Jonathan Adebola Lambo ( Faculty of Health Sciences, University of Lethbridge, Lethbridge, Canada. )
Mahmood Iqbal Memon ( Contech International, Faisal Town, Lahore, Pakistan. )
Zahid Hussain Khahro ( World Health Organization Sindh, Provincial Directorate/Expanded Program on Immunization Office, University Road, Karachi, Pakistan. )
Muhammad Ismail Lashari ( Civil Hospital Dadu, Dadu District, Pakistan. )
November 2011, Volume 61, Issue 11
Original Article
Abstract
Objectives: To estimate the incidence of neonatal tetanus (NT) and to study the factors associated with NT mortality in Dadu district, Pakistan.
Methods: This study is a retrospective analysis of surveillance data from 1993-2003. NT cases were identified from the district surveillance database and hospital records reviews. Cases were ascertained using the NT standard case definition. Clinical records of all neonates (n=416) admitted with the diagnosis of NT from 1993 through 2003 were reviewed for clinical presentation, progression and outcome. Rates, means and frequencies were calculated. Odds ratio was calculated to determine the association between potential risk factors and NT mortality. Logistic regression models were used to compute odds ratios and their associated 95% Confidence Intervals.
Results: Out of a total of 416 NT cases, 408 met the case definition. The overall case fatality rate (CFR) for NT was 30.1% (95% Confidence Interval (CI):25.6-34.6); CFR fell from 42% in 1993 to 29% in 2003 (p=0.377). NT incidence decreased from 0.90/1000 live births (LB) in 1994 to 0.18/1000 LB in 2003. Multivariable analysis showed that age at admission of 8 days or less with {Odds Ratio (OR) 9.41, CI: 2.67-33.14} or without (OR 2.62, CI: 1.52-4.50) low neonatal weight was the strongest predictor of mortality.
Conclusions: The rate of decline of neonatal tetanus incidence and case fatality was consistent with the impact of routine and supplementary immunization activities. In addition to strengthening maternal tetanus toxoid immunization coverage and hygienic delivery practices, health education focusing on increasing awareness of NT could help reduce NT mortality.
Keywords: Neonatal tetanus, Epidemiology, Surveillance, District Dadu, Case fatality, Incidence (JPMA 61: 1099; 2011).
Introduction
Neonatal tetanus (NT), caused by Clostridium tetani, is a highly fatal infection of the neonatal period. Infection occurs when the umbilical cord becomes contaminated as a result of unclean childbirth or cord-care practices. If contamination occurs when the cord is cut or within the next few days, symptoms begin 3-12 days after birth.1
The determinants of the incidence of NT relate to the cultural diversity of hygienic childbirth practices and cord care,2,3 lack of skilled attendance with delivery, parent\\\'s illiteracy,2 lack of antenatal care, including low level of immunization against tetanus,4 seasonality,2 geographical location and climate,5 prevalence of spores of C.tetani6 and rural agricultural settled populations.5 On the other hand, the determinants of mortality in NT are not well understood. Previous studies have shown that a short incubation period and low birth weight7,8 are associated with a high mortality rate and are poor prognostic indicators.
Tetanus toxoid (TT) immunization of pregnant women and of women of child bearing age has been shown to reduce the incidence and mortality of NT6 and is the World Health Organization’s (WHO) recommended strategy for NT elimination, in addition to surveillance for cases of NT in high-risk areas, and promotion of clean delivery services.9 Despite this, immunization coverage is low, particularly in rural areas of developing countries where most births occur at home with the assistance of untrained health personnel and relatives.
NT remains a public health problem in many developing countries including Pakistan, where it is one of the major causes of neonatal and infant mortality. In Pakistan, NT accounts for 18-38% and 17-22% of all neonatal and infant deaths respectively.2,10 NT has a high case fatality ratio (CFR) and community-based surveys in developing countries have reported CFRs approaching 80-90% even with treatment.11
There is a dearth of reliable information on NT incidence and mortality from rural areas where most NT cases and deaths occur.2,10 We perceive that there are important gaps in quantitative analysis of NT surveillance data and reporting in rural Pakistan to inform decision-makers who must plan and implement control measures for neonatal tetanus elimination. The present objectives were to estimate the incidence of neonatal tetanus (NT) and to study the factors associated with NT mortality in Dadu district, Pakistan.
