Emad Kudhair Abbas ( Department of Anatomy and Histology & Embryology, Collage of Medicine, University of Basra, Iraq. )
November 2011, Volume 61, Issue 11
Original Article
Abstract
Objective: To investigate the histopathological changes in laboratory mice (Mus Musculus) being given doxorubicin.
Methods: The experimental study was conducted at the University of Basra between September 2009 and March 2011. Two groups with 5 mice each were used in this study. The experimental group was given 6mg/kg of doxorubicin intraperitoneal for 15 days. The control group was given distilled water for 15 days. On day 16, post-mortem was done. Biopsies were taken from the liver and intestine of both groups. Histopathologic slides were made for examination.
Results: Changes in hepatic tissue involved were inflammatory cell infiltration, necrosis and vaculolation. While the liver of the control group was normal in structure. The intestinal section of the experimental group showed decomposition in the intestinal villi, while the intestines of control mice was normal.
Conclusion: Doxorubicin is highly toxic to the liver and intestines of laboratory mice.
Keywords: Doxorubicin, Mice, Histopathological, Iraq (JPMA 61: 1108; 2011).
Introduction
The drug Doxorubicin is an antibiotic extracted from fungi called Streptomyces peucetius.1 It is highly toxic to the cells even though it remains extracellular.2
It is absorbed rapidly in the blood and reaches a level of (700-11001 mm2) in the plasma. It has the ability to bind to blood. protein at a rate of (70%).3 Then, the drug is excreted in bile at a rate of (40-50%) and in the milk at a rate of (4-5%). It has been observed that the toxic effect specially in the liver is produced 5 days after drug administration.3
This drug is used for tumour treatment, but it has a toxic effect on the mucosal epithelium of the intestines.4 Another study has shown that the drug has a toxic effect on the crypts of the intestines and proliferation of stem cells which are responsible for generation of living cells.5
