Nadia Aslam ( Department of Pathology, Sargodha Medical College, Sargodha. )
Naima Mehdi ( Department of Pathology, Gujranwala medical college, Gujranwala. )
Mateen Izhar ( Department of Microbiology, Shaikh Zayed Hospital, Lahore. )
October 2015, Volume 65, Issue 10
Original Article
Abstract
Objective: To find the important risk factors and sources of bacteraemia in patients suffering from methicillin-resistant staphylococcus aureus bacteraemia.
Methods: The descriptive study was carried out at Shaikh Zayed Hospital, Lahore, from October 2010 to August 2011. Blood cultures were processed to isolate methicillin-resistant staphylococcus aureus. A questionnaire was completed by the participating patients suffering from bacteraemia. Information about risk factors present at the time and risk factors that served as the source of bacteraemia were noted.
Results: Total 4058 blood cultures were processed and 669(16.5%) were positive. Of them, 194(29%) cultures were found to be positive for staphylococci. Out of these 194 blood cultures, coagulase-negative staphylococci were isolated from 117(60%), and 77(40%) were positive for S. aureus. Out of these 77 samples, 26(34%) were found to be methicillin-sensitive staphylococcus aureus and 51(66%) were methicillin-resistant staphylococcus aureus. The overall frequency of methicillin-resistant staphylococcus aureus was 1.25%; 7.62% out of positive blood culture; 26.28% out of total staphylococci; and 66% out of total S. aureus. As for the source of infection, central venous pressure line 11(21.6%), post-influenza pneumonia 9(17.6%), peripheral intravenous line 8(15.7%) and dialysis line 7(13.7%) were major reasons.
Conclusion: Taking care of aseptic measures while insertion, frequent change and early removal of the central venous and dialysis lines is of critical significance.
Keywords: Bacteraemia, Methicillin, Staphylococcus aureus. (JPMA 65: 1089; 2015)
Introduction
Staphylococcus aureus (S. aureus) bacteraemia is one of the most common serious bacterial infections worldwide, Blood stream infection with the S. aureus are associated with mortality rate of about 30%.1
Studies have shown that patients with acquired immune deficiency syndrome (AIDS),2 cancer patients especially neutropenic patients,3 diabetics,4 the very young,5 elderly and intravenous (IV) drug abusers6 and patients with indwelling devices are prone to get methicillin-resistant staphylococcus aureus (MRSA) infection.7,8 MRSA infections occur more frequently in hospitals and healthcare facilities with a higher incidence rate in nursing homes and long-term care facilities.9 Use of quinolone antibiotics predisposes to MRSA infection.10 People living in confined places, having poor nutrition and poor hygiene, prisoners,11 naval training centres and soldiers12 are prone to infections with MRSA. Several reports of MRSA among athletes and football players have been published.13,14 By identifying patients at risk, attempts can be made to minimise the spread of MRSA among patients and healthcare workers, patients at risk can be isolated and MRSA care pathway can be started as early as possible.
The current study was planned to find major risk factors associated with MRSA bacteraemia which can help to identify possible areas of intervention.
Material and Methods
The descriptive study was carried out at Shaikh Zayed Hospital, Lahore, a tertiary care, university teaching hospital. S. aureus bacteraemia was defined by the isolation of that organism from blood culture bottles collected by trained phlebotomists or trained nurses by using standardised sterile technique. The total blood culture bottles received in laboratory from October 2010 to August 2011 were processed to isolate MRSA after taking written informed consent from patients. Consecutive MRSA strains were isolated from total blood cultures studied. Non-repetitive strains of MRSA were isolated from blood cultures. Hospital-associated (HA-MRSA) strains were defined as patients with MRSA infection identified after 48 hours of admission; history of hospitalisation, surgery, dialysis, or residence in a long-term care facility within 6 months of the MRSA culture date; permanent indwelling catheter or percutaneous medical device (e.g., tracheostomy tube, gastrostomy tube, or Foley\'s catheter) present at the time of culture; or a known positive culture for MRSA prior to the study period.
Cases that had none of the above features were classified as community-associated MRSA (CA-MRSA).