Methods
A retrospective analysis of NT cases in Dadu district, Pakistan was done. We reviewed and analyzed NT cases with onset of symptoms during 1993-2003. The study was restricted to all cases of admissions of infants that met the WHO case definition for NT.12 The cases not meeting the criteria for the case definition were excluded.
The data sources used to identify cases included the NT case reports that were line listed in the district surveillance database and the case records of all admissions, hospital and surveillance registers with the diagnosis of NT in the 7 hospital reporting sites (i.e. government hospitals), 5 private reporting sites (i.e. private clinics of paediatricians) and 5 tertiary referral institutions in border areas of the district for the period 1993-2003. NT case reports that were line listed in the surveillance register during 1993-2003 were checked for double reporting and placed in a separate file (NT line list).
The incidence of NT was based on the total number of cases obtained by summing the number found in the NT line list and the number found by reviewing the records of hospitals and private reporting sites whether or not they had reported a case. The district NT incidence rate per 1000 live births (LB) was calculated by using the formula:
Number of incident cases X - 1000
Total number of live births
The total live births for the study period were computed based on population data from the provincial directorate/Expanded Program on Immunization office Sindh.13 The calculation of the incidence rate takes into consideration that, despite underreporting, other independent data sources (i.e. reviews of hospital case records and registers and private sector reporting) in addition to routine NT surveillance data were included in the analysis. The case fatality ratio was calculated as the proportion of neonates who died of NT in a given year out of those who contracted it.
NT case reports, surveillance registers, case investigation and the hospital case records for all suspected NT cases from 1 January 1993 through 31 December 2003 were reviewed for clinical presentation, progression and outcome. A case of NT was defined according to the WHO standard case definition for NT:(1) normal ability to feed and cry normally for the first 2 days of life;(2) onset of illness between 3 and 28 days of age; and (3) inability to suck, followed by stiffness or spasms.12 All neonates received anti-tetanus serum, antibiotics, anticonvulsants and sedatives and all were fed with expressed breast milk as routine protocol for the care of NT cases.
For each hospitalized or line listed case, we extracted the following attributes: age at onset of symptoms, age on admission, age at death, delay before admission, weight, sex, residence, duration of hospital stay, maternal immunization with at least two doses of tetanus toxoid (TT), antenatal care, place of delivery, delivery assistance and cord care. The outcome variable was mortality from NT. This is defined as death of an infant in the neonatal period i.e. the first 28 days of life.
For descriptive analysis, frequencies, rates, means, and standard deviations were calculated. Chi-square test was done to determine trend analysis. Odds ratios (ORs) and 95% Confidence Intervals (CIs) were calculated to determine the associations between potential risk factors and NT mortality. In addition, logistic regression models were used to determine the simultaneous effect of all potential risk factors on NT mortality and to compute crude ORs and their associated 95% CIs. All exploratory analyses were done at alpha=0.05 for descriptive purposes. Data were analyzed using SPSS version 12.0 (SPSS Inc., Chicago, IL, USA). This work was part of the surveillance programme for the country and ethical review was not required.
Results
The distribution of cases by geographical areas shows several high-risk areas. The surveillance data pointed to the clustering of NT cases in all but one sub district-TB Khan. However, the routine surveillance system missed only 1 NT case which should have been detected in 1997 in the sub district of TB Khan. The persistent clustering of cases which started in 1994 had cleared by 2003 as evidenced by the reduction in the number of cases from 53 to 14. The year wise incidence of NT per 1000LB is shown in Figure.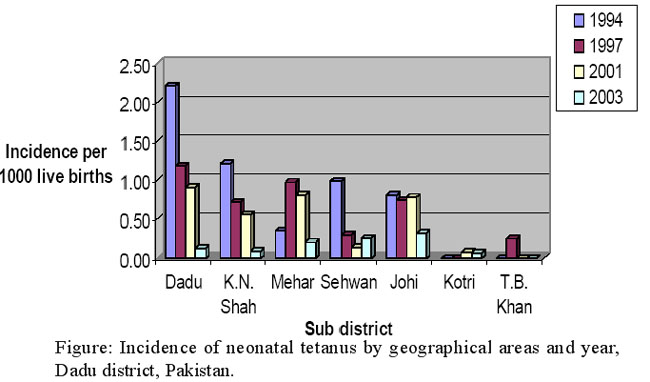
About 98.1% (408/416) of NT cases met the case definition. The final classification of NT cases were: confirmed 408; discarded (i.e. not NT cases) 7 and in 1 case the cause of death could not be ascertained. A discarded case is one which has been investigated and does not meet the criteria for the case definition. The diagnoses for the discarded cases were: neonatal death (not due to NT) 14.3 % (1/7), neonatal sepsis 57.1% (4/7) and septicemia/birth asphyxia 28.6% (2/7).
