Mirza Naqi Zafar ( Research and Diagnostic Centre, 7/14, Rimpa Plaza, Clinic Side, M.A. Jinnah Road, Karachi. )
S.H. Manzoor Zaidi ( Department of Radiotherapy, Jinnah Postgraduate Medical Centre, Karachi. )
July 1992, Volume 42, Issue 7
Original Article
ABSTRACT
Acid phosphatase (AP) reaction is a specific cytochemical marker for T-cell lymphoblastic leukaemia (1-ALL). Twenty six cases of ALL were diagnosed as 1-ALL employing cytochemical profiles including APand sheep red blood cell rosettes. Three distinct patterns of AP reaction were observed. On one end of the spectrum, 100% of the positive blasts showed the classical dot- like reaction while on the other 100% showed a scattered reaction. Between the two a mixed pattern was observed with 30- 80% of the positive blasts showing the dot-like reaction while the rest showed scattered pattern. Dot-like pattern showed L1 morphology, high counts and low SRBC rosettes while scattered showed L2 morphology, low counts and high SRBC rosettes. The pattern observed in our sedes differs from the dot-like reaction observed in T-ALL in the western series. We recommend that this pattern must be borne in mind when employing AP reaction for T- ALL diagnosis (JPMA 42: 151, 1992).
INTRODUCTION
A localized strong paranuclear acid phospliatase reaction is characteristic of human fetal thymocytes1 and in blast cells in the majority of ALL cases with T-cell markers2. In a comparative study 90% of the cases of ALL with T-lineage were strongly positive for acid phosphatase in contrast to 2% in common ALL and 10% in null-ALL3. A number of studies have shown the value of this reaction in diagnosing T-ALL with incomplete or immature T- membrane phenotype4,5. A number of workers have used cid phosphatase reaction for diagnosis of T-ALL in Pakistan6,7 and we have reported a high frequency of T-ALL6,8 in our population as compared to western population9. We have observed that a strong localized reaction is not always the rule and there is a considerable variation of acid phosphatase reaction pattern in T- ALL. This paper describes these variations in T-ALL and compares them with FAR classification of ALL, total leucocyte count, membrane markers and age of the patient in order to provide a pattern related diagnostic guideline for T-ALL.
PATIENTS AND METHODS
Pretreatment samples of bone marrow and peripheral blood were collected from twenty-six cases of ALL, 14 children ages less than 15 and 12 adults ages more than 15. Romanosky stained films were looked at and FAR variants identified using criteria outlined by FAR10. Haematological parameters included haemoglobin, leucocyrte and platelet counts and differential counts. Mononuclear cells from peripheral blood and bone marrow were separated on “Lymphoprep” (Flow Laboratories) according to Boyum11. The following cytochemical reactions were performed using Sigma Kits. Acid phosphatase, P.A.S., mycloperoxidase and alphsnapthyl esterase12-15. Sheep red blood cell rosettes were performed using fresh cells16. Using morphology criteria, cytochemistry and membrane markers, these 26 cases were diagnosed as T-ALL. Photomicrographs were with NIKON microscope under oil emersion using Agfa ASA 100 B/W film.
RESULTS

