Arshalooz J. Rahman ( Department of Pediatrics and Child Health, Aga Khan University Hospital, Karachi. )
Sohail Ashraf ( Department of Pediatrics and Child Health, Aga Khan University Hospital, Karachi. )
Farah Naz ( Department of Pediatrics and Child Health, Aga Khan University Hospital, Karachi. )
January 2011, Volume 61, Issue 1
Original Article
Abstract
Objectives: To determine the significance of pyuria as a predictor of culture proven urinary tract infections (UTI) in neonates and to assess the frequency of urinary tract anomalies in neonates with pyuria.
Methods: Prospective study conducted at the neonatal intensive care unit of a tertiary care hospital; (Liaquat National Hospital Karachi) for a period of 4 months from April 2008 to August 2008. One hundred and ten consecutive infants < 28 days of age admitted to the NICU of Liaquat National Hospital for medical reasons were included in the study. Information regarding age, gender, antenatal history, birth weight, clinical examination, laboratory findings and outcome were recorded on a questionnaire. Septic work up was performed and urine samples were collected using urethral catheterization. Patients showing any number of white blood cells on microscopy were included and their urine sent for culture. All patients had Ultrasound done during their hospital stay and those patients with any renal abnormality were further investigated with Micturating cyestoretherogram (MCUG) at 6 weeks.
Results: Out of one hundred and ten patients admitted, thirty five patients showed the presence of pus cells in urine and were included in the study. Of the 35 neonates with pyuria, 71.4% had no growth in urine cultures and 38.2 %neonates with insignificant pyuria (< 9 cells in urine) showed a positive culture. The renal ultrasound was normal in 51.4% neonates with pyuria although it was abnormal in 100% of the subjects with higher number of pus cells in urine (>20 pus cells).
Conclusion: Pyuria is not a useful marker for the diagnosis of culture proven UTI in neonates it cannot be used as an indicator of underlying renal abnormality, though it may have some utility in neonates with >20 /numerous pus cells.
Keywords: Pyuria, Urinary tract infection, Neonates (JPMA 61:70; 2011).
Introduction
Neonatal infections present with non-specific and subtle signs and various guidelines have been developed and practiced for the management of neonatal sepsis.1 Most paediatricians perform a complete sepsis work-up before starting antibiotic therapy in neonates. Management is clear cut if a source of infection is identified, but in some cases the diagnosis is not clearly established. This is especially true for urinary tract infections (UTI), where there is no cut off defined in terms of the number of pus cells for establishing the diagnosis in sick neonates.2 Significance of screening tests like nitrite positivity and gram stain on urine sample has been debated in different studies but these tests can under or over diagnose UTI in absence of urine culture results.1 Screening tests based on dipstick leukocyte esterase cannot accurately detect pyuria in young febrile children. It does not exclude pyuria, therefore using it alone without microscopy and culture can miss a lot of cases of urinary tract infection, and increase the chance of false negative results.3 A cut off of <10 WBC per high-power field in a centrifuged urine specimen has been taken as "normal urine" in few studies.3,4 However, many a times in our clinical practice we see that even in the presence of 15-20 WBCs, the urine culture is negative and vice versa. No single cut-off has high sensitivity and specificity to diagnose and exclude UTI.5 This may be due to the fact that newborns have an immature immune response therefore leucocyte count in urine cannot reliably predict UTI. Symptoms of urosepsis are more generalized with hardly any sign specific to the urinary tract. If suspected, a urine culture should be ordered before the first dose of antibiotic. Furthermore the neonate should be screened for anatomical malformations of the urinary tract as there is a higher incidence of UTI in neonates with underlying urogenital malformations.6,7
Studies have been done in infants to determine the predictive value of pyuria and urine culture but its application in neonates is controversial.7 This is due to many reasons related to not only difficulty in sample collection in neonates but also sampling techniques like bag verses catheter or suprapubic sample and prior antibiotic intake. Urine collection methods alter the diagnostic validity of urinalysis. These differences have important implications for the diagnostic and therapeutic management of children with suspected UTI.8 All these factors play a significant role in our setting. Furthermore, the diagnostic accuracy and the interpretation of urine analysis is influenced by the preparation of the specimen (centrifuged vs uncentrifuged), and the method of quantifying and reporting leukocytes (per microscopic high-power field vs per cubic millimeter). To the best of our knowledge, no study has yet examined the relationship between a positive urine culture and the number of pus cells in urine. The relationship of pyuria with urinary tract abnormalities in newborns also remains to be established, although relationship with culture proven UTI is well established.9
Therefore, we undertook this study to determine the significance of pyuria as a predictor of culture proven urinary tract infections (UTI) in neonates and to assess the frequency of urinary tract anomalies in neonates with pyuria.
