Mohammad Mehdi Hossenian ( Department of Neurology, Faculty of Medicine, Islamic Azad University of Tabriz, Tabriz City, Iran. )
Ali Khodadadi ( Faculty of Veterinary, Islamic Azad University of Tabriz, Tabriz City, Iran. )
Amir Mansour Vatankhah ( Drug Appleid Research Center, Islamic Azad University of Tabriz, Tabriz City, Iran. )
Babak Hajipour ( Young Researchers club, Tabriz Branch, Islamic Azad University, Tabriz, Iran. )
Hormoz Ayromlou ( Neurology Research Center, Tabriz University of Medical Science, Iran. )
November 2011, Volume 61, Issue 11
Original Article
Abstract
Objective: To study the potential effect of Methotrexate (MTX) on lipid peroxidation and activity of glutathione peroxidase (GPx) and superoxide dismutase (SOD) two important endogenous anti-oxidant enzymes.
Methods: Twenty healthy male Newzland rabbits weighting 1500-1800 g were divided into two groups. One group was considered as the MTX groups and the other was considered as control group. Rabbits had free access to food and tap water. Rabbits in MTX group received a single dose of MTX, 20 mg/kg intraperitoneally, a similar volume of saline was administered to control group. After 6 days rabbits were uteinized and spinal tissue excised for biochemical studies.
Results: GPX and SOD activities in spinal tissue in rabbits of MTX group significantly reduced after MTX administration compared to control group(P=0.002 vs 0. 18). An increase in the tissue MDA level was seen in the MTX group, suggesting increased lipid peroxidation. Levels of MDA were significantly higher in MTX group compared to control group(P=0.002).
Conclusion: Our data suggests that MTX treatment induces oxidative tissue damage on the spinal tissue, as assessed by increased lipid peroxidation and decreased GPX and SOD levels, so decreasing oxidative stress by anti-oxidant agents may play a key role in attenuating spinal cord injury.
Keywords: Methotrexate, Lipid peroxidation, Glutathione peroxidase, Superoxide dismutase, Oxidative stress (JPMA 61: 1096; 2011).
Introduction
Methotrexate is a cytotoxic chemotherapeutic agent, widely used not only for malignancies but also for various inflammatory diseases such as psoriasis, dermatomyositis, sarcoidosis, and rheumatoid arthritis. MTX-induced toxicity appears to be a consequence of the interaction of many factors, including the dosing schedule and length of treatment, patient risk factors, type of disease, and the presence of genetic and molecular apoptotic factors.1,2
The therapeutic use of MTX has been limited by its toxicity for the proliferating cells, especially the rapidly dividing cells of intestinal crypts. MTX is a dihydrofolate reductase (DHFR) inhibitor that blocks DNA synthesis by depleting the intracellular-reduced folate pools required for the biosynthesis of purines and thymidine, and leads to cell cycle arrest and apoptosis in many cell types.3,4
Depending on the dose, route of administration and concomitant use of other potential neurotoxic agents, MTX may affect the brain, the spinal cord, or the nerve roots. Myelitis or flaccid paralysis are the most common complications of MTX with the intrathecal administration, whereas reversible motor deficits reminiscent of transient ischaemic attacks occasionally follow high-dose intravenous administration.5
Under normal conditions, NADPH is used by glutathione reductase to maintain the reduced state of cellular glutathione, which is known as an important cytosolic antioxidant protecting against reactive oxygen species (ROS). It has been demonstrated that the cytosolic NAD (P)- dependent dehydrogenases6 and NADP malic enzyme are inhibited by MTX, indicating that the drug could decrease the availability of NADPH in cells by inhibiting pentose cycle enzymes.7 Due to the interference with the pentose phosphate shunt, MTX may also depress nucleic acid metabolism. Thus, the significant reduction in glutathione (GSH) levels promoted by MTX leads to a reduction of effectiveness of the antioxidant enzyme defense system, sensitizing the cells toROS.6 Considering the relationship between GSH and the deleterious effects of MTX, interest has focused on compounds that are capable of stimulating GSH synthesis or those that act as antioxidants.8
In addition, it is shown that the detrimental effect of MTX is partly due to its direct toxic effect by increasing ROS production.9 The mechanisms of MTX-induced toxicity have not been exactly determined yet. Recently Miketova et al10 demonstrated that MTX chemotherapy caused oxidative stress in membrane phospholipids of CNS. Oxidative stress is a known cause at toxicity for various drugs such as cisplatin, methotrexate, lithium, diazepam and isoniazid.11,12
Previous studies have reported oxidative effect of MTX in cerebellum of rats but there is not any report studying the probable effect of MTX on the levels of anti-oxidant enzymes and lipid peroxidation in spinal tissue. The aim of this research was to study the potential effect of MTX on lipid peroxidation and activity of glutathione peroxidase (GPx) and superoxide dismutase (SOD) two important endogenous anti-oxidant enzymes.