The district average annual incidence of NT was 0.55/1000LB (n=408 cases; 95% CI =0.50-0.60) during 1993-2003.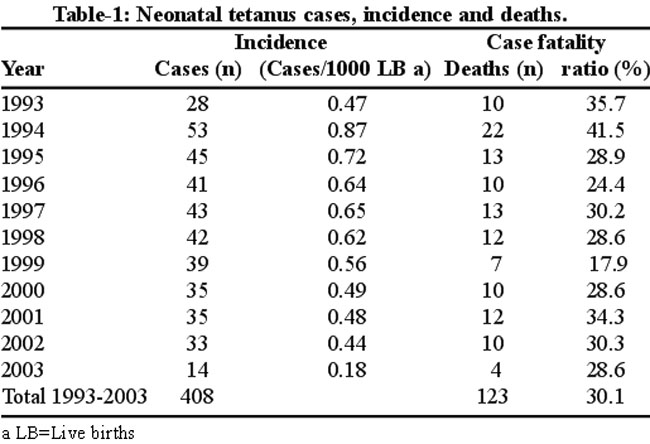
Table-1 shows the trends in the incidence, mortality, and CFR. After an initial increase of NT incidence to 0.87/1000LB in 1994, incidence decreased between 1995 and 2003 from 0.72/1000LB to 0.18/1000LB. The incidence of NT fell from 0.48/1000 in 2001 to 0.18/1000LB in 2003 (a 63% reduction) as a result of the implementation of tetanus toxoid (TT) and supplementary immunization activities (SIAs) in the district during 2001-2003. There was also a 17% reduction in the CFR in the same period (p=0.700) (Table-1).The overall CFR was 30.1% (95% CI= 25.6-34.6); 31.1% (94/302) for males and 27.4% (29/106) for females (p=0.467). The male: female sex ratio of cases was 2.9:1.
Although a higher incidence of NT cases and deaths occurred in the rainy season (July to November peak) than during the dry season but the difference was not significant (P> 0.05).
The results of the univariate analysis of risk factors showed that although there was proportionally increased mortality associated with lack of maternal immunization and antenatal care, delivery at home, delivery assistance by untrained TBAs, poor cord care, there was no significant difference in mortality between survivors and deceased (Data not shown).
The mean incubation period for cases was 6.3±2.7 days. The mean age at death was 10.7±4.6 days. The ratio of male to female at death was 3:1.The interval between the mean age at onset and mean age at death was 4.4 days. The mean delay between onset of symptoms and admission was 1.8 days. A high proportion of neonates 70.2% (172/245) had short incubation period of 6 days or less. By the 8th day of life, 69% (269/390) of the NT cases had already sought admission. Of this 74.3% (200/269) were males and 25.7% (69/269) females.
Out of 408 NT cases, there were a total of 390 admissions (95.6%): 292 males (74.9%) and 98 females (25.1%). 123 (30.1%) neonates died, 153 (37.5%) survived, and 53 (13%) discharged against medical advice. There were 79 (19.4%) cases for which outcome was missing. 355 (87%) of the NT cases resided in rural areas, 47 (11.5%) in urban centres, excluding 6 cases (1.5%) for which residence was unknown. Although the risk of NT is 8 times greater in rural than in urban areas, mortality did not significantly differ between urban and rural areas (Odds ratio (OR) =0.88, p> 0.05).
Analysis of the data by gender showed that the proportions of males and females in the rural (male: 74.3%; female: 25.7%) and urban (male: 77.1%; female: 22.9%) areas were similar so that gender was not a confounding variable. There were also statistically significant differences in mortality between survivors and deceased. The survivors had longer incubation period and greater mean body weight on admission, presented late, and had a longer hospitalization and a longer interval between onset of symptoms and admission.
