Jabeen Fayyaz ( Department of Emergency Medicine, Aga Khan University Hospital, Karachi, Pakistan. )
Arshalooz Rehman ( Department of Paediatric and Child Health, Aga Khan University Hospital, Karachi, Pakistan. )
Akhter Hamid ( Hospital of Sick Kids, Toronto, Canada. )
Munawar Khursheed ( Department of Emergency Medicine, Aga Khan University Hospital, Karachi, Pakistan. )
Nukhbha Zia ( Department of Emergency Medicine, Aga Khan University Hospital, Karachi, Pakistan. )
Asher Feroze ( Department of Emergency Medicine, Aga Khan University Hospital, Karachi, Pakistan. )
March 2014, Volume 64, Issue 3
Original Article
Abstract
Objective: To determine the signs and symptoms of acute bacterial meningitis (ABM) in different age grops of a paediatric population.
Methods: The retrospective study comprised patients who had been admitted through the Emergency Department of Aga Khan University Hospital, Karachi with the relevant diagnosis from September 2009 to September 2011. Case record forms were used to collect data from patient files. Data was collected using variables such as age, gender, presenting complaints, clinical signs and symptoms, computed tomography scan findings and final outcome of patients. There was a minimal risk of breach in patient confidentiality. SPSS 19 was used for data analysis.
Results: A total of 192 patietns were enrolled. The presenting complaint in 165 (86%) patients was fever; vomiting in 93 (48.43%); and 49 (52.68%) of them were more than 5 years old. Irritability was present in 54 (28.12%) children, of whom 27 (50%) were less than one year. Fits were present in 47 (24.47%) cases out of which 21 (44.68%) were less than one year. Neck stiffness and signs of meningeal irritation, Kerning\'s sign and Brudzincski\'s sign, were present in 53 (27.60%) patients; 26 (13.54%); and 18 (9.3%) respectively. These signs were more common in children over 5 years of age, reflected by 29 (54.7%), 16 (61.5%) and 11 (61.11%) patients respectively. On presentation, headache was found in 77 (40.10%) children among whom 56 (72.72%) were over 5 years. Besides, 151 (78.6%) patients required admission to the ward, while 40 (20.8%) were admitted in High Dependancy Unit/critical care units. Adverse outcome was observed in 6 (3.12%) patients.
Conclusion: Younger children with acute bacterial meningitis presented with non-specific signs and symptoms. Headache and signs of meningeal irritation were common findings in children over 5 years.
Keywords: Acute bacterial meningitis, Children, Emergency department, Pakistan. (JPMA 64: 296; 2014).
Introduction
Acute bacterial meningitis (ABM) is a leading cause of childhood febrile illnesses.1 The World Health Organisation (WHO) estimated that annually BM causes at least 1.2 million cases worldwide and of those 135,000 result in deaths.2 ABM is among the top 10 causes of infection-related deaths worldwide.3 Despite the advancement in diagnostic and treatment modalities, morbidity and mortality is still high in developing countries. The case fatality rate in India and other developing countries has been reported as 16-30%.4 Data from Pakistan had shown complication rate of 57% in children with ABM.5 Even if the child survives the acute illness, morbidity in the form of sequelae such as seizure disorders, hearing loss and hydrocephalus has been reported in one-third of survivors.6,7
In children, the diagnosis of ABM is difficult and challenging because of non-specific signs and symptoms. Clinical presentation of ABM usually differs with the age of patients, the infecting organism as well as the duration of symptoms. Children less than one year usually present with fever, irritability or drowsiness, excessive crying, vomiting, reluctant to feed and other non-specific signs and symptoms. Older children may have headache, vomiting, neck pain on presentation.8 Signs of meningeal irritation are considered highly indicative of meningitis. These signs include neck stiffness, Kernig\'s sign and Brudzincski\'s sign. The presences of these signs are not pathognomonic for meningitis. In about 20% of children these signs could be absent.9 Definitive diagnosis of ABM is often dependent on cerebrospinal fluid (CSF) detail report (D/R) and its culture by performing lumbar puncture.1 Common practice is to start the antibiotic till the CSF culture becomes available in 48-72 hours.10
Data from Pakistan on the clinical symptoms in different age groups in ABM is limited. Two studies from Lahore on patients with seizure episode presenting to emergency department had found that 13% and 39% of them had central nervous system (CNS) infection respectively.11,12 Data from Hyderabad had shown that ABM comprises 1.57% of total admitted patients.13 Other studies from Pakistan had focused on aspects like complications and management and the organism involved.14 The signs and symptoms in children in accordance with different age groups are still not well studied in our setting. Therefore, the current study was planned to determine the clinical presentation and outcome of ABM and to compare the signs and symptoms according to different age groups in children visiting the emergency department of a tertiary care hospital. We also looked into the length of stay and level of admission required along with the final outcome of children admitted from emergency department with ABM diagnosis.