Patients and Methods
A prospective study was conducted at the neonatal intensive care unit of Liaquat National Hospital (LNH), which is a 750 bedded, tertiary care hospital located in the city center. The Neonatal Intensive Care Unit (NICU) is a 10 bedded unit. Most of the patients admitted to the NICU are babies delivered in the LNH itself. However, the NICU accepts a limited number of referrals from outside hospitals. The average yearly neonatal admission rate to the unit is 400 -500.
The Patients (<28 days) admitted to the Neonatal unit for medical reason like sepsis, respiratory distress, hyperbilirubinaemia and prematurity were included in the study.
Patients who had received antibiotics prior to admission, neonates with known urogenital abnormality and neonates admitted for surgical reasons were excluded from the study.
The study was conducted for a period of 4 months from April 2008 to August 2008 and one hundred and ten consecutive neonates were enrolled. After informed consent, a detailed history and examination was conducted on all eligible subjects, immediately after admission. Information regarding age, gender, antenatal history, birth weight, clinical examination, laboratory findings and outcome were recorded on a pre-designed questionnaire.
A septic work up, including CBC, CRP, urine D/R, blood and urine culture was performed on all patients . Cerebrospinal fluid was not tapped as a routine and the procedure was only performed on patients where the diagnosis of meningitis was suspected. Sepsis was defined when blood and urine were positive for identical bacteria. All samples of urine were collected using urethral catheterization. All patients showing any number of white blood cells per high-power field on a centrifuged specimen of urine microscopy were included and sent for culture.
An ultrasound of the kidneys was performed on all neonates during their stay in the hospital. If a renal abnormality like hydronephrosis and hydroureter was detected on ultrasound it was further investigated with MCUG after 6 weeks. The data was analyzed using SPSS 16.0. The results were expressed in percentages.
Results
During the four month study, one hundred and ten patients fulfilled the eligibility criteria. Out of these thirty five patients (n=35) showed the presence of pus cells in urine and were included in the final analysis. Majority (57%) of the patients were female and the common presenting features were fever or temperature instability (26%), vomiting (28.6 %), lethargy (25.7%) and late neonatal jaundice (36%). The median age of the patients was 3 days (range 1-26 days). History of urinary discoloration was present in 45.7%, whereas haematuria was detected on lab investigation in 15% of the patients only. The TLC count was more than 15000 in 30% and a similar number of patients had an elevated absolute neutrophil count (>70%). Urine culture was positive in 28.7% neonates (Table-1).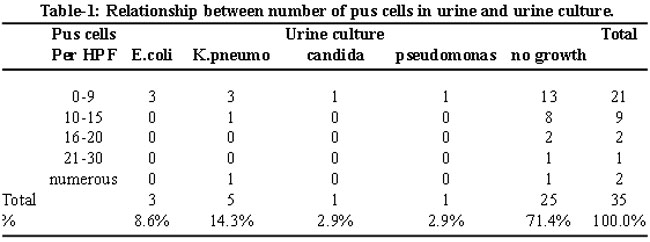
There was no relationship observed between urine and blood culture, except for one patient who grew E.coli on both blood and urine culture.
For the purpose of analysis, the numbers of pus cells in urine were categorized into four groups (Table-1). Each group was analyzed for the presence or absence of a positive urine culture. Most (60%) of the neonates had < 9 cell/hpf and 38% neonates in this group had culture positive. The group with numerous or > than 20 cells/ hpf had only one culture positive (20%). There was a significant relationship between any number of pus cells in urine and a positive culture (P=0.007). However when Fisher\'s exact test was applied to test the relationship between low number of pus cells in urine (?9 cell/hpf) and a positive urine culture the relationship was not significant (P= 0.53).
Ultrasound was performed on all subjects during their hospital stay. Nine patients (25.7%) with < 9 cells in urine had a renal abnormality detected on ultrasound (Table-2).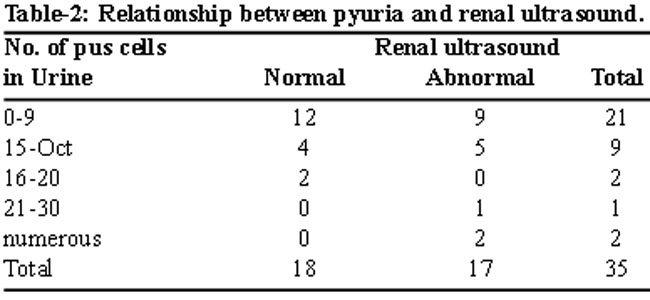
There were 3 neonates in the two groups categorized >20 / numerous pus cells and 100% of them had a renal abnormality detected on ultrasound.