Stepwise logistic regression analysis showed the simultaneous effects of all these variables on mortality (Table-2).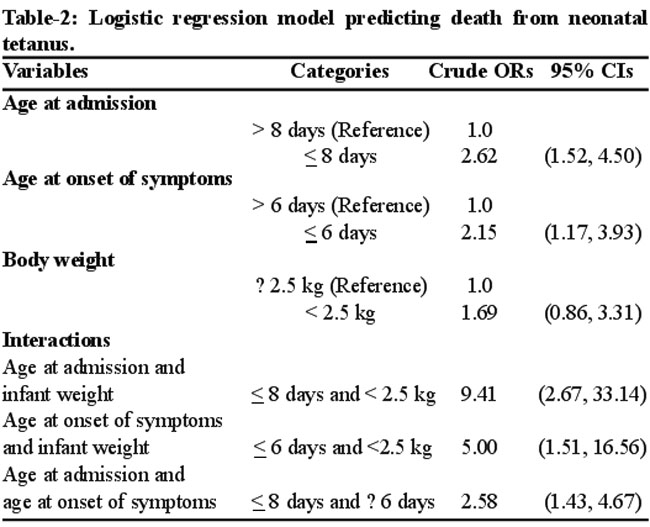
The results showed that the age at admission of 8 days or less was the strongest predictor of mortality (OR=2.62, P=0.001) while a short incubation period of 6 days or less was also a significant predictor (OR=2.15, P=0.013). However, low neonatal weight (< 2.5kg) was not a significant predictor. When the interactions between the age at admission, incubation period, and infant weight were added as covariates, the interaction term between age at admission and infant weight came up as a stronger predictor of mortality (OR=9.41, P<0.001) than that between incubation period and infant weight (OR=5.00, P=0.008), while interaction term between the age at admission and incubation period was also a significant predictor of mortality (OR=2.58, P=0.002). Thus neonates with age at admission of 8 days or less and low weight carried a significantly higher risk of mortality.
Discussion
Reducing deaths from NT has been regarded as one of the simplest and most cost-effective ways to reduce the neonatal mortality rate.14,15 The WHO global elimination target for NT is defined as less than one case of NT case per 1000LB at district level per year.12,13 Although a decline in the overall NT incidence below the elimination target was initially observed through routine immunization of pregnant women with TT, the incidence varies from one geographical area of the district to another.
Implementation of SIAs in two phases in 2001-2003 provided the final push that resulted in a marked reduction in NT incidence. Coverage with the third dose of tetanus toxoid (TT) vaccine was 84% during the first phase and 85% during the second phase. Our results confirmed that targeted TT immunization of women of childbearing age (i.e. the" high risk" approach) is an effective elimination strategy16 and can result in a larger than expected reduction in subsequent NT cases.16,17 NT cases are not randomly distributed but cluster in areas where access to clean childbirth and immunization services is limited or where environment enhance the risk of cord contamination.11,16,17
CFRs reported in our study were lower than expected based on previous studies in Pakistan. CFR can be affected by the mean delay in presentation for treatment in hospital, intensity of supportive care and the interval between mean age at onset and mean age at death.4,8,15,18 The mean delay in presentation of 1.8 days in our study was in contrast to 3.2 days reported in a previous hospital-based series in Pakistan.4 Shorter delay in presentation could imply increased awareness of the symptoms of NT causing parents to seek care earlier. The mean age at death of 10.7 days was consistent with association of 4-14 day death with NT mortality.18 The interval between mean age at onset and mean age at death of 4.4 days was also consistent with a range of 3.1-4.3 days reported in previous series studies.19
Our study showed 96% (390/408) admission rate for the NT cases admitted over the 11 year period and all of them had received a standard treatment protocol. Improved hospitalizations, use of treatment protocol and good nursing care have been shown to reduce mortality4,8,15 all of which may account for the low CFR in our study. Low CFR may be due in part to hospitalization bias for severe cases21 and the infants discharged at the mother\\\'s own request.21 Around, 13% of the NT cases in our study were discharged against medical advice and there were 19.4% for which the outcome was unknown; the former can be regarded as doomed to die.21
We observed a higher incidence of males (74%) than previously reported in both hospital and community-based studies.3,14,20 The male preponderance in our study might reflect gender bias in care seeking and to cultural practices giving preference to the survival of male children in this area. The observation that males were more likely to be admitted than females (75 v.25%) and that by the 8th day of life, admission had been sought for 74% of males compared with 26% of females further lends credence to the preferential survival of male children. Dadu is an area of animal husbandry and agriculture (> 80%) and male children are expected to be involved in these activities, but the survival of female children is equally important. Health education focusing on increasing awareness of NT and optimal utilization of medical facilities for both genders is crucial to the reduction of NT mortality. Nevertheless, TT immunization of pregnant women and women of child bearing remains the cornerstone of effective primary prevention strategy for NT.9,16,17
