Kiran Kamran ( Department of Anatomy, Foundation University Medical College, Rawalpindi. )
Muhammad Yunus Khan ( Department of Anatomy, CPSP Regional Centre, Islamabad. )
Liaqat Ali Minhas ( Depatment of Anatomy, Rawal Medical College, Rawalpindi. )
September 2013, Volume 63, Issue 9
Original Article
Abstract
Objective: To study the effects of ethanol vapour inhalation on the heart chambers of chick embryo.
Methods: The case-control study was conducted at the College of Physicians and Surgeons Pakistan regional centre in Islamabad from January to October 2007. Both experimental and control groups were divided into three sub-groups each, based on the day of the sacrifice. Each group was dissected on day 7, day 10 and day 22 or hatching whichever was earlier. The experimental sub-groups sacrificed on day 7, day 10 and on hatching, were exposed to ethanol vapours till day 6, 9 and 9 of incubation respectively. The diameter of all 4 chambers was measured in experimental hearts and compared with age-matched controls. SPSS 10 was used for statistical analysis.
Results: Ethanol vapour exposure caused widening of all heart chambers in the experimental chick embryos sacrifised on day 7 and day 10 compared to the controls. The chambers of newly hatched chick hearts showed dilatation in all the chambers except the left ventricle.
Conclusion: Ethanol vapour exposure during development affects the heart, resulting in the widening of all heart chambers. The exposure is as dangerous as drinking alcohol. Alcohol vapour exposure during development leads to progressive dilatation in different heart chambers, producing dilated cardiomyopathy.
Keywords: Ethanol vapour, Chick embryos, Heart chambers, Cardiomyopathy. (JPMA 63: 1084; 2013).
Introduction
Alcohol use during pregnancy can cause foetal abnormalities.1 Prenatal alcohol exposure exerts teratogenic effects on the developing foetus known as the foetal alcohol syndrome (FAS). Defects in the cardiovascular system appear in up to 50% of children diagnosed with FAS.2 Alcohol drinking can interfere with the normal functioning of the heart; a condition referred to as alcoholic cardiomyopathy. Alcoholic cardiomyopathy is a degenerative disease of the heart muscle characterised by a reduced capacity of the heart to pump blood (i.e., depressed cardiac output), reduced ability of the heart muscle to contract, and widening (i.e., dilatation) of all heart chambers.3 As the development of chick heart parallels that of the human heart,4 it has been used as a developmental model in various researches to study heart development and congenital heart defects.5
Nowadays a new route of alcohol intake is gaining popularity i.e. ethanol vapour inhalation through a special device known as Alcohol Without Liquid Vaporiser.6 Inhaled alcohol vapours are absorbed through blood vessels in the nose or lungs, bypassing stomach and liver. Manufacturers of this device are of the opinion that alcohol vapour inhalation has no long-lasting side-effects, but scientists are of the view that inhalation is as harmful as alcohol drinking.7 Various studies are now in progress in which ethanol vapours are given to the animals and humans to analyse its harmful effects.8,9 The purpose of this study was to assess the effects of ethanol vapour inhalation on heart chambers of chick embryos.
Materials and Methods
The experimental study was conducted at College of Physicians and Surgeons Pakistan\'s, Regional Centre in Islamabad from January to October, 2007. A total of 180 Desi (South Asian home-breed) Chicken (Gallus gallus domesticus) eggs collected from the Poultry Research Institute, Rawalpindi, were equally divided into control group \'A\' and experimental group \'B\'. Each group was further divided into 3 equal sub-groups; 1, 2 and 3 based on the day of the sacrifice. Sub-group 1 was sacrifised at day 7, sub-group 2 at day 10 and sub-group 3 on hatching or day 22 whichever was earlier. Experimental sub-group B1 was exposed to ethanol vapours from day 1 to day 6; sub-group B2 was exposed from day 1 to day 9 and sub-group B3 was also exposed to ethanol vapours from day 1 to day 9. The eggs which were from diseased mothers, cracked or stored in the refrigerator were excluded from the study.
Ethanol vapours were produced in a glass chamber containing ethanol in liquid form into which air was pumped with the help of an airpump. In line with literature, vapours produced were transmitted to the incubator where eggs were placed and the level of ethanol vapours in the incubator was monitored with the help of a breathalyzer.10-12 Concentration of ethanol in the incubator was maintained in the range of 0.75mg/l to 1.5mg/l. This particular concentration of ethanol vapours was not toxic for the chick embryos as far as their survival was concerned, but at the same time it had some adverse effect on their normal growth. This dose was determined with the help of a preliminary project.