Table I shows the cytochemical profiles on bone marrow and separated blasts cells from peripheral blood. Results are expressed as % blasts positive. It also shows % sheep red blood cell rosettes. These are % of blasts cells forming rosettes with three or more red cells. 60 to 100% of the blasts were positive for acid phosphatase. Blasts showed either a dot-like reaction or scattered activity. Low activities were seen in P.A.S.. peroxidase and NASA. % SRBC rosettes were negative in one case while upto 90% of the cells formed rosettes in others. SRBC negative case was positive for acid phosphatase. Acid phosphatase activity showed a graded pattern. Some cases showing 100% of the positive blasts giving the classical dot-like reaction (Figure 1)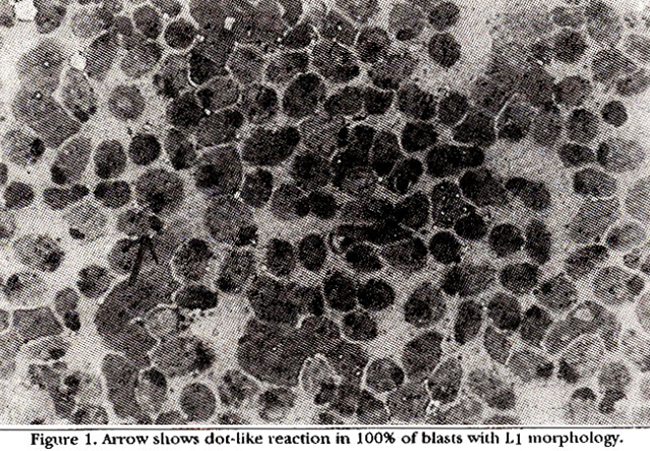
to 100% of the positive blasts showing a scattered reaction (Figure 3).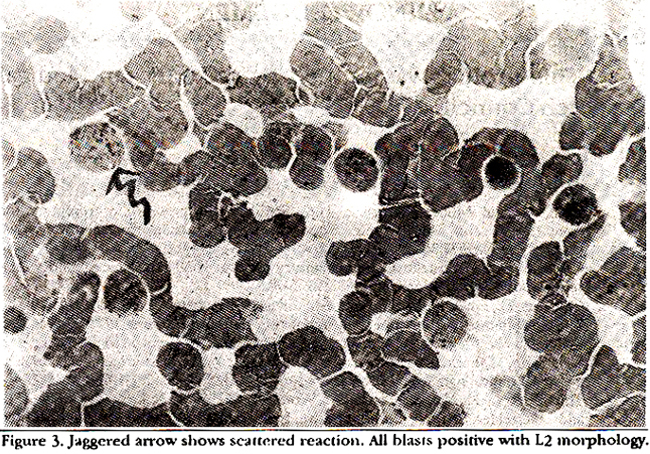
Between these two distinct patterns a mixed pattern was observed where 30-80% of the positive blasts gave the dot- like reaction and conversely 70-20% showing the scattered reaction (Figure 2).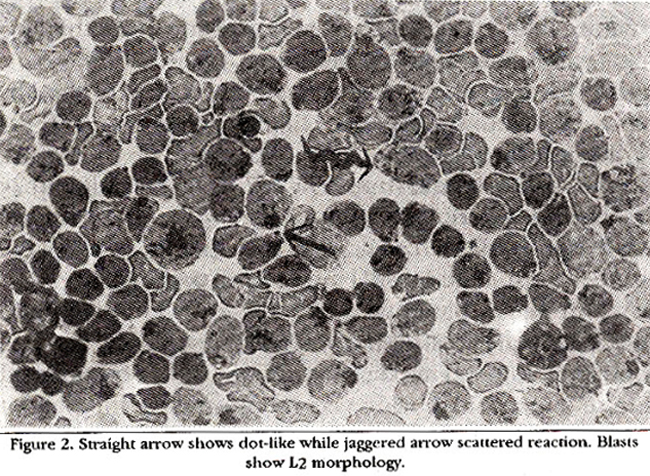
The twenty-six cases of T-ALL were grouped according to the pattern of reaction.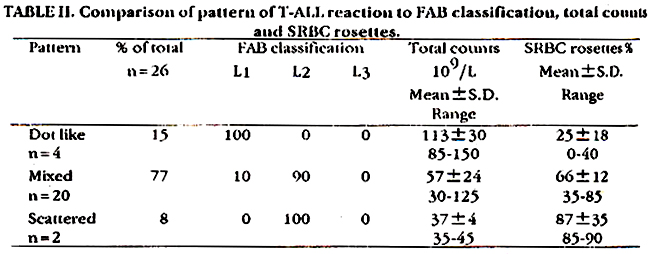
Table II shows the FAR classification, total counts and SRBC rosettes %. The dot-like reaction showed L1 morphology, high counts and low rosettes while scattered reaction showed low counts and high rosettes and L2 morphology. Mixed pattern showed L2 morphology and medium counts and rosettes. Comparison of morphology of blasts and reactivity patterns for adults and children is given in Table III.
This also shows predominance of L1 in the dot-like reaction and L2 in the mixed and scattered reaction in both age groups. However the frequency of dot-like reaction 21% is higher in children as compared to adults 8.5%.
DISCUSSION
T-ALL is associated with factors known to be of bad prognosis in ALL. Meningeal leukaemia, low remission rates and high relapse rates17 are common. high frequency of T-ALL in Pakistan6 and Asian populations in general18 necessitates its early diagnosis for effective therapy. Acid phosphatase reaction, a specific cytochemical markers for T-ALL fulfils this diagnostic need in Pakistan specially when more specific monoclonal antibody surface markers19,20 are economically non-viable for routine laboratories and remain essentially a research tool. ALI T-ALL cases in our series were positive for acid phosphatase, however the classifical dot-like reaction was neither observed in all cases nor seen in all the blasts. The description of the three patterns we have observed is worthwhile considering. Monoblastic leukaemias often give scattered pattern though size of granuoler activity is much smaller than T-ALL blasts21. Negativity of NASA reaction in T-ALL and SRBC rosettes further deviates confusion with monoblastic lineage. Dot-like reaction of acid phosphatase at electron microscopic level is found mainly in the golgi21 while the scattered pattern is observed in cytoplasmic lysosomes. Absence or low numbers of SRBC rosettes in the dot-like pattern suggests a more undifferentiated nature of this pattern and L1 as compared to the scattered pattern where SRBC rosettes are high and morphology is L2 The variation of the reaction pattern and the deviation from the classical dot-like reaction may be due to two reasons. Firstly a possible genetic variation as we have observed high frequency of T-ALL and secondly due to numericals. We have seen more cases than western studies. Whatever the reason this variation and the three patterns i.e., dot-like, mixed and scattered should be considered when acid phosphatase reaction is employed for T-ALL diagnosis.
