Madiha Syed ( Department of Paediatrics and Child Health, Aga Khan University, Stadium Road, Karachi, Pakistan. )
Fariha Batool Khawaja ( Medical Graduates, Class of 2009, Aga Khan University, Stadium Road, Karachi, Pakistan. )
Taimur Saleem ( Medical Graduates, Class of 2009, Aga Khan University, Stadium Road, Karachi, Pakistan. )
Umair Khalid ( Medical Graduates, Class of 2009, Aga Khan University, Stadium Road, Karachi, Pakistan. )
Abdul Rashid ( Department of Paediatrics and Child Health, Aga Khan University, Stadium Road, Karachi, Pakistan. )
Khadija Nuzhat Humayun ( Department of Paediatrics and Child Health, Aga Khan University, Stadium Road, Karachi, Pakistan. )
November 2011, Volume 61, Issue 11
Original Article
Abstract
Objectives: This study aims to present an experience in the management and clinical features of 88 children presenting with diabetic ketoacidosis (DKA) from Pakistan.
Methods: A retrospective medical chart review of all patients, < 15 years of age, who presented to the Aga Khan University Hospital, Karachi, Pakistan in the last ten years with a diagnosis of diabetic ketoacidosis was carried out. Severity of DKA was defined as mild (venous pH<7.30 or bicarbonate=15mEq/l), moderate (venous pH<7.2 or bicarbonate= 10 mEq/l) and severe (venous pH <7 or bicarbonate < 5 mEq/l). These classes correspond to 1st, 2nd and 3rd degrees of diabetic ketoacidosis severity respectively. Cases in which diabetic ketoacidosis had occurred at onset of diabetic diagnosis were not included in the study.
Results: Mean age was 7.5 ± 3.6 years; 58 (66%) patients were male. Twenty six patients had mild diabetic ketoacidosis, 44 had moderate while 18 had severe diabetic ketoacidosis at the time of presentation. Severity of diabetic ketoacidosis was significantly associated with the presence of infection, history of omission of insulin, poor compliance, presence of shock at time of presentation, length of stay in the hospital, final outcome (p < 0.01 for each of these associations) and Glasgow Coma Scale score (p=0.02). Mortality in this series was 3.4%.
Conclusion: Poor compliance was associated with the severity of diabetic ketoacidosis. Paediatric endocrinologists should ensure that patients and their parents understand the importance of the need for regular insulin injections and regular monitoring of blood glucose.
Keywords: Diabetic ketoacidosis, Insulin, Compliance, Karachi, Pakistan (JPMA 61: 1082; 2011).
Introduction
Diabetic Ketoacidosis (DKA) is a life-threatening paediatric metabolic emergency that is characterized by hyperglycaemia (>200 mg/dl or 11.1 mmol/l), metabolic acidosis (venous pH <7.3 and/or serum bicarbonate < 15 mEq/l) with associated glycosuria, ketonuria, and ketonaemia.1 The mortality rate for DKA in children in the developed countries has declined to 0.15% - 0.31%.2,3 However, in places with less developed medical facilities, the risk of death from DKA is greater, and children may die before receiving appropriate treatment. Recent studies have documented a 21% to 24% mortality rate in patients who develop cerebral oedema with DKA, hence accounting for most of the DKA deaths and a high rate of permanent neurologic morbidity.1,4-6 Other causes of morbidity and mortality in DKA include hypoglycaemia, infections, pulmonary oedema, central nervous system haemorrhage or thrombosis, other large vessel thromboses, cardiac arrhythmias caused by electrolyte disturbances, pancreatitis, renal failure and intestinal necrosis.1,4-6
The cornerstones of the treatment of DKA are rehydration, insulin therapy and rectification of the electrolyte disorders with particular attention to potassium, sodium and phosphate. Timely recognition of DKA and appropriate subsequent management to counter the metabolic derangements are important to minimize complications. In young children, it is often difficult to characterize the classical signs and symptoms of diabetes mellitus, such as polyuria, polydipsia and weight loss, and therefore, as many as 20-40% of the patients with type 1 diabetes present initially as DKA.1,3,7
Despite the increasing incidence of diabetes mellitus in Pakistan, the complications from this condition, including DKA, remain a neglected health issue.8 Limited literature is available regarding DKA in the Pakistani population.8-15 In particular, there are few studies available that have looked at the clinical profile and outcomes of DKA in the paediatric age group in Pakistan. In order to improve existing management protocols for DKA in children in Pakistan, it is first important for physicians to appreciate the clinical profile of DKA in children and to be cognizant of the current trends in outcomes resulting from this condition. This report describes our ten-year experience of DKA management and outcomes in a paediatric population at a tertiary care hospital in Karachi, Pakistan.