Our study confirmed the previous observation that an incubation period of 6 days or less and age on admission was significantly associated with mortality. Low neonatal weight (<2.5kg) has been reported in previous studies as a risk factor for mortality in NT7 but our study did not confirm this observation in spite of the significant difference regarding body weight between survivors and deceased. Previous studies have shown that neonates with low birth weight and short incubation of 6 days or less carried a significantly higher risk of mortality.7 However, the findings relating to the age at admission of 8 days or less, or between age at admission of 8 days or less and the incubation period of 6 days or less or between of age at admission of 8 days or less and neonatal weight (<2.5kg) as the strongest predictor of mortality are new. It is therefore likely that the weight variable may have been acting through its relationship to the incubation period or to the age of admission.
The results of our univariate analysis suggest that neonatal survival is enhanced by antenatal care, delivery in health facilities, good cord care, delivery assisted by trained health personnel and maternal immunization with at least 2 doses of TT during pregnancy. None of these variables were significant in our multivariate analysis. The use of soil and straw as delivery surfaces, illiteracy of father, possession of sheep at home,2 delivery at home,4 bundling,3 children of mothers with previous NT2,11 and application of substances, including cow dung, clarified butter (\\\'ghee\\\'),23 antimony (\\\'surma\\\'), mustard oil24 to the umbilical wound of newborns during the first several days of life have been identified as risk factors for NT mortality in rural Pakistan. The use of soil as delivery surface and possession of sheep at home are common practices in this area but the application of ghee to the umbilical stump depends on local customs. However, antimony is more widely applied to fresh umbilical wound than ghee in this area. Transmission of infection may occur while using soil as delivery surface through direct or indirect contamination of the fresh umbilical wound,2 through the hands of birth attendants while dressing the cord,25 or through contaminated hands while applying ghee or antimony to the cord. Use of safer delivery practices in general and clean delivery surfaces should be encouraged as suggested earlier.2,20
Low immunization coverage with TT and low antenatal care suggest deficiencies of the maternal and child health services and Expanded Program on Immunization (EPI) in this area. Ongoing efforts should be made to improve TT delivery to rural populations through outreach services and integration of maternal TT with antenatal care. A better understanding of the cultural barriers to vaccine delivery is urgently needed. Additional strategies such as school-based immunization will be required in order to sustain a high coverage.
Our study, which was hospital-based, has a selective referral bias. However, the risk of biased reporting of NT incidence is small as cases were representative of almost all the geographical areas of the district. Another limitation of our study is the incomplete reporting of NT cases which may lead to underestimation of the reported NT incidence. Nevertheless, our estimate was based on private- sector reporting and active search for suspected NT cases in addition to the surveillance data. Efforts should be made to improve NT reporting at the district level by increasing private- sector participation and community- based reporting of NT.
Acknowledgements
We thank Dr.Margaret L.Russell of the Department of Community Health Sciences, University of Calgary for her helpful comments on drafts of the manuscript. We acknowledge assistance of Dr.Majeed Baledai, Dr. Nisar Solangi and Dr.Shah Baig Chandio for data collection.
References
1.Schofield F. Selective primary health care: strategies for control of disease in developing world.XXII.Tetanus: a preventable problem. Rev Infect Dis 1986; 8: 144-56.
2.Quddus A, Luby S,Rahbar M,Pervaiz Y.Neonatal tetanus: mortality rate and risk factors in Loralai District, Pakistan. Int J Epidemiol 2002; 31: 648-53.
3.Bennett J, Schooley M, Traverso H, Agha SB, Boring J. Bundling: a newly identified risk factor for neonatal tetanus: implications for global control. Int J Epidemiol 1996; 25: 879-84.