Patients and Methods
The study was conducted at the Emergency department (ED) of Aga Khan University Hospital (AKUH), Karachi, from September 2009 to September 2011. This study was a retrospective chart review of all admitted patients in whom lumbar puncture was done because of suspected ABM. The patients were included if CSF D/R showed white blood cells (WBC) count >10/cmm with Neutrophil count >70%.
All patients visiting the ED during the study period who were admitted to the ward with lumbar puncture-proven ABM formed the study population.
All children aged 1 month to 14 years presenting with CSF D/R proven diagnosis of ABM were included, while children with other CNS diseases, like brain tumours, intracranial bleeds, structural malformations of brain, and those who were immune-compromised were excluded.
The study was approved by the institutional ethical review committee.
All information collected was entered in a predesigned proforma. The variables included were the basic demographics and signs and symptoms like fever, vomiting, irritability, drowsiness, seizures, headache, photophobia, bulging fontanel, neck stiffness, signs of meningeal irritations like Kernig\'s and Brudzincski\'s sign and Glasgow Coma Scale (GCS) at the time of presentation. The patients were divided into 3 age groups: less than 1 year; 1-5 years; and more than 5 years. The rationale was that the clinical sign and symptoms are different in different age groups.15,16 The percentages were than calculated according to different age groups. Duration of symptoms, level of admission, hospital length of stay and final disposition were also recorded and compared in various age groups.
Data was analysed using SPSS version 19.0. Descriptive statistics were used to describe patient\'s demographics and were expressed in frequencies and percentages. Mean and standard deviation were computed for continuous variables.
Results
Of the total 192 patients, there were 134 (69.8%) males and 58 (30.2%) females. The mean age of the children was 4.8±4.14 years. There were 53 (27.6%) children of less than one year of age, 61(31.8%) were between 1-5 years of age, and 78(40.6%) were more than 5 years (Table-1).
The common presenting complains were fever (n=163; 85.8%), vomiting (n=93, 48.4%), headache (n=77, 40.1%), irritability (n=54, 28.1%), fits (n=47, 24.5%) and drowsiness (n=26, 13.5%). Common signs observed in these patients were focal neurological signs in 1 (0.5%), bulging fontanels in 14 (7.3%), neck stiffness in 53 (27.6%), Kernig's sign in 26 (13.5%), Brudzincski's sign in 18 (9.4%), reduced or deteriorating level of consciousness in 4 (2.1%), dilated poorly reactive pupils in 2 (1%), hypertension in 1 (0.5%), and irregular respiration in 1 (0.5%) patient. The GCS score was more than 13 in 148 (77.1%) patients while less than 8 GCS score was observed in 7 (3.6%) patients. Of the 155 patients, Fundoscopy was done on 49 (25.5%) patients. Normal findings were had in 44 (89.7%), while the rest of the patients (n=5; 10.3%) had an abnormal finding on Fundoscopy.
Among all the admitted patients, 152(79.2%) were admitted in ward, 25 (13%) in surgical care unit (SCU) and 15(7.8%) in Paediatric Intensive Care Unit (PICU). Of the 192 patients, 179(93.2%) were discharged, 7 (3.6%) left against medical advice (LAMA) and there were 6 (3.1%) deaths.
