Hiba Qassem Ali ( Department of Microbiology, College of Medicine, University of Basrah, Basrah, Iraq. )
Nadham Kadham Mahdi ( Department of Microbiology, College of Medicine, University of Basrah, Basrah, Iraq. )
Mohammad Husein Al-Jowher ( Department of Surgery, College of Medicine, University of Basrah, Basrah, Iraq. )
September 2013, Volume 63, Issue 9
Original Article
Abstract
Objective: To evaluate the role of cancer antigen CA15-3 in the diagnosis, prognosis, followup and treatment of breast cancer.
Methods: The case-control study was done from October 2009 to February 2011 at the Al-Basrah Hospital in Basrah, Iraq. Blood samples were collected from 30 women with primary breast cancer before the invasive procedure. Another set of blood samples were collected from 23 of the cases after three cycles of chemotherapy. Besides, 20 serum samples were collected from apparently healthy women as a control group from the outpatient department. Enzyme-linked immunosorbent assay (ELISA) test was used for detection of serum CA15-3.
Results: Mean CA15-3 level for the cases (79.15±27.54U/ml) was significantly higher than the controls (24.34±11.68U/ml) (p<0.05), with 93.3% sensitivity and 96.6% specificity. There was a significant positive correlation with the disease stage and tumour size (p<0.05). After three cycles of chemotherapy, the CA15-3 level significantly decreased (p<0.05). Cases who developed recurrence were found to have a significantly higher CA15-3 level than those who did not (p<0.05).
Conclusion: CA15-3 is an important diagnostic, prognostic indicator and good predictor for relapse.
Keywords: Breast cancer, CA15-3, Metastases, Women. (JPMA 63: 1138; 2013).
Introduction
Cancer is a growing problem worldwide.1 Breast cancer is the third most frequent cancer in the world.2 It is by far the most frequent cancer of women (23% of all cancers), and is the most common cause of cancer-related death among women around the globe.3
Cancer antigen (CA) 15-3 is the product of MUC-1 gene, and mucins are aberrantly over-expressed in many adenocarcinomas in an under-glycosylated form and are then shed into the circulation.4 CA15-3 has been shown to be an independent predictor of first recurrence as well as a powerful prognostic indicator in patients with advanced breast cancer.5 Elevated pre-operative CA15-3 level is directly related to tumour burden and independent prognostic factors for breast cancer. It could be considered for clinical use such as predicting patient outcome and determining adjuvant treatment for better outcome.6
A study has shown that patients had serial marker values available before the development of metastatic disease; 29% had no increase in CA15-3 levels, whereas 71% of the patients had increasing marker levels before the diagnosis of metastatic disease.7 Another study showed change in CA15-3 levels with clinical course that 75% of patients with documented regressive disease had 40.5% decrease in CA15-3 levels. On the other hand, 87% with progressive disease had more than 100% increase in the levels of the antigen.8 However, CA15-3 can be elevated in the serum of patients with epithelial and non-epithelial non-mammary malignancies.9 In the followup of localised breast cancer, the CA15-3 tumour marker test is specific, but not sensitive enough to indicate the first relapse earlier than the other methods and the positive predictive value (PPV) especially remained poor in patients with a relatively good prognosis.10
The addition of other markers to the CA15-3 may increase the sensitivity during the post-surgical followup of breast cancer patients.11 However, the American Society of Clinical Oncology recommends that for monitoring patients with metastatic disease during active therapy, CA15-3 can be used in conjunction with diagnostic imaging, history and physical examination.12
The aim of the current study was to investigate the role of CA15-3 in breast cancer diagnosis, prognosis and treatment response.
Patients and Methods
The case-control study was conducted between October 2009 and February 2011 at the Al-Basrah Hospital in Basrah, Iraq. It comprised 30 women diagnosed with breast cancer by fine-needle aspiration (FNA) biopsy. Staging of the disease was performed for them by specialists and followed up for recurrence for a period of 10-17 months. Besides, 20 apparently healthy women were enrolled from the outpatient department (OPD) to serve as the control group.
Blood samples were collected from the cases prior to surgical operation for lumpectomy or mastectomy. Samples were again taken from the cases after three cycles of chemotherapy. Blood samples were collected and processed generally within an hour. The samples were centrifuged and stored in multiple tubes at -20°C. Blood samples were also collected from the 20 controls.
The study was approved by the Ethical Committee of the College of Medicine, University of Basrah, Basrah, Iraq. Informed written and verbal consent was also obtained from both the cases and the controls.
Serum CA15-3 concentration was determined by CA15-3 Enzyme Immunoassay Kit based on the principle of a solid phase enzyme-linked immunosorbent assay (ELISA), purchased from BioCheck, Inc, 323Vintage Park Drive, Foster City, CA 94404.
