad/Rawalpindi is also gratefully acknowledged. Thanks are also due to Mr. Manzar All, Technician, Immunology Department, NIH, for helping us in field studies. This study was sponsored by the WHO/TDR Special Program for Research and Training in Tropical Diseases.
REFERENCES
1. Ahmad,N., Burney,M.l. and Wazir,Y. A preliminary report on the study of Kala-Azar in Baltistan (West Pakistan). Pakistan Armed Forces Med. J., 1960; 10:1.
2. Ahmad, N. and Burney,M.I. Leishmaniasis in Northern areas of Pakistan (Baltistan). Pakistan Armed Force Med. J., 1962; 12:1.
3. Saleem,M., Anwar, CM. and Malik,LA. Visceral Leishmaniasis in children. A new focus in Azad Kashmir. JPMA., 1986; 36:230.
4. Pearson, R.D., Wheeler, D.A., Harrison, L H. and Kay, H.D. The immunobiology of Leishmaniasis. Rev. In fect.Dis., 1983;5:907.
5. Heyneman,D. Immunology of leishmaniasis. Bull. WHO., 1971; 44:499.
6. Latif, B.M., Al-Shenawi, F.A. and Al-Alousi,T.I. The indirect fluorescent antibody test for diagnosis of Kala azar infection in Iraq. Ann.Trop.Med. Parnsit., 1979; 73:31.
7. Navin, T.R., Sairra, M., Custudio, it, Stuerer, F., Porter, C.H. and Ruebusb,T.K. Epidemiological study of VL in Honduras, 1975- 1983. AmJ.Trop.Med.Hyg., 1985; 34:1069.
8. Burney, M.I., Lari,F.A. and Khan, M.A. Status of visceral leishmaniasis in northern Pakistan; a seroepidemiological assessment. Trop.Doct., 1981;11:146.
9. Edrissian, G.H. and Darabian, P. A comparison of ELI SA and IFAT in the sero-diagnosis of cutaneous and visceral leishmaniasis in Iran. Trans. Roy. Soc. Trop. Med. Hyg., 1979; 73:289.
10. Badaro, it, Jones, T.C., Carvalho, EM., Sampaio, D., Reed, S.G., Barral, A., Teixeira, R. and Johnson, W.D. Jr. New perspectives on a subclinical form of visceral leishmaniasis. J.Infect.Dis., 1986; 154:1003.
11. Paris, A., Massoud, A., El Said, S., Gadallah, M.A., Feinsod, F.M., Saah, A.J., Londner,M. and Rosen,G. Epidemiologyof human VL in El Agamy, Egypt; scrosur vey and case/control study. Ann. Trop. Med. Parasit., 1988; 82: 445.
12. Reed, S.G., Badoro, it, Masur, IT., Carvalho, E.M., Lorenco, it, Lisbio, A., Teixeira, it, Johnson, W.D. Jr. and Jones, T.C Selection of skin test antigen for American VL Am.J.Trop.Med.Hyg., 1986; 35:79.
13. Neogy, A.B., Nnndy, A., Dastidar, B.G., and Chowdbury, A.B. Leishmanin test in Indian kala-azar. Trans. it Soc. Trop. Med. Hyg., 1986; 80:454.
14. Lewis, DJ. The phlebotomine sand flies of West Pakistan (Diptera Psychoidae). Bull. Br. Mus. Nat. Hist. Entom., 1967; 19:3.
15. The Leishmaniasis. WHO Tech. Rep.Ser., 1984; 701: 67.
16. Mancianti, F., Gradoni, L, Gramicia, M. Pieri, S. and Marconcini, A. Canine leishmaniasis in the isle of Elba, Italy. Trop. Med. Parasitol., 1986; 37:110.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




