Alireza Yaghoubi ( Cardiovascular Research Centre, Tabriz University of Medical Sciences, Tabriz, Iran. )
Zahra Golmohamadi ( Cardiovascular Research Centre, Tabriz University of Medical Sciences, Tabriz, Iran. )
Azin Alizadehasl ( Cardiovascular Research Centre, Tabriz University of Medical Sciences, Tabriz, Iran. )
Rasoul Azarfarin ( Cardiovascular Research Centre, Tabriz University of Medical Sciences, Tabriz, Iran. )
September 2013, Volume 63, Issue 9
Original Article
Abstract
Objective: To explore the significance of platelet parameters, including mean platelet volume, platelet count and other haematological indices, in patients of acute coronary syndrome.
Methods: In this one-year retrospective cross-sectional study in 2010, a total of 631 patients were enrolled at the Cardiovascular Research Institute, Tabriz University of Medical Sciences, Iran, and classified into three groups: myocardial infarction, unstable angina, and Control. Cardiovascular risk factors such as smoking, diabetes mellitus, hyperlipidaemia and hypertension status were compared. Blood samples were collected for the measurement of platelet count, mean platelet volume and other haematological indices, including prothrombin time, partial thromoplastin time, blood group and Rh, haematocrit, haemoglobin, and erythrocyte sedimentation rate. SPSS 18 was used for statistical analysis.
Results: Of the total, there were 210 (33.3%; 163 men, 47 women) with myocardial infarction whose mean age was 62.7±14.2 years; 211 (33.3%; 110 men, 101 women) had unstable angina with a mean age of 63.5±13.1 years and 210 (33.3%; 117 men, 90 women) health controls with a mean age of 60.6±12.8 years. The mean platelet volume values of patients in the first two groups were significantly higher than the control group (10.14 ±1.05 fL, 9.82 ± 0.93 fL and 9.34±1.14 fL, respectively; p< 0.001). The platelet count was detected to be significantly lower in the patients than the controls (207.92±58.40×109/L, 220.18±65.81×109/L, 238±56.10×109/L, respectively, p<0.001). While blood cell count was significantly higher in those with myocardial infarction compared to the unstable angina patients and control group (P<0.001). Differences in mean values of mean platelet volume between the two patient groups was not significant, but mean values of platelet count was statistically significant between these two groups (p<0.04).
Conclusion: Mean platelet volume and platelet count may be considered prognostic markers for acute coronary syndrome.
Keywords: Platelet count, Mean platelet volume, Acute coronary syndromes, Hematological indices. (JPMA 63: 1133; 2013).
Introduction
Platelets play role in the pathogenesis of cardiovascular disorders, including atherosclerosis and its complications, such as unstable angina (UA) and myocardial infarction (MI).Thrombus is major cause of acute coronary syndromes (ACS) and platelets play an important role in developing intravascular thrombus. Because it was well proved that platelets play a central role in both initiation and propagation of ACS, prevention and treatment of platelets is important; the use of aspirin started several years ago. More recently, researchers have achieved useful results by blockade of the final common pathway in platelet aggregation.1
Patients with ACS need a therapy with an effective anti-platelets.2 Platelet activation can be detected by changes of plasma concentration in platelet counts (PC). At the same time, myocardial ischaemia and local destruction of cardiomyocyte lead to the increase troponin I concentration.3
Markers of platelet activation and haematological indices have been demonstrated to play a role in the pathophysiology of atherosclerosis and cardiovascular events. It has been shown that the mean platelet volume (MPV) is the most accurate measurement of the size of platelets, associated with platelet count, inversely. There is evidence that demonstrate MPV is an important biological variable and larger platelets have higher thrombotic potential. Platelet size has been shown to reflect platelet activity.4 Large platelets are metabolically and enzymatically more active than small platelets and platelet activation is measured via MPV.5
Risk factors such as smoking, diabetes mellitus, hypertension, hyperlipidaemia, mental stress, and obesity, acting either individually or in combination significantly play role in increasing the chances of coronary atherosclerosis. The PC and MPV were determined by automated cell counters that are routinely available in most clinical laboratories.