Methods
This study was based on a retrospective review of the medical charts of all patients < 15 years of age who presented to the Aga Khan University Hospital (AKUH) in the last ten years with a diagnosis of DKA. This diagnosis of DKA was based on finding the characteristic picture of ketonuria, hyperglycaemia (> 200 mg/dl or 11.1 mmol/l) and metabolic acidosis (venous pH <7.3 and/or serum bicarbonate < 15 mEq/l) in established cases of diabetes mellitus. Severity of DKA was defined as mild (venous pH<7.30 or bicarbonate=15mEq/l), moderate (venous pH<7.2 or bicarbonate= 10 mEq/l) and severe (venous pH <7 or bicarbonate < 5 mEq/l).1 These correspond to 1st, 2nd and 3rd degrees or grades of DKA severity respectively. These patients were then managed as in-patients at AKUH after preliminary evaluation and management in the emergency department of this hospital. The detailed evaluation of all patients included the measurement of atleast the following parameters: random blood glucose, urine ketones, serum electrolytes, arterial blood gases and complete blood count.
Once admitted, these patients were managed in the paediatric intensive care unit (PICU) or the special care unit (SCU) depending upon the clinical condition of the patient. Hourly monitoring of vital signs and blood glucose levels was done in these patients. All patients initially received intravenous rehydration and insulin infusion. Once their condition stabilized and their DKA resolved; they were shifted to subcutaneous insulin with a safe degree of overlap between the two forms of insulin. The resolution of DKA in the patients was based on the consideration of multiple factors such as the resolution of acidosis noted on arterial blood gas analysis, normalization of blood sugar levels and stabilization of the patient\\\'s clinical condition.8 Patients who didn\\\'t meet the laboratory parameters of DKA and had an alternative diagnosis that explained their signs and symptoms equally as well or better were excluded.
The Body Mass Index (BMI) was calculated by dividing the weight (in kilograms) by the square of the height (in meters). Socio-economic status of the household of the children was calculated using a set of variables that included area of residence and cumulative family income. It was divided into three categories including high, middle and low socioeconomic status. The diagnosis of cerebral oedema was suspected in the following two scenarios a) declining neurological status after initial improvement; b) persistently poor neurological status without any obvious cause. The early signs that were suspicious for this condition included headache, vomiting, lethargy and decreased arousal, relative bradycardia and hypertension while the late signs included seizures, incontinence, pupillary changes, papilledema, upgoing plantars and respiratory arrest.16 The diagnosis of cerebral oedema was verified on the basis of computed tomography (CT) scan in all the cases. Immediate management strategies employed to reduce cerebral oedema included elevation of head end of the bed, immediate reduction in rate of fluid therapy and use of intravenous mannitol. Shock in the patients was indicated by the presence of signs of systemic hypoperfusion accompanied by at least one of these: tachycardia and/or hypotension. Signs of systemic hypoperfusion were assessed by measurement of pulse characteristics, cutaneous temperature, capillary refill time, neurological status and urine output.17
HbA1c, at our institution, has been measured via high performance liquid chromatography method since the past 8 years. However, for earlier cases it was calculated by the IMS machine via electroimmunoassay. Presence of infection was indicated by a positive radiological imaging study or culture. This was supported by an elevated white blood cell count and clinical examination by the physician. The measure of compliance with regards to insulin was based on the history given by the attendants of the patients. Poor compliance was defined as missing insulin injections on multiple days, especially immediately before or during the period of illness.
Data was entered and analyzed using SPSS version 16.0. In the descriptive analysis, the mean, standard deviations and percentages were computed. Associations were evaluated using chi-square test or Fisher\\\'s exact test and univariate logistic regression where applicable. Odd\\\'s ratios were calculated for associations with a 95% confidence interval. A p-value of < 0.05 was considered statistically significant, unless otherwise specified. All ethical considerations were ensured throughout the study.
Results
A total of 88 patient charts were reviewed retrospectively in accordance with the inclusion criteria of our study. The mean age of patients was 7.5 ± 3.6 years. Fifty eight of the patients in our study were boys (66%). Twenty six patients belonged to the high socioeconomic status (SES), 44 to the middle SES and 18 were from low SES. The mean BMI was 14.4 ± 2.9 kg/m2. The mean duration of symptoms was 3.2 ± 2.7 days. Twenty six patients had 1st degree DKA, 44 had 2nd degree DKA while 18 had 3rd degree DKA at the time of presentation to the emergency department. This grading was also reflected by the blood pH of the patients; 26 had pH between 7.2-7.3, 44 had pH between 7-7.19 while the remaining had pH less than 7.
