Mehmet Emre Yurttutan ( Faculty of Dentistry, Department of Oral and Maxillofacial Surgery, Ankara University, Ankara, Turkey. )
Recep Kestane ( Faculty of Dentistry, Department of Oral and Maxillofacial Surgery, Ankara University, Ankara, Turkey. )
Ahmet Keskin ( Faculty of Dentistry, Department of Oral and Maxillofacial Surgery, Ankara University, Ankara, Turkey. )
Omur Dereci ( Faculty of Dentistry, Department of Oral and Maxillofacial Surgery Osmangazi University, Eskisehir, Turkey. )
February 2016, Volume 66, Issue 2
Original Article
Biomechanical evaluation of oversized drilling on implant stability - an experimental study in sheep
Abstract
Objective: To evaluate the effect of oversized drilling on implant success and secondary stability.
Methods: The experimental study was conducted in Turkey from January to July 2013, after approval by the ethics committee of the University of Ankara, and comprised 2 female sheep. Alumina blasted implants 4mm in diameter and 10mm in length were employed; 16 implant sites were prepared on the proximal tibias of the 2 sheep. In the right tibia, a standard preparation with 3.5mm diameter was performed in the control group, while in the left tibia, an over-preparation with 4.2mm diameter was performed at the experimental group. The implants of the control group were non-mobile, while the experimental group had rotational and vertical movements. The initial implant stability was measured using the resonance frequence analysis. Animals were sacrificed after 12 weeks, and resonance frequence analysis and reverse torque values were measured.
Results: In the experimental group, the mean resonance frequence analysis value was 60.25±7.46 (range: 49-74) at the end of the 12-week healing period. There was no statistically significant difference between the two groups (p=0.926). In the removing stage, the mean reverse torque values was 97.75±22.23N/cm (range: 68.63-138.83) for control group and 96.25±21.93N/cm (range: 63.34-126.9) for the experimental group (p=0.84).
Conclusion: Osseointegration can be achieved in the absence of primer stability in the 12-week period of healing time even for alumina-blasted implants.
Keywords: Animal study, Dental implant, Implant stability, Osseointegration, Oversized drilling. (JPMA 66: 147; 2016)
Introduction
Dental implants have been widely used to restore missing teeth for many years.1 Success and survival rate of an implant are directly associated with primary stability.2 Primary stability is achieved with initial rigid fixation of implants during the surgery and considered necessary for a successful osseointegration.3-5
There are two different processes of bone formation at implant sites; contact and distance osteogenesis. Contact osteogenesis infers new bone formation in direct contact with the implant surface. Distance osteogenesis is the new bone formation on the surfaces of the parent bone and occurs when primary stability is absent at implant site.6,7 When there is a gap between the implant surface and bone, a clot will fill the space and will be replaced by a provisional connective matrix that will act as scaffold for formation of woven bone.8-10
The current study was planned to evaluate the osseointegration degree and biomechanical properties of implants with alumina-blasted surface at oversized implant sites.
Materials and Methods
The experimental study was conducted in Turkey from January to July 2013, after approval by the ethics committee of the University of Ankara, and comprised 2 female sheep 18-24 months of age and each weighing about 30kg.
Grade IV titanium implants were sandblasted using alumina particles and the average roughness value of the surface was 1.5mm (Genamer, Ankara, Turkey). They were 4mm in diameter and 10mm in length with cylindrical design.
All surgical procedures were performed under sterile conditions with general anaesthesia. First, the surgical area was shaved, washed and disinfected with povidoneidodine. To further reduce the risk of postoperative infection, the sheep were treated with antibiotics (Iesefs 1 g, Sefriakson Na intramuscularly [IM] [I.E. UlagayIlac, Istanbul, Turkey] pre-operatively, and 1.5 g IM for 5 days postoperatively). Xylazine (Rompun, 0.2-0.5ml/kg i.m., Bayer, Istanbul, Turkey) and diclofenac potassium (diklorannon steroid anti-inflemmatuar IM, 1mg/kg, BilimIlac, Istanbul, Turkey) were administered as premedication. General anaesthesia was achieved using an intravenous (IV) injection of pentobarbital and maintained by isoflurane 3.5% (volume/volume) (Forane, Abbott Laboratories, Rungis Cedex, France), administered through an endotracheal tube. The bone surface in the proximal tibia region was exposed by an incision followed by a separate elevation of skin and periost.