4.Daud S, Mohammad T, Ahmad A. Tetanus neonatorum (a preliminary report of assessment of different therapeutic regimens). J Tropic Pediatr 1981; 27: 308-11.
5.Harfouche JK.The epidemiology of neonatal tetanus. Pak Pediatr J 1982; 6: 69-102.
6.Kessel E. Strategies for the control of neonatal tetanus. J Tropic Pediatr 1984; 30: 145-9.
7.Davies-Adetugbo AA, Torimiro SE, Ako-Nai KA. Prognostic factors in neonatal tetanus. Tropic Med Int Health 1998; 3: 9-13.
8.Einterz EM, Bates ME. Caring for neonatal tetanus patients in a rural primary care setting in Nigeria: a review of 237 cases. J Tropic Pediatr 1991; 37: 179-81.
9.Vandelaer J, Birmingham M, Gasse F, Kurian M, Shaw C, Garnier S. Tetanus in developing countries: an update on Maternal and Neonatal Tetanus Elimination Initiative. Vaccine 2003; 21: 3442-5.
10.Fikree FF, Azam SI, Berendes HW.Time to focus child survival programmes on the newborn: assessment of levels and causes of infant mortality in rural Pakistan. Bull World Health Organ 2002; 80: 271-6.
11.Whitman C, Belgharbi M, Gasse F, Torel C, Mattei V, Zoffmann H. Progress towards the global elimination of neonatal tetanus. World Health Stat Q 1992; 45: 248-56.
12.World Health Organization. Field Manual for neonatal tetanus elimination. WHO/V & B/99.14, Geneva, 1999. (Online) (Cited ………. ) Available from URL: http://whqlibdoc.who.int/hq/1999/WHO_V&B_99.14.pdf.
13.Statistics Division, Government of Pakistan. Sindh Province Population Data. From Population and Housing Census of Pakistan, 1998. Bureau of Statistics, Planning and Development Department. July 1998.
14.Dikici B, Uzun H, Yilmaz-Keskin, E, Tas T, Gunes A, Kocamaz H, et al. Neonatal tetanus in Turkey; what has changed in the last decade? BMC Infect Dis 2008; 8: 112.
15.Ertem M, Cakmak A, Saka G, Ceylan A. Neonatal tetanus in the south-eastern region of Turkey: changes in prognostic aspects by better health care. J Tropic Pediatr 2004; 50: 297-300.
16.Expanded Programme on Immunization. The "high-risk" approach: the WHO-recommended strategy to accelerate elimination of neonatal tetanus. Wkly Epidemiol Rec 1996; 71: 33-6.
17.Aylward RB, Mansour E, Oon EA, et al. The Role of Surveillance in a \\\'High Risk\\\' Approach to the Elimination of Neonatal Tetanus in Egypt. Int J Epidemiol 1996; 25: 1286-91.
18.Eregie, CO, Ofovwe, G. Cluster survey on neonatal tetanus mortality in Nigeria: observation on some clinical aspects. J Tropic Pediatr 1993; 39: 372-3.
19.Expanded Programme on Immunization. Neonatal tetanus survey, Indonesia.Wkly Epidemiol Rec 1982; 57: 395.
20.Chai F,Prevots DR,Wang X,Birmingham M,Zhang R.Neonatal tetanus incidence in China,1996-2001,and risk factors for neonatal tetanus, Guangxi Province, China. Int J Epidemiol 2004; 33: 551-7.
21.Stanfield JP, Galazka A. Neonatal tetanus in the world today. Bull World Health Organ 1984; 62: 647-69.
22.Traverso HP, Kamil S, Rahim H, Samadi AR, Boring JR, Bennett JV. A reassessment of risk factors for neonatal tetanus. Bull WHO Health Organ 1991; 69: 573-9.
23.Bennett J, Azhar N, Rahim F, Kamil S, Traverso H, Killgore G, et al. Further observations on ghee as a risk factor for neonatal tetanus. Int J Epidemiol 1995; 24: 643-7.
24.Raza SA, Akhtar S, Avan BI,Hamza H, Rahbar MH. A matched case-control study of risk factors for neonatal tetanus in Karachi, Pakistan. J Postgrad Med 2004; 50: 247-51.
25.Leroy O, Garenne M.Risk factors for neonatal tetanus in Senegal. Int J Epidemiol 1991; 20: 521-6.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