Symptoms and admission patterns were also compared for the three age groups (Table-2, Figure).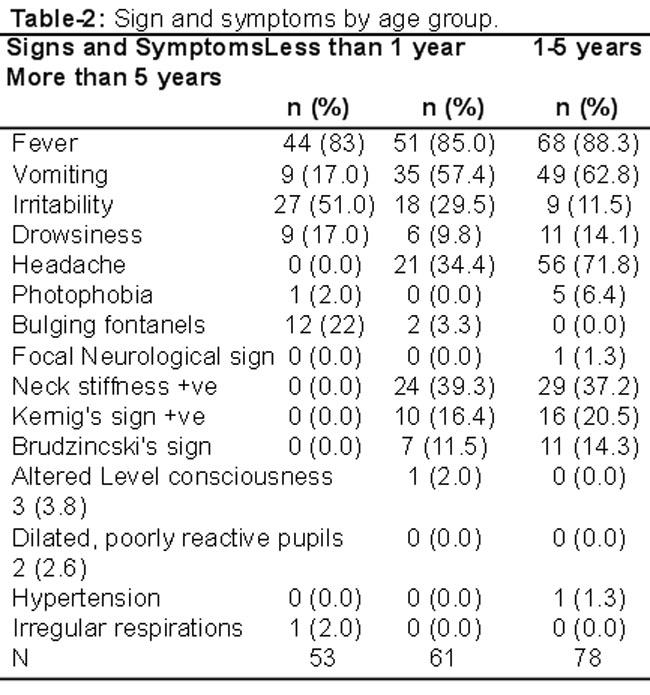

Discussion
Results show that most (29.7%) children diagnosed as having ABM were infants less than one year and were more sick requiring high dependency unit admission. Children who were diagnosed as ABM had fever as the major complaint in all age groups. The typical signs of meningeal irritation like nuchal rigidity, Kernig\'s sign and Brudzincski\'s sign are not very specific to meningitis as they could appear in upper respiratory tract infections, pneumonia and other bacterial infections.17,18 Hard clinical signs and symptoms of ABM are present in older age groups. The younger the patient, there is less likelihood of getting specific neurological signs.19
ABM is one of the leading causes of early death if not suspected and diagnosed promptly.20 The fatality rate is high because of lack of immunisation covering the bacterial pathogen. The recent inclusion of Hemophilus influenza B vaccine in expanded programme for immunisation (EPI) holds the hope of decreasing cases with this organism, especially in children less than 5 years.21 However, the pneumococcal and meningococcal infections still prevail in our community.22,23 Early diagnosis depends on early recognition of signs and symptoms, and intervention is required within a few hours of diagnosis. Emergency room management is the key in improving the survival rate in children. The recognition of ABM in ED is a difficult task because of non-specific signs and symptoms and literature review has shown that there is no consensus on any clinical prediction rules that could predict the possibility of ABM in children.24 Our study has reflected the importance of clinical signs and symptoms according to age groups which would help the clinician to recognise them early. Investigations take a few hours and at times even labs are not very suggestive of this dreadful condition, leaving the ED physician in a dilemma to start the treatment or not.25
Haemodynamic instability indicates either disseminated infection or raised intracranial pressure (ICP). Prior antibiotic usage is still a problem in our community. We strongly feel that in era of modern technology none of the investigations can rule out the possibility of meningitis. Good history, focussed clinical examination and high index of suspicion are still the key to diagnose ABM.
In terms of limitations it was a single-centre retrospective chart review and its results cannot be generalised. The age groups were divided according to the experts\' panel and the reference in the literature is not well developed. Confounding factors like prior antibiotic therapy had been taken into account.
Conclusion
Younger children with ABM presented with non-specific signs and symptoms. Headache and signs of meningeal irritation were common findings in children over 5 years of age.
References
1. Sa¡ez-Llorens X, McCracken GH. Bacterial meningitis in children. Lancet 2003; 361: 2139-48.
2. Black RE, Cousens S, Johnson HL, Lawn JE, Rudan I, Bassani DG, et al. Global, regional, and national causes of child mortality in 2008: a systematic analysis. Lancet 2010; 375: 1969-87.
3. Chaudhuri A, Martin PM, Kennedy PGE, Andrew Seaton R, Portegies P, Bojar M, et al. EFNS guideline on the management of community�acquired bacterial meningitis: report of an EFNS Task Force on acute bacterial meningitis in older children and adults. Eur J Neurol 2008; 15: 649-59.
4. Sallam AK. Etiology and presentation of acute bacterial meningitis in children at Al-Thawrah Hospital, Sana\'a, Yemen. J Ayub Med Coll Abbottabad 2004; 16: 40-3.