Blood cultures were processed according to routine laboratory procedures. Blood culture bottles containing blood sample and tryptic soya broth (TSB) were incubated at 37ºC. Subculture was done on solid media chocolate agar and MacConkey agar after 48 hours. All the plates were incubated at 37ºC and read after 18 to 24 hours. Plates showing no growth were re-incubated and read after 24 hours. No growth was declared after 48 hours of incubation. Gram-positive cocci were selected on the basis of colonial morphology. These were confirmed by gram stain and then subjected to catalase test. Catalase-positive gram-positive cocci were labelled as staphylococci and subjected to slide coagulase and deoxyribonuclease (DNase) test. DNase-positive and slide coagulase positive were labelled as S. aureus. In case of discrepancy between the result of slide coagulase test and DNase test, final confirmation was based on tube coagulase test. For oxacillin susceptibility testing, S. aureus strains were inoculated on Mueller-Hinton agar plates. After placing 1ug oxacillin disk, the plates were incubated at 35ºC and the results were read after a full 24 hours, according to Clinical and Laboratory Standards Institute (CLSI) recommendations.15 Based on above-mentioned procedures the strains were finally classified as MRSA and stored for the purpose of the study. Frequency of MRSA bacteraemia was calculated by keeping record of all the blood cultures in the Microbiology department and number of MRSA isolated for the duration of study.
A questionnaire was completed by collecting basic clinical data of these patients. Number of different risk factors present in patients like presence of indwelling devices, assisted ventilation, immunosuppressive illness, co-morbidities such as cancer, diabetes, heart failure, hypertension, respiratory disease, intestinal disease and renal failure, was recorded. Previous hospital admission within 3 months before documentation of bacteraemia and operative procedures were included in risk factors. Information about source of bacteraemia, line site infection, soft tissue and bone infection, pneumonia including ventilator-associated pneumonia (VAP), endocarditis or any other illness was collected from hospital records.
MRSA-positive blood cultures from total blood cultures were reported as frequency and percentage. Number of CA-MRSA and HA-MRSA were separated from MRSA isolates from blood cultures and were reported as frequency and percentage.
Risk factors for MRSA bacteraemia were reported as frequencies and percentage.
Results
Total 4058 blood cultures were processed 2175(53.6%) were of male patients, while 1883(46.4%)were of female patients. Overall, 669(16.5%) were positive. Of them, 194(29%) cultures were found to be positive for staphylococci. Out of these 194 blood cultures, coagulase-negative staphylococci were isolated from 117(60%), and 77(40%) were positive for S. aureus. Out of these 77 samples, 26(34%) were found to be methicillin-sensitive S. aureus (MSSA) and 51(66%) were MRSA. The overall frequency of MRSA was 1.25%; 7.62% out of positive blood culture; 26.28% out of total staphylococci; and 66% out of total S. aureus.
Out of 51 MRSA cases, 7(14%) were CA-MRSA and 44(86%) were HA-MRSA.
Of the 51 MRSA patients, 11(21.5%) were less than one month old, 4(8%) were between 1 month and1 year of age, 1(1.9%) was between 1-12 years of age, 9(17.6%) were in 12-40 years age group, 14(27.4%) were in 40-60 years of age, and 12(23.5%) patients were over 60 years of age. Of the 51 patients with MRSA-positive blood cultures, 33(64.7%) were males and 18(35.3%) were females; the male-to-female ratio being1.8:1 (p=3.84).
Central venous pressure line 11(21.6%), post-influenza pneumonia 9(17.6%), peripheral intravenous line 8(15.7%) and dialysis line 7(13.7%) were major reasons.
As for the source of infection, 113 indwelling devices were seen in 43(84.3%) patients (Table-1).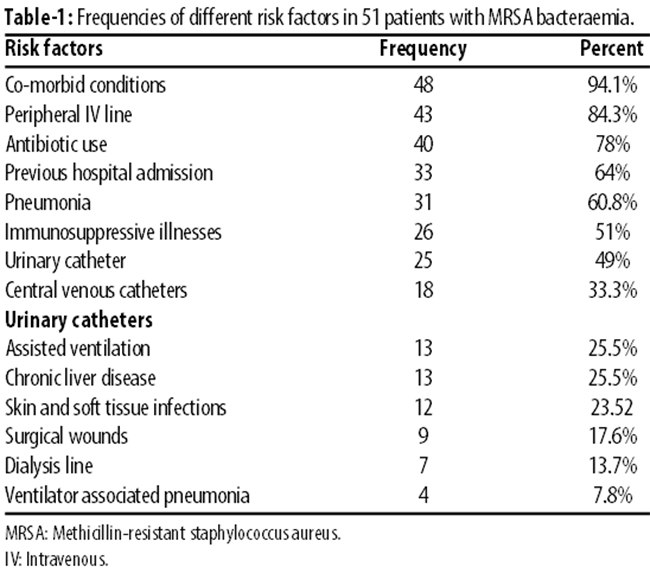
Co-morbid conditions were present in 48(94%) patients having 121 co-morbid conditions. Renal failure was seen in 12(25%) patients, 10(21%) patients were bed-ridden with cerebrovascular accident (CVA) and were having bed sores; 31(64.5%) patients had respiratory tract infection (RTI); 15(31.2%) had urinary tract infection (UTI).