The CA15-3 conjugate reagent prepared by the entire 1ml of conjugate concentrate to 21ml of the enzyme conjugate diluents. Washing buffer was prepared by adding 50ml of the buffer to 950ml of distilled water.
Statistical analysis was performed by SPSS 17. Pearson test was used for the determination of correlation between the measured parameters. One-way analysis of variance (ANOVA) test was used for the determination of relation between different breast cancer stages and the control group. P<0.05 was considered statistically significant.
Results
The age of the 30 patients ranged between 27 and 70 years, with a mean of 48.6±11.27 years. After the initial sampling, 7 (23.3%) patients could not be followed up either because of poor compliance or lack of cooperation. The mean age of the 20 controls was 47.15±10.95 years.
A significantly higher serum CA15-3 level was seen in the cases (79.15±27.54U/ml) than the controls (24.34± 11.68U/ml) (p<0.05) (Table-1).
The cutoff level of 40U/ml was set according to the manufacturer\'s instructions.
The sensitivity and specificity of CA15-3 in the detection of breast cancer was 93.3% and 96.6% respectively.
A statistically significant (p<0.05) increase was noticed in CA15-3 values across breast cancer stages; from stage I to stage III (Table-2).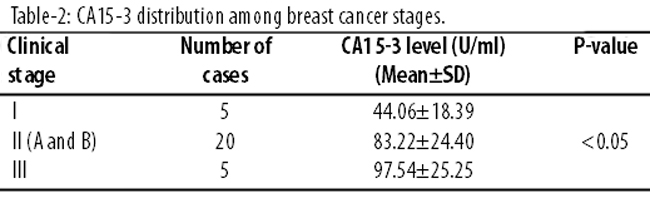
The preoperative CA15-3 values were significantly higher than the values after three cycles of chemotherapy (p<0.05) (Table-3).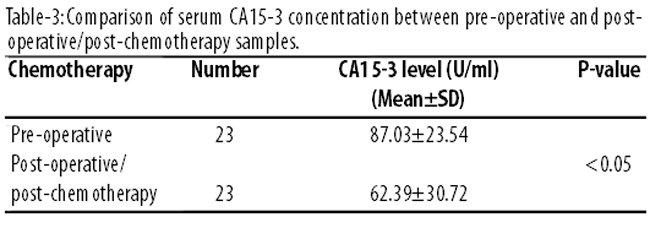
Six (26%) patients who developed recurrence exhibited a significantly higher CA15-3 level than the 17 (73.91%) patients who did not (Table-4).
No correlation was found between CA15-3 level and patient\'s age or lymph node metastasis, but a highly significant correlation was found between CA15-3 level and the disease stage as well as tumour size (Table-5).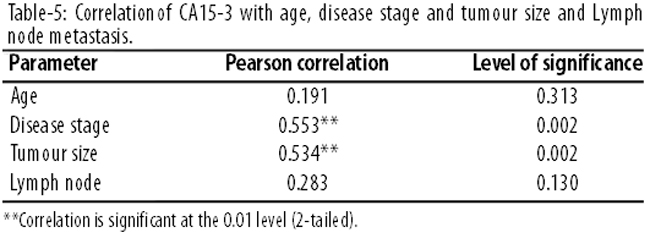
Discussion
There exists a conflict of opinion about the use of CA15-3 as a diagnostic tumour marker. Some have a low opinion about its value in diagnosing breast cancer, specially at early disease stages.13 Others find it to be a sensitive marker.7,14
The present study, like most others found a significantly higher CA15-3 level for breast cancer patients in comparison with controls.15,16 There is also a debate over CA15-3 sensitivity in breast cancer detection. It has been quoted at 76.1%,15 and 41%.7 The present study has recorded a sensitivity of 93.3% and a specificity of 96.6% while others have reported a sensitivity of 100%.15 This high sensitivity and high specificity gives CA15-3 test a value in breast cancer detection. Its level was significantly increased with advancing disease stage, and it was higher in cases than the controls at all stages. The results were in line with literature.16,17
The study also agrees with an earlier study reporting a positive correlation (>25%) between CA15-3 level of disease stages and tumour size, while no correlation was found with age, menopausal status and lymph node status.17 The marker, as such, is also sensitive for monitoring breast cancer patients.7,16
The rise of CA15-3 may proceed clinically for radiological diagnosis of recurrence by a mean lead time from 6 to 9 months.19 Elevated CA15-3 level is predictive of a poor response to chemotherapy. In addition, persistently elevated CA15-3 levels post chemotherapy predict a reduced disease-free survival following treatment in locally advanced breast cancer.20 The present study found an elevated CA15-3 levels after three cycles of chemotherapy for patients who developed recurrence. A study, however, found no significance of the post-operative level in predicting breast cancer recurrence.21 The clinical interest of CA15-3 remains the early detection of the first recurrence. In this regard, more than 75% of first recurrences (all sites included) are associated with a significant rise in CA15-3 levels, with a particular sensitivity for bone, liver, and lung metastases which appear lower for loco-regional recurrences.19 CA15-3 test is an important prognostic predictor.