5. Rashid J, Rehman Z, Shahzad U, Khan MMN. Frequency of acute complications in pyogenic meningitis in children. Pak Paed J 2008; 32: 208-14.
6. Neuman HB, Wald ER. Bacteral Meningitis in Childhood at the Children\'s Hospital of Pittsburgh: 1988-1998. Clin Pediatr 2001; 40: 595-600.
7. Laeeq A, Ali L, Ahmad M. Assessment of hearing loss after childhood meningitis Pak Paed J 2002; 26: 125-31.
8. Singhi S, Singhi P. Baranwa AK. Bacterial meningitis in children: critical care needs. Indian J Pediatr 2001; 68: 737-47.
9. Oliver LG, Harwood-Nuss AL. Bacterial meningitis in infants and children: a review. J Emerg Med 1993; 11: 555-64.
10. Bryant K, Marshall GS. Most cerebrospinal fluid cultures in children with bacterial meningitis are positive within two days. Pediatr Infec Dis J 1999; 18: 732-3.
11. Raza AB, Naz F, Malik MA, Bano I. Common causes of seizures in children presenting in paediatric medical emergency. Pak Paed J 2010; 34: 133-8.
12. Rana MN, Hussain RR, Amir K, Samdani A, Rathore AW, Ahmed TM. Seizures in children with fever presenting in accident and emergency department of Children\'s Hospital, Lahore. Pak Paed J 2010; 34: 204-7.
13. Afzal M. Children presenting with seizures- A hospital based study. J Coll Physicians Surg Pak 1999; 9: 132-5.
14. Ahmad I, Haq I, Rehman H, Khattak AA, Khan FM. Bacterial meningitis in children. J Postgrad Med Inst 2004; 18: 523-8.
15. Amarilyo G, Alper A, Ben-Tov A, Grisaru-Soen G. Diagnostic accuracy of clinical symptoms and signs in children with meningitis. Pediatr Emerg Care 2011; 27: 196-9.
16. Ward MA, Greenwood TM, Kumar DR, Mazza JJ, Yale SH. Josef Brudzinski and Vladimir Mikhailovich Kernig: signs for diagnosing meningitis. Clin Med Res 2010; 8: 13-7.
17. De Cauwer HG, Eykens L, Hellinckx J, Mortelmans LJ. Differential diagnosis between viral and bacterial meningitis in children. Eur J Emerg Med 2007; 14: 343-7.
18. Oostenbrink R, Moons KG, Theunissen CC, Derksen-Lubsen G, Grobbee DE, Moll HTA. Signs of meningeal irritation at the emergency department: How often bacterial meningitis? Pediatr Emerg Care 2001; 17: 161-4.
19. Curtis S, Stobart K, Vandermeer B, Simel DL, Klassen T. Clinical features suggestive of meningitis in children: a systematic review of prospective data. Pediatrics 2010; 126: 952-60.
20. Salih KEMA, El Nageeb Suliman Saeed MS, Karsani REA, Salih AA, Ibrahim SA. Characteristics of morbidity and mortality in children less than 5 years of age with acute bacterial meningitis in Omdurman, Sudan. J Medicine Medical Sci 2011; 2: 920-4.
21. Zaidi AK, Khan H, Sherali AR, Lasi R; Sindh Meningitis Study Group. Burden of Haemophilus influenzae type b disease in Pakistani children. East Mediterr Health J 2010; 16: 590-4.
22. Chugh Y, Kapoor AK, Kastury N, Srivastava AK, Bhargava A, Sharma A. Study of antimicrobial sensitivity pattern of Gram-positive CSF isolates among children suffering from septic meningitis in a tertiary care hospital. J Indian Acad Clin Med 2011; 12: 274-82.
23. Zaidi AK, Khan H, Lasi R, Mahesar W; Sindh Meningitis Group. Surveillance of pneumococcal meningitis among children in Sindh, southern Pakistan. Clin Infect Dis 2009; 48: S129-35.
24. Maguire JL, Kulik DM, Laupacis A, Kuppermann N, Uleryk EM, Parkin PC. Clinical prediction rules for children: a systematic review. Pediatrics 2011; 128: e666-e77.
25. Stevenson M. To Tap or Not: Bacterial Meningitis & Complex Febrile Seizures. AAP Grand Rounds 2010; 24: 41-52.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