Studies have shown MPV, as an indicator of platelet activation, suggesting enhanced values in patients with ACS and MI.6 Finding in a study in Iran confirmed that not only could on-admission MPV predict impaired reperfusion and in-hospital cardiac events in acute ST segment elevation myocardial infarction (STEMI) patients treated with percutaneous coronary intervention (PCI), but also it could be considered a practical way to determine higher-risk patients.7
The MPV and mean platelet component in cardiovascular disease (CAD) have been subjects of numerous researches, because it is controversial, and the review of recent literature shows this parameter to be an independent predictive factor for the progression of CAD. Recent reports did not confirm a correlation between the MPV and platelet hyper-aggregatibility nor a mean platelet component as a marker of an enhanced platelet activation.
The aim of this study was to assess the role of MPV, PC, white blood cell (WBC) and other haematological indices like haemoglobin (Hb), haematocrit (Hct) and erythrocyte sedimentation rate (ESR) in hospitalised patients with ACS and MI and to find out a correlation.
Patients and Methods
The retrospective cross-sectional hospital-based study was carried out on 631 patients over a period of one year in 2010 at the Cardiovascular Research Centre, Tabriz University of Medical Sciences, Iran. Institutional ethical committee clearance was obtained and so was informed consent.
Samples were selected from hospitalised patients with diagnosis of ACS who were hospitalised within the first 24 hours of their chest pain into coronary care. Diagnosis of ACS was made by assessing unstable chest pain, typical electrocardiographic (ECG) changes and/or elevation of cardiac enzymes.
The data of 631 consecutive patients from their medical records with the diagnosis of non-ST elevation (NSTE) ACS who were hospitalised within the first 24 hours of their chest pain into coronary care were collected. Patients were classified into three groups. Group A: patients with MI; Group B: patients with UA; and Group C: healthy controls with no history of heart disease and a normal electrocardiogram.
Patients were considered in sub-groups depending on smoking status and if they had or did not have diabetes mellitus (DM), hyperlipidaemia and hypertension. Hypertension was diagnosed as blood pressure above 140/90mmHg or being on anti-hypertensive therapy. DM was diagnosed as fasting blood glucose above 125mg/dl or being on anti-diabetic drugs. Patients who were smoking before hospitalisation were accepted as smokers. Patients with severe hepatic or renal impairment were excluded. Patients with more than 24h of unstable chest pain and patients who were received anti-coagulant, anti-platelet or anti-inflammatory drugs, that might interfere with the interpretation of results, at least 5 days before blood sampling were excluded. Patients were followed up in the coronary care unit (CCU) and treated by doctors who were blinded to the study. Before the administration of anti-platelet or anti-coagulant drugs, except aspirin, blood samples were collected for prothrombin time (PT), partial thromboplastin time (PTT), blood group and Rh, Hct, Hb, ESR, PC and MPV determination through the brachial vein during hospitalisation into tubes with ethylenediaminetetraacetic acid (EDTA). Samples were studied within 20 min with an automated cell counter (Sysmex Kx-212N, Japan).
Values were expressed as mean±standard deviation. The means of continuous variables were compared with independent samples t-test (between two groups MI and UA) and the one way analysis of variance (ANOVA) test (among the three study groups). Chi-square tests were used to compare categorical variables, such as status of smoking, diabetes, gender, hyperlipidaemia and hypertension. All of the tests were two-tailed; p<0.05 was considered statistically significant. SPSS 18.0 was used for data analysis.
Results
Of the total, there were 210 (33.3%; 163 men, 47 women) with myocardial infarction whose mean age was 62.7±14.2 years; 211 (33.3%; 110 men, 101 women) had unstable angina with a mean age of 63.5±13.1 years and 210 (33.3%; 117 men, 90 women) health controls with a mean age of 60.6±12.8 years.
Risk factors in patients with MI and UA were compared (Table-1).
There were a significant relationship between gender, DM, number of diseased vessels, hypertension, smoking and hyperlipidaemia and type of diseases of MI and UA. There was no association between blood group, Rh and type of disease.