A total of 16 implant sites were prepared in the proximal tibias. In the right tibia, a standard preparation with 3.5mm diameter was performed in the control group, while, in the 16 implant sites in the left tibia, an over-preparation with 4.2mm diameter was performed in the experimental group (Figure-1).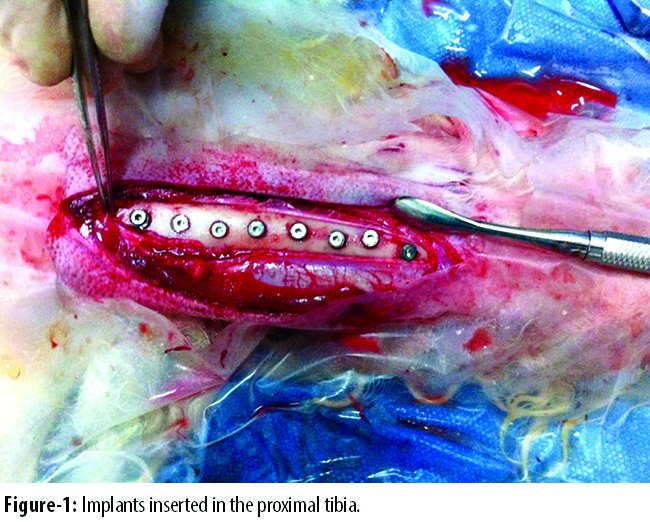
The initial implant stability was measured using the resonance frequence analysis (RFA) (Ostell Mentor, Integration Diagnostics AB, Göteborg, Sweden). The RFA measurement was repeated five times and the average was recorded as final. However, RFA value for the experimental group could not be measured because the value was not within the detectable range of the device. Cover screws were applied onto the implants. However, the implants of the control group were non-mobile, while the experimental group had rotational and vertical movements. The operation was completed after suturing of the flap margins in layers.
The tibias of the sheep were splinted to avoid breaking (Figure-2).
The splints and the sutures were removed 3 weaks later. Animals were fed a standard diet throughout the healing period. Animals were sacrificed after 12 weeks.
Upon sacrifice of the animals, RFA values were measured and the block including the implants was retrieved. The block was immediately trimmed to the proper dimensions and embedded into a container containing fast curing methyl methacrylate. Following the polymerisation of the acrylic, the specimen was transferred to the testing setup including a special torque measurement device (Mark-10, MGT 12, USA) connected to the internal octagonal connection of the implant via a specially-designed castmount. Torque force was applied in counter clockwise direction until the implant was turned. The bone specimens were kept moist throughout the testing procedure by spraying with saline solution to mimic in vivo physiological conditions. The peak torque achieved by complete loosening of the implant was recorded by the device as Ibin, and Ibin values were converted to N/cm values.
Mean value, standard deviation as well as range were calculated for each outcome variable. Paired-t test was used for statistical evaluation of initial and postoperative RFA values for each implant in the control group. T-test was used for statistical evaluation of postoperative RFA values of the control and experimental groups and statistical evaluation of reverser torque (RT) values of the control and experimental groups. Correlation between RFA and RT was investigated by the Spearman rank correlation test. P<0.05 was considered statistically significant.
Results
There were no complications during the course of the study and no implant had to be excluded on account of clinical instability.
In the insertion stage, the mean RFA value was 56.87±3.24 (range: 52-62) for the control group. There was no initial RFA value for the experimental group because they were not in the detectable range..
RFA values in the 12th week increased to a mean of 60.5±7.61 (range: 48-77) in the control group. The change from baseline value was statistically significant (p=0.031).
For the experimental group the mean RFA value was 60.25±7.46 (range: 49-74) at the end of the 12-week healing period. RFA values between the groups were not statistically significant (p=0.926).