Material and Methods
All experimental protocols were approved by the Ethics Committee of Tabriz University of medical sciences. Twenty healthy male Newzland rabbits weighting 1500-1800 g were divided into two groups. One group was considered as the MTX groups and the other was considered as control group. Rabbits had free access to food and tap water. Rabbits in MTX group received a single dose of MTX, 20 mg/kg intraperitoneally, a similar volume of saline was administered to control group. After 6 days rabbits were uteinized and spinal tissue excised for biochemical studies.
Measurement of GPx activity: The glutathione peroxidase (GPx) activity was determined as described by Paglia and Valentine.13 After mixing 40 Ml of the sample with 10 Ml of t-butyl hydroperoxide and a solution of distilled water containing 10 mg glutathione (GSH), GSH reductase, NADPH, buffer (0.25 M KH2PO4 and 0.025 M Na2EDTA), and 940 Ml Kbuffer, the GPx activity was measured at 10-second intervals for 60 seconds by recording the rate of light absorption.
Measurement of SOD activity: SOD activity was determined as described by Sun et al.14 This method depends on the inhibition of nitroblue tetrazolium (NBT) reduction by xanthine-xanthine oxidase used as a superoxide generator. One SOD activity was expressed as the amount of enzyme that causes 50% inhibition of the rate of NBT reduction. SOD activity was designated as unit for mg/protein of spinal tissue.
Determination of Lipid Peroxidation: The lipid peroxidation products in spine was determined by measuring malondialdehyde (MDA). The method of Yagi et al.15 was used. Briefly, 20 µl of sample was placed in a glass centrifuge tube. 4.0 ml of 1/12 N H2SO4 was added and mixed gently. An amount of 0.5 ml of 10% phosphotungstic acid was added and mixed. After being allowed to stand at room temperature for 5 min, the mixture was centrifuged at 1600 g for 10 min. The supernatant was discarded and the sediment mixed with 2.0 ml of 1/12 N H2SO4 followed by 0.3 ml of 10% phosphotungstic acid. The mixture was centrifuged at 1600 g for 10 min. The sediment was suspended in 1.0 ml of distilled water, and 1.0 ml of 0.67% (w/v) TBA reagent was added. The reaction mixture was heated at 95 °C for 60 min. After cooling with tap water, 5.0 ml of n-butanol was added and the mixture was shaken vigorously. After centrifugation at 1600 g for 15 min, the n-butanol layer was taken for fluorometric measurement at 553 nm with excitation at 515 nm.
Statistical analysis: Data were expressed as means ± SD. Differences among groups were tested for statistical significance using the Mann-Whitney U test. A P value of less than 0.05 denotes the presence of a statistically significant difference.
Results
Data on the GPX and SOD activities of the spinal tissue of rabbits are shown in table. GPX and SOD activities in spinal tissue in rabbits of MTX group are significantly reduced after MTX administration compared to control group (P=0.002 vs P=0.018) (Table).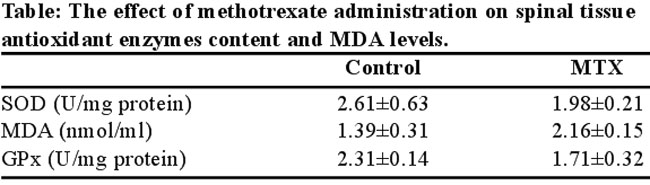
The MDA levels in the spinal tissue are shown in table. An increase in the tissue MDA level was seen in the MTX group, suggesting increased lipid peroxidation. Levels of MDA were significantly higher in MTX group compared to control group (P=0.002) (Table).
Note. The rabbits received methotrexate 20mg/kg and after 6 days spinal tissue was studied for measuring antioxidant enzymes content and MDA levels. The values are shown as a mean ±SD for rabbits in each group. SOD: Superoxide dismutase, GPx: Glutathione peroxidase , MDA: Malondialdehyde.