CA15-3 level for the cases was higher than the controls. It increased with the increase in disease stage and decreased after three cycles of chemotherapy, with higher levels for patients who developed recurrence than patients who did not.
CA15-3 level exhibited a positive relation with disease stage and tumour size, while it was not related to axillary lymph node status and age.
Conclusion
CA15-3 is an important diagnostic, prognostic indicator and good predictor for relapse in breast cancer patients.
References
1. World Health Organization. National Cancer Control Programmes: Policies and Managerial Guidelines. Geneva, Switzerland: WHO Press; 2002.
2. Parkin DM, Pisani P, Ferlay J .Global cancer statistics. CA Cancer J Clin 1999; 49: 33-64.
3. Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin 2005; 55: 74-108.
4. Perey L, Hayes DF, Maimonis P, Abe M, O\'Hara C, Kufe DW. Tumour selective reactivity of a monoclonal antibody prepared against a recombinant peptide derived from the DF3 human breast carcinoma-associated antigen. Cancer Res 1992; 52: 2563-8.
5. Park BW, Oh JW, Kim JH, Park SH, Kim KS, Kim JH, et al. Preoperative CA 15-3 and CEA serum levels as predictor for breast cancer outcomes. Ann Oncol 2008;19: 675-81.
6. Berrutia A, Tampellini M, Tortaa M, Buniva T, Gorzegno G, Dogliotti L. Prognostic value in predicting overall survival of two mucinous markers: CA 15-3 and CA 125 in breast cancer patients at first relapse of disease. European J Cancer 1994; 30: 2082-4.
7. Geraghty JG, Coveney EG, Sherry F, O\'Higgins NJ, Duffy MJ. CA 15-3 in patients with locoregional and metastatic breast carcinoma. Cancer 1992; 70 : 2831-4.
8. Berberoglu U, Ceyhan B, Erçakmak N, Sezerdogdu V. The value of new tumor marker CA 15-3 in diagnosis and monitoring of patients with breast cancer. J Islamic Academy Sci 1989; 2: 113-7.
9. Colomer R, Ruibal A, Genolla J, Salvador L. Circulating CA 15-3 antigen levels in non-mammary malignancies. Br J Cancer 1989; 59: 283-6.
10. Kokko R, Holli K, Hakama M. CA15-3 in the follow-up of localized breast cancer: a prospective study. European J Cancer 2002; 38: 1189-93.
11. D\'Alessandro R, Roselli M, Ferroni P, Mariotti S, Spila A, Aloe S, et al. Serum tissue polypeptide specific antigen (TPS): a complementary tumor marker to CA 15-3 in the management of breast cancer. Breast Cancer Res Treat 2001; 68: 9-19.
12. Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S, et al. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol 2007; 25: 5287-312.
13. Duffy MJ. Serum tumor markers in breast cancer: are they of clinical value? Clin Chem 2006; 52: 345-51.
14. Estevam FR, Augusto SF, Rodrigues SA, Pinheiro MR, Monteiro AF. Apoptosis and production of TNF-b by tumor-associated inflammatory cells in histological grade III breast cancer. Cancer Immunol Immunother 2003; 54: 671-6.
15. Agyei Frempong MT, Darko E, Addai BW. The use of carbohydrate antigen (CA) 15-3 as a tumor marker in detecting breast cancer. Pak J Biol Sci 2008; 11: 1945-8.
16. Thriveni K, Krishnamoorthy L, Ramaswamy G. Correlation study of Carcino Embryonic Antigen & Cancer Antigen 15.3 in pretreated female breast cancer patients. Indian J Clin Biochem 2007; 22: 57-60.
17. Cioffi M, Fratta M, De Lucia D. CEA, TPA and CA 15-3 in breast cancer. Cancer Detect Prev 1993; 17: 14-8.
18. Lumachi F, Basso SM, Brandes AA, Pagano D, Ermani M. Relationship between tumor markers CEA and CA 15-3, TNM staging, estrogen receptor rate and MIB-1 index in patients with pT1-2 breast cancer. Anticancer Res 2004; 24: 3221-4.
19. Antoine E, Kayitalire L, Spielmann M. CA 15-3 in breast cancer. Rev Med Interne 1994; 15: 650-62.
20. Al-azawi D, Kelly G, Myers E, McDermott EW, Hill AD, Duffy MJ, et al. CA 15-3 is predictive of response and disease recurrence following treatment in locally advanced breast cancer. BMC Cancer 2006; 6: 220. doi:10.1186/1471-2407-6-220
21. Ebeling FG, Stieber P, Untch M, Nagel D, Konecny GE, Schmitt UM, et al. Serum CEA and CA 15-3 as prognostic factors in primary breast cancer. Br J Cancer 2002; 86: 1217-22.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




