Farooq Ali ( Department of Pathology, Hi-Desert Medical Center, Joshua Tree, California 92252, USA. )
June 1987, Volume 37, Issue 6
Special Communication
Abstract
It was observed that individuals at high risk for AIDS who are exposed to domestic cats may not develop AIDS or ARC. This may be due to protection induced by the Feline Leukemia Virus which is widespread in cats and has not been demonstrated to be pathogenic for humans. Recently developed FeLV subunit vaccines have been recognised as useful models for potential human retrovirus vaccines, but the microstructure of HIV is not conducive to subunit immunogenecity. Due to probable conserved inter-species antigenic determinants and virally-coded membrane neoantigens between FeLV and HIV, FeLV vaccine itself (Leukocell) may be effective in humans. Trials with Leukocell or attenuated strains of FeLV grown in human cell cultures, in simians, and if protective against AIDS and/or retrovirally induced neoplasia, in high-risk human populations is suggested.
AIDS VACCINE
The author’s interest in the Acquired Immunodeficiency Syndrome (AIDS) stems from a conversation in 1983 with Professor Waterson1 at the Royal Postgraduate Medical School in Hammersmith, London, when the similarity between a recently described human T-cell leukemia virus and the Feline Leukemia Virus (FeLV) was discussed. This author practiced for the last seven years in or adjacent to communities in Southern California which have a high incidence of AIDS and where individuals follow high-risk behaviours (for AIDS) with hitherto relative freedom from social persecution or legal prosecution, viz. Hollywood, Los Angeles, some high desert communities in and around Joshua Tree and cities in the Imperial Valley including El Centro where there are also high rates of drug abuse. The initial idea was to gather data for a speculation, subsequently abandoned, that the Human T-Cell Lymphotropic Virus/Lymphadenopathy-Associated Virus/Human Immunodeficiency Virus (HTLV-III/LAV/HIV) may have originated from a mutated FeLV instead of simian-human transmission as is now widely accepted.
Fifteen reportedly homosexual or bisexual males whom the author knew of were followed informally over periods varying from two to seven years and were questioned as to whether they were exposed to pet cats. Ten of these who either had pet cats or whose regular partners did, did not develop AIDS or AIDS-related complex (ARC).
Two individuals who did not have clinical symptoms of AIDS or ARC but had a history of substance abuse, and had positive HTLV-III serology reported that they “hated cats”, but kept dogs.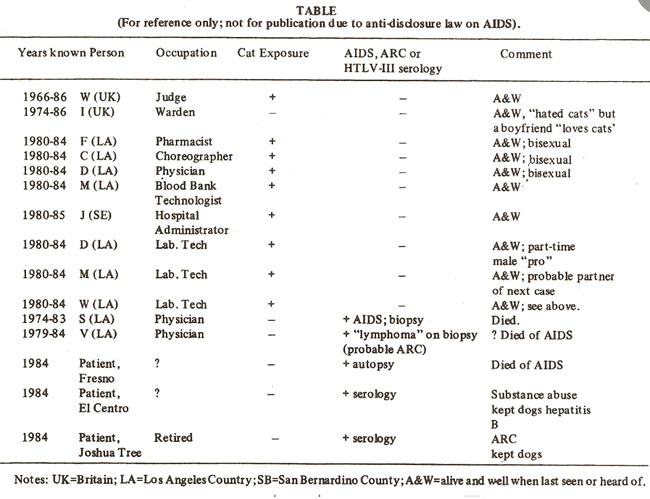
The three remaining individuals who died of AIDS-related complications reportedly did not have pets; these cases were confirmed by serology, biopsy or autopsy. As all of these persons except for three patients in the “positive” group were professionals, colleagues or health-care workers and due to strict anti-disclosure laws in the State of California on AIDS it was not feasible to do a formal case-control retrospective study with statistical analysis of the significance of the negative association. However, due to the urgency inherent in the possibility of a global AIDS pandemic, this is presented as a clinical observation and with reference to animal models, so that retrospective studies may be done in areas where AIDS is endemic, and if the association is confirmed, simian trials of a FeLV or subunit based vaccine may be carried out.