Therefore, it is recommended to give doxorubicin along with other anti cancer drugs in order to reduce its toxicity.6,7 Good treatment response was seen in patients with stomach cancer, when both Fluorouracil and doxorubicin were given together.8 These drugs could lead to complications as blood poisoning and decompensation among patients with lung cancer.9 The aim of this study was to investigate the histopathological changes among treated laboratory mice with doxorubicin.
Material and Methodsand
This was an experimental study in which two groups of mice strain (Balb / c) with 5 mice each, were used. The experimental group was given 6 mg/kg of doxorubicin. Production Company (Pharmacia & Upjohn SPA) Italy, intrapritoneal for 15 days.10 The control group was given distilled water for 15 days. On day 16, post-mortem was done. Biopsies were taken from the liver and intestines of both groups. Histopathologic slides were prepared for histopathological examination.11
Results
Changes in hepatic tissue were Necrosis (Figure-1),
inflammatory cell infiltration (Figure-2)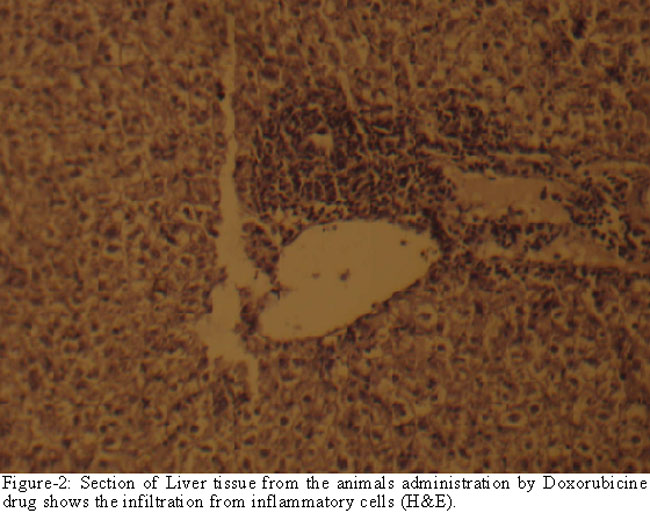
and vacuolation (Figure-3).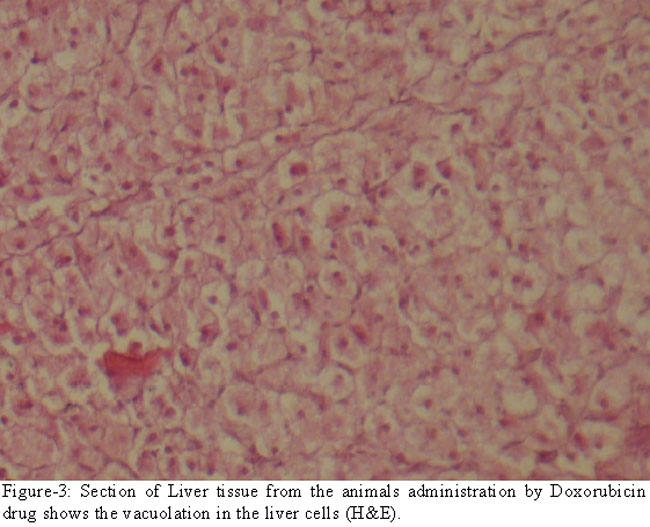
The liver of the control group was normal in structure (Figure-4).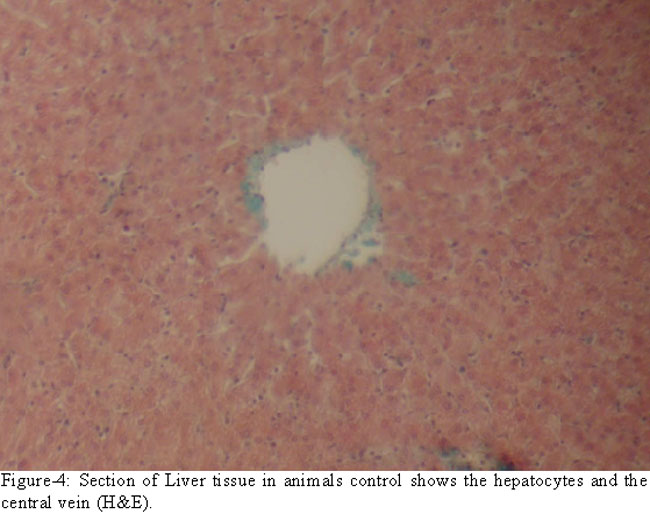
The intestinal section of the experimental group showed decomposition in the intestinal villi (Figure-6).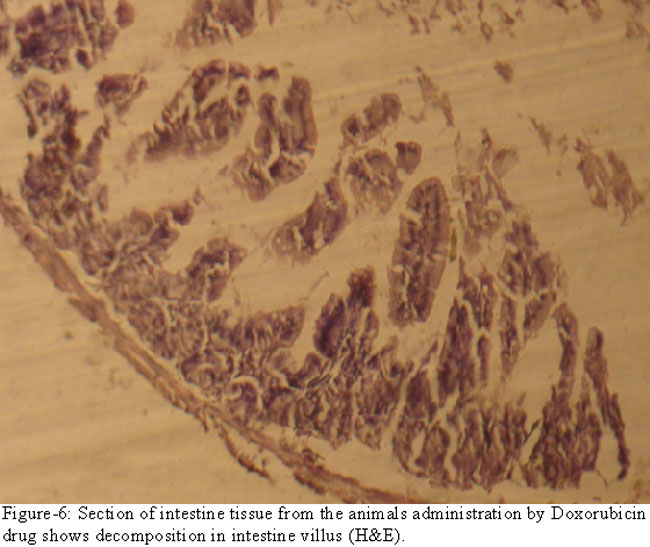
While the intestines of the control mice were normal (Figure-5).
Discussion
It is clear from the present work that the drug Doxorubicin has a toxic impact on organs especially liver and intestines.
The explanation for the observed vacuolation in the liver might be due to increase in the disruption of fat metabolism in addition to form of lipoproteins, which turn to triglycerides (Triglycerol), which later leads to fat infiltration in some organs.12
Another study has shown that the abnormal histopathological changes in the liver can be attributed to increased apoptotic widely and inflammatory response.13
An indicator for liver damage was also investigated by the raised level of enzymes GOT and GPT and (Alkailineaes) among treated patients.14 The hepatic tissue changes would reduce a conversion of Hb to meta Hb and that would lead to lack of carrying oxygen and eventually lead to cell death.