The mean WBC, PT and PTT was significantly raised in the groups A and B compared with controls (Table-2).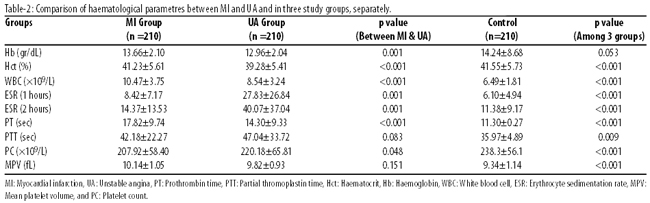
There were significant difference in mean of ESR (1 and 2 hours) and PT and PTT between groups A and B (p<0.001). ESR (1and 2 hours) was raised in group B compared to the control group.
WBC was significantly raised in the MI group compared to the UA and control groups (p<0.001).There was no relationship between WBC and age and smoking status. The means of Hb (p<0.001) [but not Hct (p<0.053)] decreased significantly in groups A and B compared to the control group.
The mean of MPV was significantly raised in groups A and B compared to the control group (10.14 ±1.05, 9.82 fL ± 0.93 fL, 9.2±0.96 fL, respectively; p<0.001). There was no significant difference in mean MPV between groups A and B (p<0.151). A significant difference was seen when the mean PC was compared between the groups A and B with the control group (207.92±58.40×109/L, 220.18± 65.81×109/L, 238±56.10×109/L, respectively; p<0.001) and significant difference was seen in mean PC between groups A and B (p<0.048) . Mean PC in UA patients was higher than in MI patients. There was no relationship between mean MPV and gender and no correlation was seen between the MPV and hb and Hct values.
Discussion
In ACS, rupture of unstable atherosclerotic plaque triggers a thrombogenic cascade leading to clinical events. However, platelet reactivity is critically important in the formation and propagation of intracoronary thrombus.8 MPV, one of the markers indicating the function of platelets, is a simple and easy measurement.5
This study recruited 631 patients with a mean age of 63.08±13.65 years, which was 8 years more than the mean age of the patients in the Vakili H study in Iran in 2009,7 but in accordance with Huczek study.9 Besides 64.84% of our patients were men, and this percentage was almost similar to that in other studies on the correlation between MPV and ACS (70-80%). Distribution of risk factors was similar to that in another study carried out specifically on the relation between the risk factors of ACS and MPV.6
In some studies results failed to demonstrate a difference in mean PC between patients with CAD and those with normal coronary arteries.9 This study found that MPV was raised in patients who had suffered an acute coronary event compared with controls. This is in agreement with the results of similar studies.7,8,11-14
Chu H et al in 2011 showed MPV is significantly associated with ACS in patients with acute chest pain and is an early and independent predictor.15 Chu SG et al. review demonstrated that elevated MPV is associated with acute MI, mortality following MI, and restenosis following coronary artery intervention.16
Some studies have shown higher MPV values in patients with UA than those with MI,17 but our study found no such difference which compares well with a study by Senaran et al.18 PC was significantly lower in our MI and UI patients compared with the controls which was in agreement with other studies.3,19,20
In our study PC was higher in UA patients than MI patients, whereas Mathur et al. in 2001 showed MPV was higher in UA patients than in those diagnosed with MI and PC was lower in UA than in MI.16 Avramakis et al’s study in 2007 showed that MPV was significantly raised in all MI and UI patient groups except in DM; non-smokers with UA or MI.20
In this research UA and MI patients had significantly higher WBC counts compared to the controls and is similar to most other studies such as Avramakis et al’s study.20 Besides, in our study, in MI patients group significantly higher WBC were observed than in UA group and controls. We suggest the association of an immunologic process in coronary disease that is similar to other studies.21 Libby et al. reported that higher baseline WBC counts were associated with greater extent of CAD.22
There is some evidence to suggest that inflammation and the WBC itself may directly contribute to coronary thrombosis, impaired perfusion, and reperfusion injury.23 Enhanced MPV has been reported in subjects with cardiovascular risk factors, such smoking,6 hypertension,24 DM,25 obesity,26 and hypercholesterolaemia,27 suggesting a common mechanism by which these factors may increase the risk of cardiovascular disease. Our study is in agreement with literature.
Our study showed significant difference in mean Hb and Hct between MI and UA patients, also controls unlike the Yilmaz4 and Khandekar6 studies. Also, significant difference were seen in means of ESR1 and ESR2, PT between MI and UA patients as well as compared to control group, but difference of PTT was not significant.