In the removing stage, the mean RT values were 97.75±22.23 N/cm (range: 68.63-138.83) for control group and 96.25±21.93 N/cm (range: 63.34-126.9) for the experimental group (p=0.84) (Table).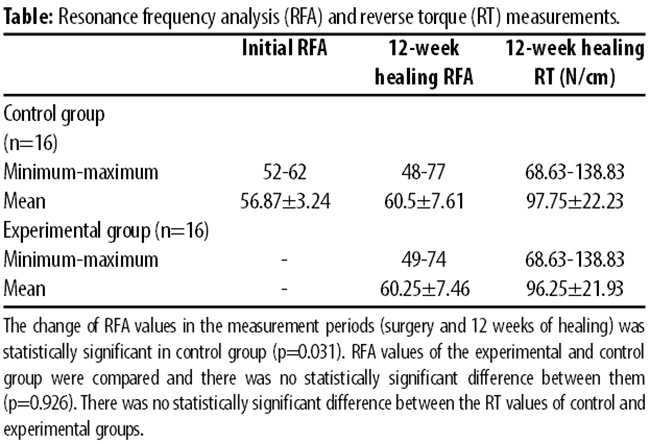
There was statistically significant correlation between RFA and RT values for experimental group (p<0.001; r=0.826) and also for the control group (p=0.001; r=0.765).
Discussion
Primary stability is the immobilisation of the dental implants at the initial stage of healing and achieved by press-fitting of the implant into the undersized-prepared implant site. It is suggested that primary stability is a prerequisite for successful dental implant osseointegration.4
Primary stability is achieved by direct bone implant contact and initiates contact osteogenesis. However, if a gap occurs between implant and parent bone, new bone formation occurs on the surfaces of parent bone and advances in the direction of implant threads, which is called distant osteogenesis.11 Mobility of an implant after insertion indicates oversized implant beds. Mobility grades of an implant during surgery can be classified as A) non-mobile implant, B) light rotation with feeling of resistance, C) rotation without resistance, and, D) rotation and lateral and/or vertical movements.5 A study suggested that implants placed without primary stabilisation and with rotational mobility achieve a degree of osseointegration that is equivalent to non-mobile implantsin 4 weeks.12
The RFA technique is widely used to assess the changes in bone-implant interface during osseointegration. In a study, RFA values of the study group with non-primarily stabilised implants were similar with control group, including primarily stabilised implants at the end of 4-week osseointegration period.13 In our study, the mean values of RFA in experimental and control groups at the end of 12-week osseointegration period were respectively 60.25 and 60.50. There was no statistically significant difference between experimental and control groups. The experimental group reached a satisfactory level of RFA value similar to control group even if implants were placed without primary stability. Also, mean RT values were similar and there was no statistically significant difference between experimental (96.25) and control (97.75) groups. Secondary stabilisation was successful in experimental group as well as in control group.
One study suggested that osseointegration still occurs even if the contact between implant surface and implant bed has deliberately been avoided.7 In several previous studies, it is suggested that adequate osseointegration can occur even if primary stability is avoided.12,14,15
Surface characteristic of a dental implant is considered to be an important factor for secondary stability. Moderately roughened hydrophilic surfaces and hydroxyapatite (HA)-coated surfaces are known to be effective in promoting bone healing and increasing the rate and degree of osseointegration.16-18 A study reported that HA-coated implants can reach adequate osseointegration degree when primary stability is not provided.13 The implants in this study were roughened with alimuna particles and there was no surface-coating. However, implants in the experimental group reached a satisfactory level of osseointegration compared to the control group.
RT and RFA measurements are basic tools for evaluation of stability and degree of osseointegration and are usually used in experimental implant studies to assess primary and secondary stability of the implants. In the present study, RT and RFA values of the experimental group at the end of the 12-week healing period were not statistically significant different from control group, indicating that osseointegration occurred at oversized implant sites was as successful as at normal-sized implant sites. These findings were in accordance with other experimental studies which were conducted on rabbits and mongrel dogs.13-15
In our view, loose implants should be avoided from micro movement and bacterial microflora to achieve solid osseointegration. In this regard, using sheep tibia instead of jaw bone may be considered a drawback of this study. Rich bacterial microflora and chewing movements may be considered a disadvantage of the osseointegration process of loose implants. Because of this, bone-level and submerged implants were used in our study to minimise these disadvantages. Some similar studies conducted on dog are in accordance with the results of the current study.12,13
Conclusions
Primary stability is necessary for healthy bone formation and initiation of contact osseogenesis. If primary stability cannot be established, distance osteogenesis initiates in the bone implant interface. Osseointegration can be achieved in the absence of primer stability in the 12-week period of healing time even for alumina-blasted implants.