Discussion
Since the antitumour drugs are being increasingly utilized as adjuvant therapy for patients at high risk for recurrent disease , a major concern is the complications associated with chemotherapy. MTX, the widely used drug in antimetabolite cancer therapy or in various forms of arthritis, is known to have toxic effects due to oxidative reactions that take place during its metabolism in the liver. Due to its lack of specificity toward malignant cells, MTX may cause toxicity in different tissues.16,17
The result of the present study indicated that MTX administration in a single dose of 20 mg/kg body weight caused a significant decrease in the activities of GPX and SOD and increased MDA levels in spinal cord tissue of rabbits. A lot of studies have been conducted to clarify the underlying mechanisms of neurotoxicity caused by MTX.18-20 The mechanism of MTX-induced neurotoxicity has been elucidated in great detail.21 Recent advances in medicine have demonstrated that oxygen radicals and hydrogen peroxides are associated with the undesired adverse effects of several anti-tumour drugs.22 ROS generation, through the production of hydroxyl radicals, can lead to cellular damage. Possible mechanism of ROS generation is due to high activities of purine-catabolizing enzymes such as ADA and XO. The mitochondrial membrane dysfunction leads to impaired ATP metabolism with increased production of purine degradation products such as adenosine, inosine, hypoxanthine and xanthine which are substrates for ADA and XO.23 Free radicals are expected to play a role in MTX-induced toxicity. Lipid peroxidation, mediated by oxygen free radicals, is believed to be an important cause of destruction and damage to cell membranes and has been suggested to be a contributing factor to the development of MTX-mediated tissue damage, as confirmed by the elevated MDA levels in the present study.
To reduce the detrimental effects of ROS, besides diminishing its production, organisms have developed their own antioxidant mechanisms including low-molecular-weight antioxidant molecules, for example glutathione, melatonin and various antioxidant enzymes, such as SOD and GPx and glutathione reductase. Superoxide dismutase (SOD), an oxygen radical scavenger, which converts the superoxide anion radical present in the upper stream of reactive oxygen metabolism cascade, will afford protection from cell damage. There were reports describing the efficacy of SOD on I/R injury of the liver.24 SOD catalyses the dismutation of the superoxide anion (O2-) into H2O2; GSH-Px is a selenoprotein, which reduces lipidic or nonlipidic hydroperoxides as well as H2O2 while oxidizing GSH.25 In our study, we found that MTX administration impaired these enzymes activities, as indicated by the markedly lower activities compared with control group group. Uzar et al26 indicated that MTX administration in a single dose of 20 mg/kg body weight caused a significant increase in the activities of ADA and in the levels of NO in spinal cord tissue of wistar rats.Uzar et al12 study showed that MTX causes lipid peroxidation with increased malondialdehyde in rat cerebellum tissue. Likewise, the result of this experimental study may also suggest that spinal cord toxicity is related to MTX-induced oxidative stress.
Conclusion
In summary, our data suggests that MTX treatment induces oxidative tissue damage on the spinal tissue, as assessed by increased lipid peroxidation and decreased GPX and SOD levels, so decreasing oxidative stress by anti-oxidant agents may play a key role in attenuating spinal cord injury.
References
1.Cetinkaya A, Bulbuloglu E, Kurutas EB, Kantarceken B.N-acetylcysteine ameliorates methotrexate-induced oxidative liver damage in rats. Med Sci Monit 2006; 12: BR 274-8.
2.Neuman MG, Cameron RG, Haber JA, Ratz GG, Malkiewicz IM, Shear NH. Inducers of cytochrome P450 2E1 enhance methotrexate-induced hepatotoxicity. Clin Biochem 1999; 32: 519-36.
3.Li T, Ito K, Sumi S, Fuwa T, Horie T. Protective effect of aged garlic extract (AGE) on the apoptosis of intestinal epithelial cells caused by methotrexate. Cancer Chemother Pharmacol 2009; 63: 873-80.
4.da Silva CP, de Oliveira CR, da Conceicao M, de Lima P. Apoptosis as a mechanism of cell death induced by diVerent chemotherapeutic drugs in human leukemic T-lymphocytes. Biochem Pharmacol 1996; 51: 1331-40.
5.Brock S, Jennings HR. Fatal acute encephalomyelitis after a single dose of intrathecal methotrexate. Pharmacotherapy 2004; 24: 673-6.
6.Babiak RM, Campello AP, Carnieri EG, Olivera MB. Methotrexate: pentose cycle and oxidative stress. Cell Biochem Funct 1998; 16: 283-93.
7.Caetano NN, Campello AP, Carnieri EGS, Kluppel ML, Oliveira MB. Effects of methotrexate (MTX) on NAD(P)+ dehydrogenases of HeLa cells: malic enzymes, 2-oxoluterate and isocitrate dehydrogenases. Cell Biochem Funct 1997; 15: 259-64.