Table-1 shows the distribution of the socio-demographic characteristics along with some other baseline variables at presentation of the study population in relation to the severity of the DKA. Presence of infection, history of omission of insulin and poor compliance were factors that were seen to be significantly associated with the severity of DKA at presentation (p < 0.01).
Shock was present in a total of 17 (19.3%) patients at the time of presentation. Out of these 17 patients, 13 (76.5%) had grade 3 DKA. Eight patients (9%) had a Glasgow Coma Scale Score of 8 or less. Blood cultures were positive in only 4 patients (4.5%). The mean values of various important serum parameters in patients included: random blood sugar: 480 mg/dl ± 128 mg/dl (26.6 mmol/l ± 7.1 mmol/l), 2) sodium: 133 ± 9.4 mEq/l, 3) potassium: 4.5 ± 0.8 mEq/l, 4) bicarbonate: 15 ± 6 mEq/l, 5) HBA1c: 9% ± 2.5%.
Seven patients (8%) required admission to the paediatric intensive care unit (PICU). The mean duration of stay in the hospital was 6.4 ± 2.9 days. Presence of shock at the time of presentation, Glasgow coma scale (GCS) score, length of the stay in the hospital and final outcome in terms of resolution of condition or death were significantly associated with the degree of severity of DKA (p values of <0.01, 0.02, <0.01 and <0.01 respectively). The associations between biochemical and clinical parameters with the degree of severity of DKA are presented in Table-2.
Bicarbonate bolus, although no longer recommended (1), was used in 30 patients (36.1%) who were from the earlier part of our review period. Similarly insulin bolus is also no longer used (1) and 38 patients (43.2%) who received it were from the earlier part of our study population. The mean duration of the insulin infusion was 29.2 ± 22.2 hours. The mean duration of acidosis was 19.1 ± 19.5 hours. A total of 6 patients (6.8%) developed cerebral oedema in our series. There were 3 deaths; therefore the mortality rate was 3.4%. All the 3 patients who died had 3rd degree DKA as well as cerebral oedema. One of these patients had concomitant sepsis; the 2nd one had severe acute respiratory distress syndrome while the 3rd patient had cerebral oedema without any other pathology. Presence of cerebral oedema, need for mechanical ventilation, low socioeconomic status, low serum sodium level, low serum potassium level, low arterial blood pH, presence of infection and stay in the paediatric intensive care unit were factors that impacted the outcome of the patient. The association between different variables and outcome is presented in table 3.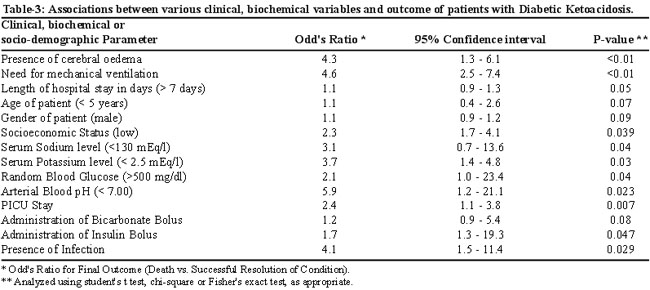
Review at the time of the last follow-up, of these patients in the out-patient clinic after discharge showed that 74 (87.1%) patients were taking insulin therapy at home. Fifty nine (69.4%) patients were compliant in the receipt of insulin therapy. Reasons of non-compliance were unfortunately not documented.
Discussion
Most of the studies of DKA in paediatric population have enrolled subjects up to the age of 20. However, since 15 years is the cut-off between paediatric and adult admissions at our hospital, we reviewed files of all patients younger than 15 years of age. Our data shows no association between age and severity of DKA. These findings are also consistent with the results of a study in which although age was found to be significantly associated with the incidence of DKA, but not with its severity.18 Our data also showed no significant correlation between severity of DKA and other demographic factors like gender, socio economic status, body mass index and family history of diabetes and this is supported by other reports in literature.19
In children with DKA, initial consciousness level is closely related to acidosis.1-3 In our study, the severity of acidosis as a reflection of the severity of DKA, appeared to have a clinically significant association with the GCS at the time of presentation. Acidosis has been reported as an independent predictor of the level of consciousness in DKA, as it affects the GCS even without the development of cerebral oedema.20
We found that presence of infection, poor compliance and omission of insulin had a positive correlation with the degree of severity of DKA in our cohort. Specific defects in innate and adaptive immune function have been identified in diabetic patients making them prone to infection and in turn to the development of DKA.21 In a study done in Egypt, infection was found to precede the diagnosis of DKA in 21.9% cases.22 This association is clearer in the context of extreme metabolic disturbances as shown in our study, where infections have been found to significantly affect the severity of DKA. A study from Manchester23 based on a 6-year retrospective review of 135 diabetic children found that abnormal insulin treatment behaviour was an important factor in the development of DKA episodes in established diabetic children. This is in agreement with a study conducted in Dundee where adherence to insulin treatment protocols was found to be inversely related to hospital admissions for DKA suggesting that poor adherence to insulin treatment was a major factor in DKA hospitalizations.3 This notion is confirmed by our results where history of insulin omission had a direct correlation with DKA severity (p-value of < 0.01).