Yilmaz et al. study4 demonstrated MPV was not associated with the number of diseased vessels which was in contrast with the Yang et al study.29 In Cooke et al’s study there was no relation between mean PC and severity of CAD.10
In our study, MPV increased significantly in MI patients compared to the controls, and PC in MI patients was significantly lower than in UA patients and controls, alike.6,12,19
Most studies suggest MPV to be associated adversely with PC. One study showed that an increase in MPV was inversely correlated with platelet count.11
Our study showed that difference in MPV between MI patients and UA patients was not significant, like Khandekar et al. in India, but contrary to Yilmaz et al.4 In our study, difference of mean PC between MI and UA patients was significant, and in agreement with Yilmaz et al.4 In the Khandekar et al. study,6 it was not significant. PC between MI patients and controls is significant like the Khandekar study, while MPV was significantly higher in ACS group compared to the controls.
Our data demonstrated that the increased MPV contributes to the prethrombotic state in acute ischaemic syndromes. Whether the relationship is causal, and whether MPV should influence practice or guide therapy, remains unknown. Various drugs, including losartan and lipid-lowering therapies may reduce MPV, but no study to date has shown that lowering MPV reduces cardiovascular risk.29
Further studies are needed to evaluate whether MPV provides added value in identifying patients at enhanced clinical risk and whether therapeutic modification of this marker may lead to improved cardiovascular care.
Conclusion
High MPV could be a risk factor for ACS and MI. Since MPV measurement is a simple, feasible, economic and practical method, it should be considered along with other conventional risk factors in ACS.
References
1. Grover A, Bahl A, Vijayvergiya R, Kumar RM, Thingan ST. Changing trends of cardiovascular disease in India: implications for prevention based on 2001 census. Indian J Cardiol 2003; 6: 59-3.
2. Bigalke B, Lindemann S, Gawaz M. Platelet activation in acute coronary syndrome and interventional therapy. Hamostaseologie 2007; 27: 338-43.
3. Butkiewicz AM, Dymicka-Piekarska V, Kemona-Chetnik I, Kemona H, Bychowski J, Stogowski A. Platelet activation in unstable angina depending on troponin I concentration. Pol Merkur Lekarski 2005; 18: 13-6.
4. Yilmaz MB, Cihan G, Guray Y, Guray U, Kisacik HL, Sasmaz H, et al. Role of mean platelet volume in triagging acute coronary syndromes. J Thromb Thrombolysis 2008: 26: 49-54.
5. Endler G, Klimesch A, Sunder-Plassmann H, Schillinger M, Exner M, Mannhalter C, et al. Mean Platelet volume is an independent risk factor for myocardial infarction but not for coronary artery disease. Br J Haematol 2002; 117: 399-404.
6. Khandekar MM, Khurana AS, Deshmukh SD, Kakrani AL, Katdare AD, Inamdar AK. Platelet volume indices in patients with coronary artery disease and acute myocardial infarction: an Indian scenario. J Clin Pathol 2006; 59: 146-9.
7. Vakili H, Kowsari R, Namazi MH, Motamedi MR, Safi M, Saadat H, et al. Could mean platelet volume predicts impaired reperfusion and in-hospital major adverse cardiovascular event in patients with primary percutaneous coronary intervention after ST-elevation myocardial infarction? J Teh Univ Heart Ctr 2009; 4: 17-23.
8. Martin JF, Bath PMW, Burr ML. Influence of platelet size on outcome after myocardial infarction. Lancet 1991; 338: 1409-11.
9. Huczek Z, Kochman J, Filipiak K, Horszczaruk G, Grabowski M, Piatkowski R, et al. Mean platelet volume on admission predicts impaired reperfusion and long-term mortality in acute myocardial infarction treated with primary percutaneous coronary intervention. J Am Coll Cardio 2005; 46: 284-90.
10. Cooke J, Murphy T, McFadden E, O\'Reilly M, Cahill MR. Can mean platelet component be used as an index of platelet activity in stable coronary artery disease? Haematology 2009; 14: 111-4.
11. Smyth DW, Martin JF, Michalis L, Bucknall CA, Jewitt DE. Influence of platelet size before coronary angioplasty on subsequent restenosis. Eur J Clin Invest 1993; 23: 361-7.