Source of MRSA bacteraemia was recorded separately (Table-2).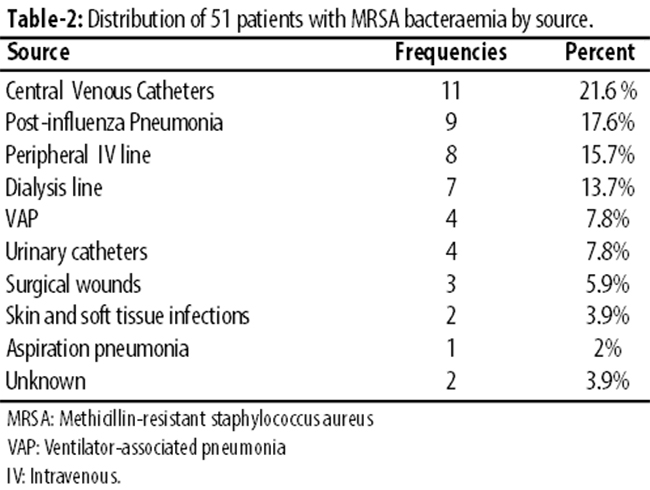
Of the 51 MRSA-positive patients, 43(84%) had Peripheral IV line, but 8(18.6%) had IV line as the source of infection. Among 18(35%) patients with central venous pressure (CVP) line, 11(61%) had CVP as the source. Among 7(13.7%) patients having dialysis line, 7(100%) had dialysis line site infection. Among 25(49%) patients with urinary catheters, 4(16%) had UTI as the source. Likewise, in 13(25.4%) patients with assisted ventilation, VAP was found in 4(30.7%). Skin and soft tissue infection was present in 12(23.5%) patients, but was the source of infection in only 2(16.6%). Surgical wounds were present in 9(17.6%) patients, but was the source in 3(11.1%). Further, 31(61%) patients had RTI, but was the source in 9(29%). Lastly, 22(43%) patients had nasogastric (NG) tube, but it was not found to be the source in any patient (0%) (Table-3).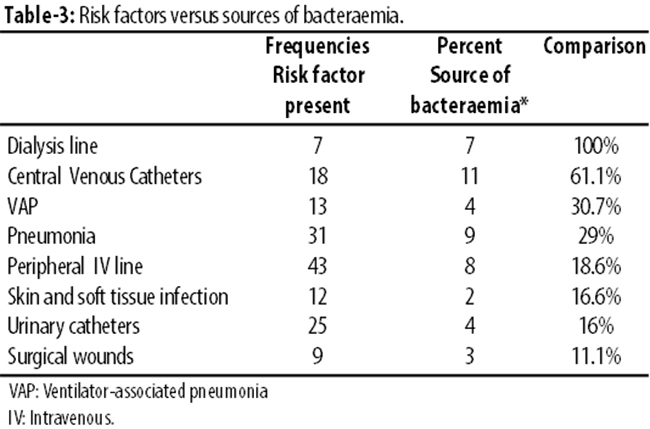
Discussion
Identifying high-risk group and taking preventive measures is important as MRSA is a lethal organism when it enters the blood stream.16 Patients who have recently been discharged or transferred from another hospital, are at increased risk of carrying MRSA.17,18 In this study, recently discharged patients or referred patients from other hospitals were found to be at increased risk of MRSA bacteraemia. Previous hospital admission is a risk factor for MRSA colonisation at the time of admission because nasal colonisation can lead to MRSA infection. Studies, including this study, found previous hospital admission as a predisposing factor for MRSA infection.19,20 Colonisation and infection by S. aureus are known to be significantly associated with infection among hospitalised patients.21
Some studies suggest that measures to reduce the incidence of hospital-acquired MRSA bacteraemia in acute general hospitals should focus on improving infection control procedures for the insertion and care of central lines and urinary catheters.22 In most instances the development of MRSA bacteraemia is probably a two-stage process with acquisition of the organism and colonisation of skin or superficial sites followed, after a variable period, by invasion of the bloodstream. Central or peripheral vascular lines are the most obvious routes for direct invasion into the bloodstream.22 Indwelling central venous catheters and dialysis lines were the most important causes of MRSA bacteraemia in the study. Therefore, it can be expected that by taking care of IV lines, taking standard precautions before insertion and frequent change of IV catheters can prevent risk of MRSA bacteraemia. Patients who had central line inserted also frequently had peripheral IV line, urinary catheter and NG tube. Also, patients with multiple indwelling devices were found to be at increased risk as presence of multiple indwelling devices not only shows severity of co-morbid conditions, but also exposes patient to risk of acquiring MRSA infection from any of indwelling devices. In such cases, additional care for every indwelling device should be taken because patient can get MRSA from any or multiple indwelling devices. Among risk factors that became source, dialysis line site infection was the most important, followed by central venous line site infection.18,22,23 Other important sources were pneumonia, including post-influenza24 as well as VAP and urinary catheter-associated infection.22
A prolonged hospital stay and prior exposure to broad spectrum antimicrobial therapy also appear to predispose patients to MRSA.17,25,26 In our study, 78.4% patients included had history of antibiotic use; most of the patients were taking third-generation cephalosporins or quinolones at the time of diagnosis. Therefore, injudicious use of antibiotics should be avoided.