Cerebral oedema is the most threatening complication of DKA in children, occurring in 0.3% - 1% of DKA episodes.1,5 It has a high mortality rate of 21%-24% in different studies, where as 21%-26% of the survivors suffer from neurologic sequelae.1,4-6 This is further reinforced by our study in which the development of cerebral oedema significantly affected the outcome of our patients. Hence, it is very important to prevent and recognize the early warning signs of cerebral oedema, such as decreased arousal, lethargy after initial improvement, headache, vomiting, relative bradycardia and relative hypertension.16,24 A review highlights an increased risk of the development of cerebral oedema with mechanical ventilation in patients with severe diabetic ketoacidosis. This could explain why, in our study, patients who were intubated had a significantly worse clinical outcome.25
DKA, represents a decompensated phase of diabetes mellitus, which may require PICU admission, especially in the presence of cardiovascular instability, inability to protect the airway, altered state of consciousness, the presence of acute abdominal signs or symptoms suggestive of acute gastric dilatation26 Our study reports a significant association between PICU stay and a worse patient outcome, which can be explained by the fact that all the mentioned indications for PICU admission adversely affect the outcome in these patients.
The results of our study suggest that most of our patients developed DKA because they had omitted insulin doses; also poor compliance was associated with severity of DKA in the study population. An earlier study from Pakistan showed that insulin omission precipitated DKA in upto 38% pediatric patients with established diabetes.8 In order to prevent the development of DKA in children in our setting, physicians will have to ensure that patients and their parents/guardians understand the importance of the need for regular insulin injections and regular monitoring of blood glucose. This can be reinforced by patient and parent education on each clinic visit and regular follow ups. It is also important to educate them about sick-day management.27 Another factor that can help achieve this is the establishment and maintenance of a 24-hours diabetic help line which can guide the carers of diabetic children after the clinic hours. There is also a strong need for the dissemination of information about diabetes and DKA to community physicians and general population.
We acknowledge that this was a single centre study at a tertiary care hospital in Karachi that relied primarily on retrospective data for derivation of conclusions. However, despite the retrospective nature of the study, we have tried to define some contributors of outcomes in a large sample that may be helpful in improving practices.
Conclusions
Poor compliance is an important modifiable precipitating factor for diabetic ketoacidosis in children in our population. Education of patients and their parents/guardians with regards to the importance of regular blood sugar monitoring and insulin dosing should be considered an important long-term management strategy. Further studies are needed to improve the data about DKA in the paediatric population in Pakistan.
References
1.Dunger DB, Sperling MA, Acerini CL, Bohn DJ, Daneman D, Danne TP, et al. European Society for Paediatric Endocrinology/ Lawson Wilkins Paediatric Endocrine Society consensus statement on diabetic ketoacidosis in children and adolescents. Paediatrics 2004; 113: e133-40.
2.Curtis JR, To T, Muirhead S, Cummings E, Daneman D. Recent trends in hospitalization for diabetic ketoacidosis in Ontario children. Diabetes Care 2002; 25: 1591-6.
3.Edge JA, Ford-Adams ME, Dunger DB. Causes of death in children with insulin dependent diabetes 1990-96. Arch Dis Child 1999; 81: 318-23.
4.Vanelli M, Chiarelli F. Treatment of diabetic ketoacidosis in children and adolescents. Acta Biomed 2003; 74: 59-68.
5.Glaser N, Barnett P, McCaslin I, Nelson D, Trainer J, Lovie J, et al. Risk factors for cerebral edema in children with diabetic ketoacidosis. The Pediatric Emergency Medicine Collaborative Research Committee of the American Academy of Pediatrics. N Engl J Med 2001; 344: 264-9.
6.Edge JA, Hawkins MM, Winter DL, Dunger DB. The risk and outcome of cerebral oedema developing during diabetic ketoacidosis. Arch Dis Child 2001; 85: 16-22.