Chick embryos were taken out from their shells on the respective days of sacrifice and stored in phosphate-buffered formalin. Chick hearts were taken out through dissection of the anterior thoracic wall. The heart of the hatched chicks was measured. Length was measured from ascending aorta to the apex of the heart, while width was measured at the level of atrioventricular junction.
The hearts were processed for paraffin embedding and serial sections of heart were taken from the frontal plane to measure chambers. The sections were stained with haematoxylin and eosin.
Diameter of left atrial cavity was measured by taking the maximum transverse diameter of the cavity between left atrial wall and interatrial septum. Diameter of right atrial cavity was measured by taking the maximum transverse diameter of the cavity between right atrial wall and interatrial septum. Diameter of left ventricular cavity was measured by taking the maximum transverse diameter of the cavity between the left ventricular wall and interventricular septum. Diameter of right ventricular cavity was measured by taking the maximum transverse diameter of the cavity between the right ventricular wall and interventricular septum.
The data obtained was analysed using SPSS 10. Student T-test was applied to the quantitative data and p value of less than or equal to 0.05 was considered significant.
Results
The heart length and width of the experimental group B3 was significantly more than that of control group A3 (p<0.001) (Table-1).
The diameter of atrial cavities of B1 was more than that of A1 (Figure-1).
However, this difference was not statistically significant (Table-2).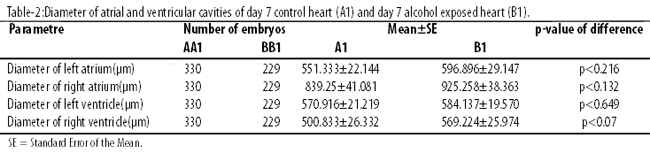
The diameter of ventricular cavities of B1 was more than that of A1, but the difference was again not statistically significant.
The diameter of left atrial and left ventricular cavity was more in B2 than that of A2 (Figure-2).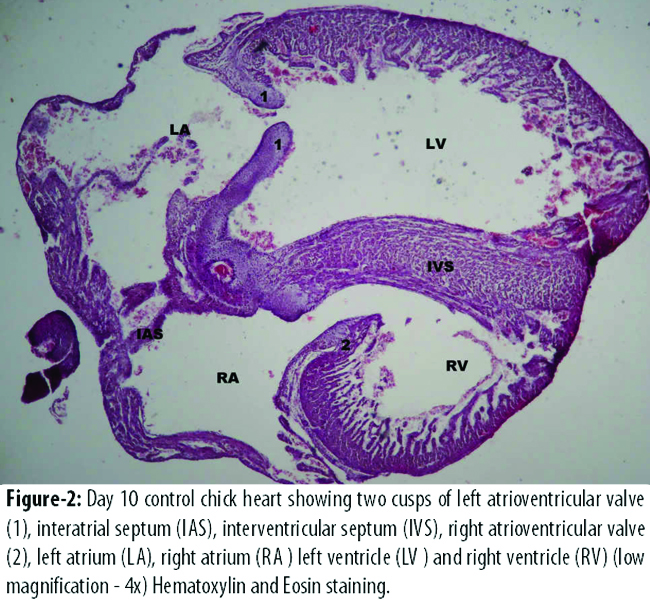
However, the difference was not statistically significant. The diameter of right atrial and right ventricular cavity was significantly more in B2 than that of control group A2 (Table-3).
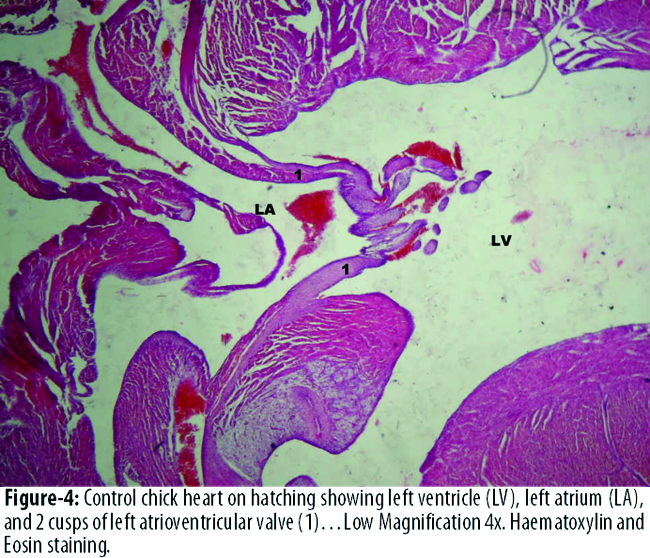
The diameter of left atrial cavity was significantly more in B3 than that of A3. The diameter of right atrial and right ventricular cavity was more in B3 than A3, but this difference was statistically not significant. The diameter of left ventricular cavity was more in A3 than that of experimental group B3, but the difference was not statistically significant. (Figures-3, 4).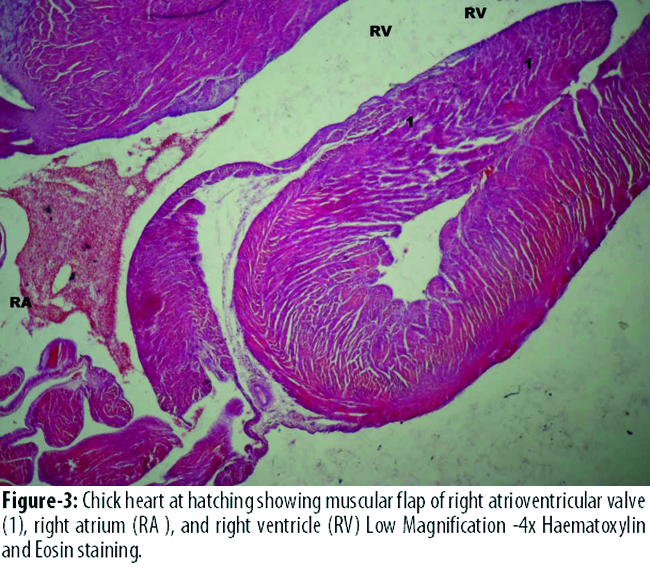
Discussion
The length and width of newly-hatched chick heart showed that the experimental group B3 had significant cardiomegaly compared to control group A3. Organomegaly has been seen in researches done on chronic alcoholic drinkers.13 This study shows that even alcohol inhalation can lead to this condition.
At day 7, the chick embryos in the experimental group showed an increase in diameter of all the four chambers of the heart compared to the chick embryos in the control group. However, this result was not statistically significant. The increase in diameter of all the heart chambers represents a form of dilated cardiomyopathy. Since, in the present study, the sole cause of dilatation in heart chambers was alcohol, it can be labelled as alcoholic cardiomyopathy. Catherine et all gave 20% ethanol in drinking water to