The toxic effect of this drug would lead to destruction of the mucous layer of the intestinal villi which in turn cause a reduction in cell proliferation, that can be explained by the inhibition of manufacturing DNA.15
The treated patients would suffer from diarrhoea due to mal absorption of nutrients from the intestinal lumen.16,17
A dose of 20 mg/kg given intraperitoneal into rabbits lead to apoptosis of intestinal mucosa, after 6 hours post injection.18 While a dose of 6 mg/kg would induce severe morphological changes in the small intestinal 3 days after treatment.18 There is a relation between the dose of the drug and the toxic effect on the tissue of the body.
Conclusion
This study showed the toxic effects of doxorubicin on mice livers and intestines. These changes were identified by examining the tissue sections under the light microscope.
References
1.McEvoy GK. American hospital formulary serviee: Drug information 2000; 905.
2.Gaset SA. The effect of antineoplastic drug doxorubicin in breast cancer. Eur J Pharmacol 1993; 12: 7-13.
3.American Society of Hospital Pharamacist Technical. Assistance bulletin on handing cytotoxic and hazardous drug. Am J Hosp Pharam 1990; 17: 1033-49.
4.Arcamone F. Properties of anti tumor anthracycline and neo development s in their application: Cain memorial award lecture. Cancer Res 1985; 45: 5995-9.
5.John MB, Joseph GJ, David AS. Radiochemo therapy for acute leukemia. J Moffitt Cancer Center 2002; 11.
6.Cullinan SA, Moertal GG, Wieand HS, O\\\'Connell MJ, Poon MA, Krook JE, et al. Controlled evaluation of three druge combination regimens versus fluorouracial alone for therapy at advanced gastric cancer. North Central Cancer Treatment Group. J Clinical Oncol 1994; 12: 412-6.
7.Bates NP, Williams MV, Bessell EM, Vaughan Hudson G, Vaudhan Hudson B. Efficacy and toxicity of vinblastive, bleomycin, and methoterxate with involved - field radio - therapy in clinical stage 1A and 11 Hodgkins Disease: a British National Lymphoma Investigations Pilot Study. J Clin Oncol 1994; 12: 288-96.
8.Bjerknes,p. Chemotherapy induced intestinal damage. Nature 2004; 449: 1003.
9.Barker N, Watering Mvd, Clevers H. The intestinal stem cells. Genes Development 2007; 22: 1856-64.
10.Balanchund RJ, Hori K, Blanchard DC, Hall J. Ethanol effects in aggression of rat selected for different levels of aggressioness. Pharmacol Biochem Behav 1987; 27: 641-4.
11.Luna LC. Manual of histological staining Methode of the armed forces. USA: McGraw-Hill, inc. 1968; pp 32.
12.Knight T, Fariss M, Farhood A, Jaeschke H. The role of lipid peroxidation as a mechanism of liver after acetaminophen in mice. Toxic Sci 2003; 76: 229-36.
13.Rash AR, Abdella EM. Modulatory effects of Rosemary leaves Aqueous on doxorubicin-induced histological lesions, Apoptosis and oxidative stress in mice. Iranian J Cancer Prev 2010; 3: 1-22.
14.Helmrath MA, Fong JJ, Dekaney CM, Henning SJ. Rapid expansion of intestinal secretory lineages following a massive small bowel resection in mice. Am J Physiol 2007; 292: G 213-22.
15.Lipkin M. Growth &development of gastrointestinal cells. Annu Rev Physiol 2005; 47: 175-97.
16.Kojima S, Lizuka H, Yanaguchi H, Tanuma S, Kochi M, Ueno Y. Antioxiodative activity of benzylideneascorbate and its effect on advimycin-induced cariotoxiacty. Anti-cancer Res 1994; 14: 1875-80.
17.Tweedale P, Eden OB. Pulmonary function in childhood leukemia survivors. Med Pediatric Oncol 1989; 17: 149-54.
18.Gramm G. Methaemoglobin level in young children after treatment with cisplatine. Int J Epid 1999; 10: 309.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