7.Levy-Marchal C, Patterson CC, Green A; Eurodiab ACE Study Group. Europe and Diabetes. Geographical variation of presentation at diagnosis of type 1 diabetes in children: the EURODIAB Study. European and Diabetes Diabetologia 2000; 44 (Suppl 3): B: 75-80.
8.Lone SW, Siddiqui EU, Muhammad F, Atta I, Ibrahim MN, Raza J. Frequency, clinical characteristics and outcome of diabetic ketoacidosis in children with type-1 diabetes at a tertiary care hospital. J Pak Med Assoc 2010; 60: 725-9.
9.Jabbar A, Farooqui K, Habib A, Islam N, Haque N, Akhter J. Clinical characteristics and outcomes of diabetic ketoacidosis in Pakistani adults with Type 2 diabetes mellitus. Diabet Med 2004; 21: 920-3.
10.Shera AS, Miyan Z, Basit A, Maqsood A, Ahmadani MY, Fawwad A, et al. Trends of type 1 diabetes in Karachi, Pakistan. Pediatr Diabetes 2008; 9: 401-6.
11.Akhter J, Jabbar A, Islam N, Khan MA. Diabetic ketoacidosis in a hospital based population in Pakistan. J Pak Med Assoc 1993; 43: 137-9.
12.Qayyum AA, Lone SW, Ibrahim MN, Atta I, Raza J. Effects of diabetes self-management education on glycaemic control in children with insulin-dependent diabetes mellitus. J Coll Physicians Surg Pak 2010; 20: 802-5.
13.Naveed D, Bilal N, Nasir B, Lodhi BUR. Precipitating factors for diabetic ketoacidosis. KUST Med J 2009; 1: 6-8.
14.Sheikh AAI, Malloush A. Diabetic ketoacidosis: more cases seen during summer vacation. Pak J Med Sci 2009; 25: 512-5.
15.Niaz Z, Razzaq A, Chaudhary UJ, Awais M, Yaseen MA, Naseer I, et al. Mortality review of diabetic ketoacidosis in Mayo Hospital, Lahore - Pakistan. Biomedica 2005; 21: 83-5.
16.Shastry RM, Bhatia V. Cerebral edema in diabetic ketoacidosis. Indian Pediatr 2006; 43: 701-8.
17.Singh D, Chopra A, Pooni PA, Bhatia RC. A clinical profile of shock in children in Punjab, India. Indian Pediatr 2006; 43: 619-23.
18.Rodacki M, Pereira JR, Nabuco de Oliveira AM, Barone B, MacDowell R, Perricelli P, et al. Ethnicity and young age influence the frequency of diabetic ketoacidosis at the onset of type 1 diabetes. Diabetes Res Clin Pract 2007; 78: 259-62.
19.Angus VC, Waugh N. Hospital admission patterns subsequent to diagnosis of type 1 diabetes in children: a systematic review. BMC Health Serv Res 2007; 7: 199.
20.Edge JA, Roy Y, Bergomi A, Murphy NP, Ford-Adams ME, Ong KK, et al. Conscious level in children with diabetic ketoacidosis is related to severity of acidosis and not to blood glucose concentration. Pediatr Diabetes 2006; 7: 11-5.
21.Flood RG, Chiang VW. Rate and prediction of infection in children with diabetic ketoacidosis. Am J Emerg Med 2001; 19: 270-3.
22.Ismail NA, Kasem OM, Abou-El-Asrar M, El-Samahy MH. Epidemiology and management of type 1 diabetes mellitus at the ain shams university pediatric hospital. J Egypt Public Health Assoc 2008; 83: 107-32.
23.Smith CP, Firth D, Bennett S, Howard C, Chisholm P. Ketoacidosis occurring in newly diagnosed and established diabetic children. Acta Paediatr 1998; 87: 537-41.
24.Bohn D, Daneman D. Diabetic ketoacidosis and cerebral edema. Curr Opin Pediatr 2002; 14: 287-91.
25.Tasker RC, Lutman D, Peters MJ. Hyperventilation in severe diabetic ketoacidosis. Pediatr Crit Care Med 2005; 6: 405-11.
26.Magee MF, Bhatt BA. Management of decompensated diabetes. Diabetic ketoacidosis and hyperglycemic hyperosmolar syndrome. Crit Care Clin 2001; 17: 75-106.
27.Bismuth E, Laffel L. Can we prevent diabetic ketoacidosis in children? Pediatr Diabetes 2007; 8 (Suppl 6): 24-33.
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