ACKNOWLEDGEMENT
This project was funded by the Pakistan Medical Research Council.
REFERENCES
1. Stein, H. and Muller-Hermelink, H.K. Simultaneous presence of receptors for complement and sheep red blood cells on human (octal thymocytes. Br.J. Haematol., 1977;36:225-30.
2. Catovsky, D. and costello, C Cytochemistry of normal and leukaemic lymphocytes; a review. BasicAppL Histochem., 1980;23:255-70.
3. Catovsky, D., Cherchi, M., Gresves, M.F., Pain, C, Janossy, C. and Kay, H.E.M. The acid phosphatase reaction in acute lymphoblastic leukaemia. Lancet, 1978;1:749-51.
4. Brouet, J.C., Valensi, F., Daniel, M.T., Flandrin, G., Preued Hoome, IL and Seligmann, M. Immunological classificatinn of acute lymphoblastic leukaemia; evaluadon otita clinical significance in a hundred patients. Br.J. Haematol., 1976;33:319- 28.
5. Thiel, B., Rods, H., Netzel, B., Huhn, D., Wondisch, G.F., Haas, R.J., Bender-Gotze, C.L and Thierfelder, S. T-zell antigen positive. E-Rossetten negative akute lymphoblasten leukaemie. Blut.,1978;36:363-68.
6. Zafar, M.N. and Zaidi, S.H.M. Acid phoaphataae positive T- lymphoblaslic leukaemia in Pakistani children. J.Pak.Med.Assoc., 1985;35:6-9.
7. Alvi, E.A., Saleem, M., Ahmed, P., Rehman, Z., Anwar, M. and Ahmed H. A study of hundred cases of acute leukaemia in Northern Pakistan with reference to French-American and British (FAB) Cooperative Group Classification. Pakistan. J. Pathol., 1990;1:87-92.
8. Zafar, MN. and Zaidi, S.M.H. Multiparameter studies in T-cell acute lymphoblastic leukaemia (T-ALL) in Karachi children.). CPSP., 1991;1:20-23.
9. Benett, J.M., Catovaky, D., Daniel, MT.. Flandrin, 0., Gallon, D.A.0., Gralnick, HR. and Sultan, C The morphological classilicstion of acute lymphoblaatic leukaemis concordance among obacrvors and clinical correlations. Br.J. Haematol., 1981;47:553-61.
10. Bennett, 3M., Catovsky, D., Daniel, MT., Flandrin, C., Galton, D.A.G., Gralniclc,H.R. and Sultan, C. Proposal for the classification of acute leukaemias. Br.J. Haematol., 1976;33:451- 58.
11. Boyum, A. A one-stage procedure for isolation of granulocytes and lymphocytes from human blood cells in a 1g gravity field. Scand. J. Clin. Lab. lnvest.,1968; 21 (Suppl 97):51-52.
12. Goldberg. A.F. and Barks, T. Acid phosphatase activity in human blood cells. Nature, 1962;195:297-300.
13. Loftier, H. Indicationsand limitsof cytochemistry in acute leukaemias. Recent results in cancer research, 1973;43:57-62.
14. Kaplow, LS. Simplified myeloperoxidase stain using benzidine dihydrochloride. Blood, 1965;26:215- 19.
15. Higgy, K.E. and Burns, G.F.J. Discrimination of B.Tand null lymphocytes byeacerase cytochemistry. Scand. 3. Haematol., 1977;18:437-48.
16. WHO/IARC Workshop. Identification, enumeration and isolation of B and T lymphocytea from human peripheral blood. Scand.J. Immuno., 1974;3:521-24.
17. Ravindranath, Y., Kaplan, 1 and Zuelzer, W.W. Significance of mediastinal mass in acute lymphoblaatic leukaemia. Pediatrics, 1975:55:889-93.
18. Kamat, D.M., Gopal, R., Advani, SM. etal. Pattern of subtypes of acute lymphoblastic leukaemia in India. Leuk.Res., 1985;9:927- 34
19. Chart, LC, Pcgrsm. S.M. and Greaves, M.F. Contribution of immunophenotype to the dassification and differential diagnosis of acute leukaemia. Lances, 1985;1:475-79.
20. Foon, K.A. and Todd, R.F. Immunological classification of Ieulcaemia and lymphoms. Blood, 1986;68:1-31.
21. O\\\'Brien, H., Catovsky, D. and Costello, C. Ultrastructural cytochemistry of leukaemic cells; characterization of the early small granules of monoblasta. BrJ. Haematol., 1980;45:201-8.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