adult chickens of two months\' age.14 They specifically studied left ventricle through echocardiography and histopathology. They found that the chickens developed left ventricle dilatation and left ventricular dysfunction. Another research done on a mice model showed that ethanol\'s metabolite acetaldehyde reduces myocardial contractility, disrupts mitochondrial function and produces apoptosis, all leading to myocardial damage and cardiac dysfunction.15 Cardiac dysfunction leads to decreased heart pumping, volume overload and eventually cardiac dilatation.
The diameter of left atrial and left ventricular cavity was more in experimental group B2 than that of control group A2, but, the difference was not statistically significant. The diameter of right atrial and right ventricular cavity was significantly more in experimental group B2 than that of control group A2. These findings were similar to the findings seen at day 7. The diameter of all the cavities was more in the day 10 experimental group than that of day 10 control group, presenting a form of dilated cardiomyopathy due to alcohol. Alcoholic cardiomyopathy accounts for 33% of all dilated cardiomyopathies. It is manifested by cardiomegaly, cardiac hypertrophy, compromised ventricular contractility and cardiac output.16 In another study done by Cheng et al, it was seen that chronic alcohol consumption causes inhibition of myocyte contraction and relaxation and dysfunctional calcium regulation leading to the development of alcoholic cardiomyopathy.17
The diameter of all the cavities was more in experimental group B3 than that of control group A3 except the diameter of left ventricular cavity which was more in the control group. This shows a persistence of dilatation in experimental hearts as compared to the controls except the left ventricle. One of the possible causes of dilated cardiomyopathy is the damage done to the developing heart due to alcohol exposure during the initial 10 days of incubation. Alcohol decreases ejection volumes and increases end systolic volume, resulting in volume overload and cardiac dilatation.14 Experimental studies show that alcohol and its metabolite acetaldehyde can disrupt myocardium by decreasing myocardial protein synthesis and cellular apoptosis.18 Uchenna et al found that a 39-year-old female after alcohol intake in fertility potions for 8 years developed dilated cardiac chambers.19
Left ventricle was not dilated in experimental hearts on hatching. This could be attributed to ethanol withdrawal. Chick embryos were exposed to alcohol vapours till day 10 after which alcohol was withdrawn. Ethanol withdrawal causes improvement of left ventricular systolic and diastolic functions. Earlier ethanol withdrawal in subjects with marked alcoholic dilated cardiomyopathy show improvement in left ventricular systolic function and symptoms of heart failure.19 That is why ethanol withdrawal at day 10 could be a cause of decreased left ventricular diameter in this study, indicating that there might be an improvement in its function. Another possibility could be increased ventricular wall hypertrophy which could have decreased the diameter of the chamber since alcohol damage induces cardiomyocyte hypertrophy seen in previous researches after alcohol drinking.14,20
Conclusion
Alcohol vapour inhalation during heart development causes cardiomegaly and cardiac chamber dilatation, resulting in dilated cardiomyopathy.