12. Pizzulli L, Yang A, Martin JF, Lüderitz B. Changes in platelet size and count in unstable angina compared to stable angina or non-cardiac chest pain. Eur Heart J 1998; 19: 80-4.
13. Varol E, Akcay S, Ozaydin M, Erdogan D, Dogan A. Mean platelet volume in patients with coronary artery ectasia. Blood Coagul Fibrinolysis 2009; 20: 321-4.
14. Senen K, Topal E, Kilinc E, ten Cate H, Tek I, Karakoc Y, et al. Plasma viscosity and mean platelet volume in patients undergoing coronary angiography. Clin Hemorheol Microcirc 2010; 44: 35-41.
15. Chu H, Chen WL, Huang CC, Huang HY, Kuo HY, Gau CM, et al. Diagnostic performance of mean platelet volume for patients with acute coronary syndrome visiting an emergency department with acute chest pain: the Chinese scenario. Emerg Med J 2011; 28: 569-74.
16. Chu SG, Becker RC, Berger PB, Bhatt DL, Eikelboom JW, Konkle B, et al. Mean platelet volume as a predictor of cardiovascular risk: a systematic review and meta-analysis. J Thromb Haemost 2010; 8: 148-56.
17. Mathur A, Robinson MS, Cotton J, Martin JF, Erusalimsky JD. Platelet reactivity in acute coronary syndromes: evidence for differences in platelet behaviour between unstable angina and myocardial infarction. Thromb Haemost 2001; 85: 989-94.
18. Senaran H, Ileri M, Altinba? A, Ko?ar A, Yetkin E, Oztürk M, et al. Thrombopoietin and mean platelet volume in coronary artery disease. Clin Cardiol 2001; 24: 405-8.
19. Kiliçli-Camur N, Demirtunç R, Konuralp C, Eskiser A, Ba?aran Y. Could mean platelet volume be a predictive marker for acute myocardial infarction? Med Sci Monit 2005; 11: CR387-92.
20. Avramakis G, Papadimitraki E, Papakonstandinou D, Liakou K, Zidianakis M, Dermitzakis A, et al. Platelets and white blood cell subpopulations among patients with myocardial infarction and unstable angina. Platelets 2007; 18: 16-23.
21. Butkiewicz AM, Kemona H, Dymicka-Piekarska V, Bychowski J. Beta-thromboglobulin and platelets in unstable angina. Kardiol Pol 2003; 58: 449-55.
22. Sabatine MS, Morrow DA, Cannon CP, Murphy SA, Demopoulos LA, DiBattiste P, et al. Relationship between baseline white blood cell count and degree of coronary artery disease and mortality in patients with acute coronary syndromes: a TACTICS-TIMI 18 (Treat Angina with Aggrastat and determine Cost of Therapy with an Invasive or Conservative Strategy- Thrombolysis In Myocardial Infarction 18 trial) substudy. J Am Coll Cardiol 2002; 40: 1761-8.
23. Libby P, Simon DI. Inflammation and thrombosis: the clot thickens. Circulation 2001; 103: 1718-20.
24. Nadar S, Blann AD, Lip GY. Platelet morphology and plasma indices of platelet activation in essential hypertension: effects of amlodipine-based antihypertensive therapy. Ann Med 2004; 36: 552-7.
25. Papanas N, Symeonidis G, Maltezos E, Mavridis G, Karavageli E, Vosnakidis T, et al. Mean platelet volume in patients with type 2 diabetes mellitus. Platelets 2004; 15: 475-8.
26. Coban E, Ozdogan M, Yazicioglu G, Akcit F. The mean platelet volume in patients with obesity. Int J Clin Pract 2005; 59: 981-2.
27. Pathansali R, Smith N, Bath P. Altered megakaryocyte-platelet haemostatic axis in hypercholesterolaemia. Platelets 2001; 12: 292-7.
28. Yang A, Pizzulli L, Luderitz B. Mean platelet volume as marker of restenosis after percutaneous transluminal coronary angioplasty in patients with stable and unstable angina pectoris. Thromb Res 2006; 117: 371-7.
29. Coban E, Afacan B. The effect of rosuvastatin treatment on the mean platelet volume in patients with uncontrolled primary dyslipidemia with hypolipidemic diet treatment. Platelets 2008; 19: 111-4.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