It is. suggested that the individuals who did not get AIDS may have been protected by exposure to the ubiquitous ‘FeLV. About 30% to 50% of all domestic cats carry the FeLV provirus without showing Signs of disease2 and half of the cats which roam freely have episodes of FeLV viremia3. It is postulated that a class of retroviruses oncogenic in mesodermal tissues in different species, including FeLV-strains and HTLVs, evolved from a common ancestor and therefore share antigenic determinants4, some of which are conserved between species and may include virally coded membrane neoantigens. Citing a parallel to the DNA variola-vaccinia viruses, where passage through different hosts alters pathogenecity while retaining immunogenecity, FeLV which has passed through felines is non-pathogenic to humans, whereas the closely related Simian AIDS (SAIDS) virus and HIV passed through primates show cross-pathogenecity in humans and monkeys.
FeLV was discovered by Jarrett et a15 in 1964, and on the basis of experimental transformation of human and canine cell lines in tissue culture was classified by the National Cancer Institute as an agent of moderate oncogenic hazard potential for humans, in the 1970s; but subsequent studies have not shown pathogenecity to humans6. Studies in cats show many similarities between FeLV-induced disease and human AIDS, including production of a feline acquired immune deficiency syndrome (FAIDS), induction of various mesodermal malignancies, opportunistic infections2,7 and gastrointestinal and nervous system involvement2,8. However, host patho. genecity is significantly different in that most cats develop protective antibodies and do not develop disease. Ott2 describes three types of antigens in FeLV: virion antigens including internal group-specific and envelope type-specific antigens; cell surface antigens; and soluble antigens. Antibodies against internal antigens are produced in small amounts and are not protective. Recent efforts at producing vaccines against mammalian retroviruses including HIV have been focuied on membrane and soluble antigen subunits: these have been summarized by Hunsmann9. These consist of antibodies against envelope polypeptides, in which some antigenic determinants are conserved between mammalian species (gp 70), expressed on membranes of infected cells as well as virus particles and other immune stimulating complexes (iscoms) 10. However, Hunsmann9 notes that purified HTLV viruses are not a good source of envelope glycoprotein complexes; probably corresponding to a lack of knobbly protrusions such as seen on FeLV on electron microscopy. Osterhaus et al9 reported in 1985 that a subunit vaccine consisting of the gp7O/85 of FeLV was effective in preventing a leukemic viremia in cats subsequently challenged with FeLV and elicited multiple antibodies including a monoclonal antibody to the gp7O/85 epitope, capable of neutralizing all three subtypes of FeLV. They proposed this as a model for iscom vaccines against other mammalian retroviruses.
Subsequently, an effective FeLV vaccine capable of inducing neutralising antibodies was developed for veterinary use based on soluble tumor antigens in FeLV infected cells (STAV) including gp7O, p27, p15, p12 and plO; as well as Feline oncovirus associated membrane antigens (FOCMA); which were harvested from infected transformed cell lines; and produced and marketed commercially by Smith Kline as “Leukocell”; this work has been reviewed by Mastro et al.11
Recent nucleotide sequencing and gene mapping on AIDS viruses by Hahn et al12 showed that in HIV there is greatest genetic variation in the gag and em’ oncogenes; and also that within the em’ gene, there are hypervariable regions interspersed with areas of strong conservation. It is postulated that the hypervariable regions allow for genetic drift by incorporation of “normal” human cell membrane antigens into the envelope which protect adjacent virally-coded membrane antigens; however, if the former are feline rather than human, it may facilitate recognition of the latter by human NK cells or unaffected T4+ lymphocytes.
Hahn et al12 also showed that in the gag coding for p15, only about 5% of the base sequences differ in virus strains. This may mean that there is enough homology between FeLV p15 present in STAV vaccines and AIDS p15 for the former to stimulate a response to the latter. Again, Hunsmann9 notes that gp 85 on viral surfaces is a heterodimer linked via disulfide bridges to a p15 which he characterizes as a membrane inserted env product; and that antigenic determinants responsible for protection by vaccines are located on gp7O which is more immunologic when complexed to gp8S. He also cites animal studies in which heterologous AKR virus and FeLV glycoprotein complexes (gp7O/85-pls) immunizations were effective in preventing against Friend virus leukemia in mice. Further, as Shaw and Harper (cited by Hahn et al12) have shown that
only a small proportion of 14+ lymphocytes are infected in patients with chronic infection, FeLV subunit vaccines may be useful in AIDS carriers or patients with ARC.