References
1. Ponnappa BC, Rubin E. Modeling alcohol\'s effects on organs in animal models. Alcohol Res Health 2000; 24: 93-104.
2. Ren J, Wold LE, Natavio M, Ren BH, Hannigan JH, Brown RA. Influence of prenatal alcohol exposure on myocardial contractile function in adult rat hearts: role of intracellular calcium and apoptosis. Alcohol Alcohol 2002; 37: 30-7.
3. Zhang X, Li SY, Brown RA, Ren J. Ethanol and acetaldehyde in alcoholic cardiomyopathy: from bad to ugly en route to oxidative stress. Alcohol 2004; 32: 175-86.
4. Taber LA. Biophysical mechanisms of cardiac looping. Int J Dev Biol 2006; 50: 323-32.
5. Sakabe M, Matsui H, Sakata H, Ando K, Yamagishi T, Nakajima Y. Understanding heart development and congenital heart defects through developmental biology: a segmental approach. Congenit Anom(Kyoto) 2005; 45: 107-18.
6. Wikipedia: The Free Encyclopedia. Alcohol Without Liquid. (Online) Last Updated: May 23, 2010. (Cited 2010 September 19.) Available from URL: http://en.wikipedia.org/wiki/Alcohol_without_liquid.
7. The New York Times. Citing safety, states ban alcohol inhalers. The New York Times. (Online) October 8, 2010. (Cited 2012 November 23). Available from URL: http://www.nytimes.com/2006/ 10/08/us/08whiskey.html?_r=0.
8. Ristuccia RC, Spear LP. Sensitivity and tolerance to autonomic effects of ethanol in adolescent and adult rats during repeated vapour inhalation sessions. Alcohol Clin Exp Res 2005; 29: 1809-20.
9. Nadeau V, Lamoureux D, Beuter A, Charbonneau M, Tardif R. Neuromotor effects of acute ethanol inhalation exposure in humans: a preliminary study. J Occup Health 2003; 45: 215-22.
10. Kamran K, Khan MY, Minhas LA. Effects of ethanol vapour exposure on survival of chick embryos. J Coll Physicians Surg Pak 2009; 19: 150-3.
11. Kamran K, Khan MY, Minhas LA .Ethanol vapour induced growth suppression in chick embryo. Ann Pak Inst Med Sci 2010; 6: 164-7.
12. Kamran K, Khan MY, Minhas LA. Teratogenic effects of ethanol vapour exposure on chick embryos. J Pak Med Assoc 2011; 61: 328-31.
13. Wong JL, Arango-Viana JC, Squires T. Heart, liver and spleen pathology in chronic alcohol and drug users. J Forensic Leg Med 2008; 15: 141-7.
14. Morris N, Kim CS, Doye AA, Hajjar RJ, Laste N, Gwathmey JK. A pilot study of a new chicken model of alcohol-induced cardiomyopathy. Alcohol Clin Exp Res 1999; 23: 1668-72.
15. Guo R, Ren J. Alcohol dehydrogenase accentuates ethanol-induced myocardial dysfunction and mitochondrial damage in mice: role of mitochondrial death pathway. PLoS One 2010; 5:e8757. doi:10.1371/journal.pone.0008757.
16. Aberle II NS, Ren J. Experimental assessment of the role of acetaldehyde in alcoholic cardiomyopathy. Biol Proced Online 2003; 5: 1-12.
17. Cheng HJ, Grant KA, Han QH, Daunais JB, Friedman DP, Masutanis, et al. Up-regulation and functional effect of cardiac ?3-adrenoreceptors in alcoholic monkeys. Alcohol Clin Exp Res 2010; 34: 1171-81.
18. Smith SM. Alcohol-induced cell death in the embryo. Alcohol Health Res World 1997; 21: 287-97.
19. Uchenna D.I, Anyalechi JI, Jesuorobo DE. Alcoholic cardiomyopathy in a 39 year old female: a case report. The Internet Journal of Cardiology 2011; 10. doi: 10.5580/170.
20. Zhang B, Turdi S, Li Q, Lopez FL, Eason AR, Anversa P, et al. Cardiac overexpression of insulin-like growth factor 1 attenuates chronic alcohol intake-induced myocardial contractile dysfunction but not hypertrophy: roles of Akt, mTOR, GSK3beta, and PTEN. Free Radic Biol Med 2010; 49: 1238-53.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