Acknowledgments
We are grateful to the Ministry of Science, Industry and Technology, Republic of Turkey, for supporting the study.
References
1. Albrektsson T, Jansson T, Lekholm U. Osseointegrated dental implants. Dent Clin North Am. 1986; 30:151-74.
2. Friberg B, Jemt T, Lekholm U. Int J Early failures in 4,641 consecutively placed Brånemark dental implants: A study from stage 1 surgery to the connection of completed prostheses. Int J Oral Maxil Impl. 1991; 6:142-6.
3. Sennerby L, Thomsen P, Ericson LE. A morphometric and biomechanic comparison of titanium implants inserted in rabbit cortical and cancellous bone. Int J Oral Maxillofac Implants. 1992; 7:62-71.
4. Lioubavina-Hack N, Lang NP, Karring T. Significance of primary stability for osseointegration of dental implants. Clin Oral Implants Res. 2006; 17:244-50.
5. Rodrigo D, Aracil L, Martin C, Sanz M. Diagnosis of implant stability and its impact on implant survival: a prospective case series study. Clin Oral Implants Res. 2010; 21:255-61.
6. Davies JE. Understanding peri-implant endosseous healing. J Dent Educ. 2003; 67:932-49.
7. Sivolella S, Bressan E, Salata LA, Urrutia ZA, Lang NP, Botticelli D. Osteogenesis at implants without primary bone contact: an experimental study in dogs. Clin Oral Implants Res. 2012; 23:542-9.
8. Berglundh T, Abrahamsson I, Lang NP, Lindhe J. De novo alveolar bone formation adjacent to endosseous implants. Clin Oral Implants Res. 2003; 14:251-62.
9. Abrahamsson I, Berglundh T, Linder E, Lang NP, Lindhe J. Early bone formation adjacent to rough and turned endosseous implant surfaces. An experimental study in the dog. Clin Oral Implants Res. 2004; 15:381-92.
10. Rossi F, Botticelli D, Pantani F, Pereira FP, Salata LA, Lang NP. Bone healing pattern in surgically created circumferential defects around submerged implants: an experimental study in dog. Clin Oral Implants Res. 2012; 23:41-8.
11. Coelho PG, Marin C, Teixeira HS, Campos FE, Gomes JB, Guastaldi , et al. Biomechanical evaluation of undersized drilling on implant biomechanical stability at early implantation times. J Oral Maxillofac Surg. 2013;71:e69-75.
12. Jung UW, Kim S, Kim YH, Cha JK, Lee IS, Choi SH. Osseointegration of dental implants installed without mechanical engagement: a histometric analysis in dogs.Clin Oral Implants Res. 2012; 23:1297-301.
13. Jung UW, Kim S, Lee IK, Kim MS, Lee JS, Kim HJ. Secondary stability of microthickness hydroxyapatite-coated dental implants installed without primary stability in dogs. Clin Oral Implants Res. 2014; 25:1169-74.
14. Orenstein IH, Tarnow DP, Morris HF, Ochi S. Factors affecting implant mobility at placement and integration of mobile implants at uncovering. J Periodontol. 1998; 69:1404-12.
15. Ivanoff CJ, Sennerby L, Lekholm U. Influence of initial implant mobility on the integration of titanium implants. An experimental study in rabbits.Clin Oral Implants Res. 1996; 7:120-7.
16. Wennerberg A, Albrektsson T. On implant surfaces: a review of current knowledge and opinions. Int J Oral Maxillofac Implants. 2010; 25:63-74
17. Park YS, Yi KY, Lee IS, Han CH, Jung YC. The effects of ion beam-assisted deposition of hydroxyapatite on the grit-blasted surface of endosseous implants in rabbit tibiae. Int J Oral Maxillofac Implants. 2005; 20:31-8.
18. Lang NP, Salvi GE, Huynh-Ba G, Ivanovski S, Donos N, Bosshardt DD. Early osseointegration to hydrophilic and hydrophobic implant surfaces in humans. Clin Oral Implants Res. 2011; 22:349-56.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