The recent gene mapping studies by Hahn et al12 also showed that there is genetic variation with time due to mutations in HTLV-IlI in patients with AIDS, suggesting that strains evolve in parallel from a common progenitor virus, but that some type of interference mechanism prevents simultaneous infection by more than one major genotypic form of virus.
The present author suggests that as some tumor antigens may be conserved between FeLV and HIV, and interfering mechanisms are generated, Leukocell or. FeLV cultured in human cell lines may be effective vaccines against AIDS. Also, Ott2 notes that development of FOCMA antibodies in cats enables them to resist or even reject FeLV induced tumors, even though the virus is not neutralized. This suggests that Leukocell or monoclonally derived anti-membrane neoantigen antibodies may be useful even in patients who have developed Kaposi’s sarcoma or other malignancies secondary to AIDS; and possibly in other human neoplasms expressing viral neoantigens, or in whom a retroviral etiology is suspected. It is therefore suggested that “Leukocell” or similar subunit vaccines made from FeLV-transformed human cell lines, or attenuated strains of FeLV itself be tested in simians and if effective against retrovirally induced neoplasms or protective against subsequent challenge with the simian SAIDS virus, in human populations atbigh risk.
DNA viruses such as herpes or papovaviruses which are often associated with AIDS13, may be helper viruses. Therefore, prospective trials may be constructed to include Acyclovir therapy as well as vaccination.
ACKNOWLEDGEMENTS
This paper is dedicated to the memory of the late Professor A.P. Waterson, virologist, teacher, and friend. The author wishes to thank Mr. Y. Yasin, M.A. (Oxon) for technical assistance. This paper was read at the 2nd International Conference of the Pakistan Association of Pathologists, 5th - 8th December, 1986, Karachi.
REFERENCES
1. Waterson, A.P. Human cancers and human viruses. Br. Med. J., 1982; 284:446.
2. Ott, R.L. Feline leukemia virus infection, in Feline medicine Edited by Pratt, D.W. Santa Barbara, USA, American Veterinary Publications, 1985, p. 123.
3. Francis, D.P., Essex, M. and Hardy, W.D. Jr. Excretion of feline leukaemia virus by naturally Infected pet cats. Nature, 1977;269:252.
4. Hardy, W.D. Jr., Hirshaut, Y, and Hess, P. Detection of the feline leukemia virus and other mammalian oncornaviruses by immunofluorescence, in unifying concepts of leukemia. Edited by Dutcher, R.M. and Chieco-Bianchi, L. Basel, Karger, 1973; p.7 78.
5. Jarrett, W.F., Crawford, E.M., Martin,W.B. et al. A virus-like particle associated with Ieukaemia (Lymphosarcoma). Nature, 1964; 202 : 567.
6. Hardy, W.D. Jr. The feline leukemia virus. J. Am. Anim Hosp. Ass., 1981; 17:951.
7. Rojko, J.L. and Olsen, R.G. The iminunobiology of the feline leukemia virus. Vet. Immunol. !mxnunopathol., 1984; 6:107.
8. Jarrett, J.O. Feline leukaemia virus, in Feline medicine and therapeutics. Edited by Chandler, E.A. Hilbery, A.D.R. and Gaskefi, C.J. London, Blackweli, 1985, p. 271,1985: 271-83.
9. Hunsmann, G. Subunit vaccines against exogenous retroviruses; overview and perspectives. Cancer Res, 1985; 45 (Suppl.) : 4691 s.
10. Osterhaus, A., Weijer, K., Uytdehaag, F., Jarrett,0., Sundquist, B. and Morein, B. Induction of protective immune response in cats by vaccination with feline leukemia virus iscom. J. Immunol., 1985;135 :591.
11. Mastro, J.M., Lewis, M.G., Mathes, L.E. et a!. Feline leukemia vaccine; efficacy, cxnitents and probable mechanism. Vet. Immunol. Immunopathol., 1986; 11 :205.
12. Hahn, Bil., Shaw, G.M., Taylor, M.E. et al. Genetic variation in HTLV-IH/LAV over time in patits with AIDS or at risk for AIDS. Science, 1986; 232: 1548.
13. Frklman-Kien, A.E. Viral origin of hairy leukoplakia. Lancet, 1986; 2 : 694.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