Patients suffering from different immunosuppressive illnesses, diabetes mellitus, AIDS, cancer patients, substance abusers or receiving immunosuppressive therapy were found prone to MRSA bacteraemia. Other studies also found patients suffering from AIDS,1 cancer patients,3 diabetics,17,23,27 patients receiving immuno-suppressive therapy to be at increased risk.
A total of 121 co-morbid conditions were recorded in 51 patients during this study. Patients suffering from co-morbid conditions (renal insufficiency and dialysis patients,7,23,26 bed-ridden patients with bed sores,22 liver cirrhosis) were found to be at increased risk of infection with MRSA. Therefore, increased care should be taken for such patients during hospital stay. Also, patients suffering from tuberculosis were found to be at increased risk of MRSA infections.
Conclusion
Central venous lines and dialysis lines are important risk factors as well as source of bacteraemia. Therefore, taking care of aseptic measures while insertion, and frequent change and early removal of these devices are recommended.
References
1. Thwaites GE, Edgeworth JD, Gkrania-Klotsas E, Kirby A, Tilley R, Török M. E, et al. Clinical management of Staphylococcus aureus bacteraemia. Lancet Infect Dis 2011; 11: 208-22.
2. Cenizal MJ, Hardy RD, Anderson M, Katz K, Skiest DJ. Prevalence of and Risk Factors for Methicillin-Resistant Staphylococcus aureus (MRSA) Nasal Colonization in HIV-Infected Ambulatory Patients. J Acquir Immune Defic Syndr 2008; 48: 567-71.
3. Smiley S, Almyroudis N, Segal BH. Epidemiology and Management of Opportunistic Infections in Immunocompromised Patients with Cancer. Abstr Hematol Oncol 2005; 8: 20-30.
4. Eleftheriadou I, Tentolouris N, Argiana V, Jude E, Boulton AJ. Methicillin-Resistant Staphylococcus aureus in Diabetic Foot Infections. Drugs 2010; 70: 1785-97.
5. Kuint J, Barzilai A, Regev-Yochay G, Rubinstein E, Keller N, Maayan-Metzger A. Comparison of community-acquired methicillin-resistant Staphylococcus aureus bacteremia to other staphylococcal species in a neonatal intensive care unit. Eur J Pediatr 2007; 166: 319-25
6. Al-Rawahi GN, Schreader AG, Porter SD, RoscoeD L, Gustafson R, Bryce E A. Methicillin-Resistant Staphylococcus aureus Nasal Carriage among Injection Drug Users: Six Years Later. J Clin Microbiol 2008; 46: 477-9.
7. Lu PL, Tsai JC, Chiu YW, Chang FY, Chen YW, Hsiao CF, et al. Methicillin-resistant Staphylococcusaureus carriage, infection and transmission in dialysis patients, healthcare workers and their family members. Nephrol Dial Transplant 2008; 23: 1659-65
8. O\'Neill E, Pozzi C, Houston P, Smyth D, Humphreys H, Robinson DA, et al. Association between methicillin susceptibility and biofilm regulation in Staphylococcus aureus isolates from device-related infections. J Clin Microbiol 2007; 45:1379-88.
9. Mody L, Kauffman CA, Donabedian S, Zervos M, Bradley SF. Epidemiology of Staphylococcus aureus Colonization in Nursing Home Residents. Clin Infect Dis 2008; 46: 1368-73.