8.Sener G, Ek?io?lu-Demiralp E, Cetiner M, Ercan F, Sirvanci S, Gedik N, et al. L-Carnitine ameliorates methotrexate-induced oxidative organ injury and inhibits leukocyte death. Cell Biol Toxicol 2006; 22: 47-60.
9.Neuman MG, Cameron RG, Haber JA, Katz GG, Malkewcz IM, Shear NH. Inducers of cytochrome P450 2E1 enhance methotrexate-induced hepatotoxicity. Clin Biochem 1999; 32: 519-36.
10.Miketova P, Kaemingk K, Hockenberry M, Pasvogel A, Hutter J, Krull K, et al. Oxidative changes in cerebral spinal fluid phosphatidylcholine during treatment for acute lymphoblastic leukemia. Biol Res Nurs 2005; 6: 187-95.
11.Husain K, Morris C, Whitworth C, Trammell GL, Rybak LP, Somani SM: Protection by ebselen against cisplatin-induced nephrotoxicity: antioxidant system. Mol Cell Biochem 1998; 178: 127-33.
12.Uzar E, Koyuncuoglu HR, Uz E, Yilmaz HR, Kutluhan S, Kilbas S, et al. The activities of antioxidant enzymes and the level of malondialdehyde in cerebellum of rats subjected to methotrexate: Protective effect of caffeic acid phenethyl ester. Mol Cell Biochem 2006; 291: 63-8.
13.Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Met 1967; 70: 158-69.
14.Sun Y, Oberley LW, Li Y. A simple method for clinical assay of superoxide dismutase. Clin Chem 1988; 34: 497-500.
15.Yagi K. A simple fluorometric assay for lipoperoxide in blood plasma. Biochem Med 1976; 15: 212-6.
16.Goto E, Tomojiri S, Okamoto I, Tanaka K. Methotrexate poisoning with acute hepatorenal dysfunction. J Toxicol Clin Toxicol 2001; 39: 101-4.
17.Cetiner M, Sener G, Sehirli AO, Ek?io?lu-Demiralp E, Ercan F, Sirvanci S, et al. Taurine protects against methotrexate-induced toxicity and inhibits leukocyte death. Toxicol Appl Pharmacol 2005; 209: 39-50.
18.Kishi S, Griener J, Cheng SC, Das SH, Cook EH, Pei D, et al. Homocysteine, pharmacogenetics, and neurotoxicity in children with leukemia. J Clin Oncol 2003; 21: 3084-91.
19.Linnebank M, Pels H, Kleczar N, Farmand S, Fliessbach K, Urbach H, et al. MTX-induced white matter changes are associated with polymorphisms of methionine metabolism. Neurology 2005; 64: 912-3.
20.Uz E, Sogut S, Sahin S, Var A, Ozyurt H, Gulec M, et al. The protective role of caffeic acid phenethyl ester (CAPE) on testicular tissue after testicular torsion and detorsion. World J Urol 2002; 20: 264-70.
21.Vezmar S, Becker A, Bode U, Jaehde U. Biochemical and clinical aspects of methotrexate neurotoxicity. Chemotherapy 2003; 49: 92-104.
22.Miyazono Y, Gao F, Horie T. Oxidative stress contributes to methotrexate-induced small intestinal toxicity in rats. Scand J Gastroenterol 2004; 39: 1119-27.
23.Fadillioglu E, Erdogan H, Iraz M, Yagmurca M. Effects of caffeic acid phenethyl ester against doxorubicin-induced neuronal oxidant injury. Neurosci Res Commun 2003; 33: 132-8.
24.Minor T, Isselhard W,Yamamoto Y, Obara M, Saad S. The effects of allopurinol and SOD on lipid peroxidation and energy metabolism in the liver after ischemia in an aerobic/anaerobic persufflation. Surg Today 1993; 23: 728-32.
25.Michiels C, Raes M, Toussaint O, Remacle J. Importance of Se-glutathione peroxidase, catalase, and Cu/Zn-SOD for cell survival against oxidative stress. Free Radic Biol Med 1994; 17: 235-48.
26.Uzar E, Sahin O, Koyuncuoglu HR, Uz E, Bas O, Kilbas S, et al. The activity of adenosine deaminase and the level of nitric oxide in spinal cord of methotrexate administered rats: Protective effect of caffeic acid phenethyl ester. Toxicology 2006; 218: 125-33.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