10. Weber SG, Gold HS, Hooper DC, Karchmer AW, Carmeli Y, et al. Fluoroquinolones and the risk for methicillin-resistant Staphylococcus aureus in hospitalized patients. Emerg Infect Dis 2003; 9: 1415-22.
11. Centers for Disease Control and Prevention. Methicillin-resistant Staphylococcus aureus skin or soft tissue infections in a state prison-Mississippi, 2000. MMWR Morb Mortal Wkly Rep 2001; 50: 919-22.
12. Kallen AJ, Driscoll TJ, Thornton S, Olson PE, Wallace MR. Increase in community-acquired methicillin-resistant Staphylococcus aureus at a Naval Medical Center. Inf Cont Hosp Epi 2000; 21: 223-6.
13. Kazokova SV, Hageman JC, Matava M, Srinivasan A, Phelan L, Garfinkel B, et al. A Clone of Methicillin-Resistant Staphylococcus aureus among Professional Football Players. N Engl Med 2005; 352:468-75.
14. Benjamin HJ, Nikore V, Takagishi J. Practical Management: Community-Associated Methicillin-Resistant Staphylococcus aureus (CA-MRSA): The Latest Sports Epidemic. Clin J Sport Med 2007; 17: 393-7.
15. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing; Twenty first Informational Supplement. [online] 2011 [cited 2014 Dec 14]. Available from: URL: http://antimicrobianos.com.ar/ATB/wp-content/uploads/2012/11/M100S22E.pdf
16. Archer GL, Climo MW. Staphylococcus aureus Bacteremia_ Consider the Source. N Engl J Med 2001; 344: 55-6.
17. Moniri R, Musav GA, Fadavi N. The prevalence of nasal carriage methicillin-resistant Staphylococcus aureus in hospitalized patients. Pak J Med Sci 2009; 4: 656-9
18. Tacconelli E, Venkataraman L, De Girolami PC, D\'Agata EM. Methicillin-resistant Staphylococcus aureus bacteraemia diagnosed at hospital admission: distinguishing between community-acquired versus healthcare-associated strains. J Antimicrob Chemother 2004; 53:474-9.
19. Casas I, Sopena N , Esteve M , Quesada MD, Andrés I , Matas L, et al. Prevalence of and Risk Factors for Methicillin-Resistant Staphylococcus aureus Carriage at Hospital Admission. Infect Control and Hosp Epidemiol 2007; 28: 1314-7
20. Ho KM, Robinson JO. Risk factors and outcomes of methicillin-resistant Staphylococcus aureus bacteraemia in critically ill patients: a case control study. Anaesth Intensive Care 2009; 37: 457-63.
21. Von Eiff C, Becker K, Machka K, Stammer H, Peters G. Nasal carriage as a source of Staphylococcus aureus bacteremia. Study Group. N Engl J Med 2001; 344: 11-6.
22. Carnicer-Pont D, Bailey KA, Mason BW, Walker AM, Evans MR, Salmon RL. Risk factors for hospital-acquired methicillin-resistant Staphylococcus aureusbacteremia: a case-control study. Epidemiol Infect 2006; 134: 1167-73
23. Laupland KB, Ross T, Gregson DB. Staphylococcus aureus Bloodstream Infections: Risk Factors, Outcomes, and the Influence of Methicillin Resistance in Calgary, Canada, 2000-2006. J Infect Dis 2008, 198: 336-43
24. Hageman JC, Uyeki TM, Francis JS, Jernigan DB, Wheeler JG, Bridges CB, et al. Community-acquired pneumonia caused by Staphylococcus aureus, 2003-04 influenza season. Emerg Infect Dis 2006; 12: 894-9.
25. Como-Sabetti KJ, Harriman KH, Fridkin SK, Jawahir SL, Lynfield R. Risk factors for community-associated Staphylococcus aureus infections: results from parallel studies including methicillin-resistant and methicillin-sensitive S. aureus compared to uninfected controls. Epidemiol Infect 2011; 139: 419-29
26. Sacar S, Sayin K S, Turgut H, Cevahir N, HircinCenger D, Tekin K. Epidemiology and associated factors for Nosocomial methicillin-resistant Staphylococcus aureus infection in a tertiary-care hospital. Epidemiol Infect 2010; 138: 697-701
27. Tentolouris N, Petrikkos G, Vallianou N, Zachos C, Daikos GL, Tsapogas P, et al. Prevalence of methicillin-resistant Staphylococcus aureus in infected and uninfected diabetic foot ulcers. Clin Microbiol Infect 2006; 12: 186-9.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