Seventeen patients had an evidence of renal pathology on the initial ultrasound. These subjects were followed up with an Micturating cysto uretherogram (MCUG) at the age of 6 weeks and nine showed evidence of underlying congenital renal anomaly (Table-3).
Seven of these neonates had < 9 cell/hpf and two had >20 / numerous pus cells. (Fisher Exact P value= 0.31).
Discussion
Neonatal sepsis is one of the leading causes of neonatal morbidity and mortality in the developing world. Urosepsis particularly due to Urinary tract infection has a major contribution and is an important cause of serious bacterial infection in neonates affecting 1 in 3 babies with proven bacterial infection, and it may be the first indicator of underlying structural abnormalities.10 In neonatal age there are no direct symptoms related to the urinary tract. Presentation is vague which makes urinary tract infection a less likely source of infection.11 Pyuria has been documented as a sign of urinary infection in neonate, however it is still not clear what number of pus cells in urine should be considered abnormal and whether the cutoff of 10 cells/high power field stands valid for neonates or lesser number of pus cells should be considered as cutoff. Most authors support pyuria of <10 cell /hpf as being indicative of presence of low risk of bacterial infections.12,13 However, in our study we had the experience of having culture positive in the presence of less number of cells (pyuria 1-9 cells/hpf). Some authors agree with the fact that the sensitivity, specificity, and positive predictive value of the urine analysis is low and only a third to half of patients with positive urine culture results can be identified correctly.14 This decreases the significance of pyuria as a screening tool in this age group. Neonates have an immature immune system usually with leucocytosis in the blood in case of sepsis.15 However, uroepithelial responses have not been studied in detail in this age group, decreased quantity of glycosaminglycans (GAGS) in urothelial cell in children with UTI have been documented in cases with underlying anatomical malformation but there is no data supporting leucocyte urothelial responses.16
Therefore the dipstick as a method of urine analysis has a less reliable screening tool for infants and should not be used to exclude urinary tract infection as a single test modality.3 The practice of sending urine culture as a part of septic screen in newborns is still in practice and is justified as urine analysis may miss a lot of cases of UTI in neonates especially in premature neonates.7 Long term consequences of recurrent UTI can be devastating in children leading to renal scarring, hypertension and chronic kidney disease.17
In this study unlike outside data, 57 % of the subjects were female.12,14 Most of the newborns were admitted with non specific symptoms like temperature instability (hyperthermia or hypothermia), lethargy, vomiting and hyperbilirubinaemia. All of these symptoms can be seen in neonatal sepsis and are not specific to urinary tract infections. Other studies in neonates have reported similar findings.18
Urinary tract infection was the source of bacteraemia in only one neonate in the present study. Joshi et al, have similarly reported a very low rate (0.4%) of bacteraemia due to underlying UTI.19
Altered urine colour was the presenting symptom in a significant number of neonates but a small number of urine specimens showed haematuria, the probable underlying reason for this was hyperbilirubinaemia rather than UTI.
Klebsiella pneumoniae, was the commonest pathogen identified in urine culture in our study, whereas most studies from the west have documented E.coli as the predominant organism.20
Urinary tract abnormalities as an under lying cause of UTI has been recognized for a long time and complete evaluation is warranted by ultrasound and MCUG once infection is diagnosed.14 Recently newer modalities like Comprehensive ultrasound are being recommended to replace diagnostic modalities like DMSA, MCUG to reduce the radiation hazard.21 In this study there were more females with underlying abnormality unlike other data which favours severe reflux disease in males or an equal preponderance.22,23 The the most common urinary tract abnormality detected on ultra sound was hydronephrosis and MCUG at 6weeks of age which demonstrated grade 4 and 5 VUR in a significant number of subjects. Vesicouretral Reflux VUR has been documented as a leading cause of renal scarring and chronic kidney disease among children. Urinary tract abnormality was documented on MCUG at six weeks follow up in 50% of our study group as well.21,22
Generalizibility of the results of this study may be limited because of small sample size, larger studies are needed to establish a definite correlation between presence of pyuria and culture positivity.
Conclusion
Pyuria alone is not a reliable screening tool for UTI and can miss culture positive Urinary tract infection. Pyuria is also not a reliable marker for underlying urogenital abnormalities. Late neonatal jaundice due to non haemolytic causes should warrant urine culture as this may be the only sign on presentation. Efforts should be made for early diagnosis of UTI in neonates as it can have devastating consequences if missed.
References
1.Mathur NB. Neonatal sepsis. Indian Pediatr 1996; 33: 663-74.
2.Paci A, Giorgi PL, Vizzoni L, Ceccarelli M. [Pyuria in the premature infant]. Riv Clin Pediatr 1964; 74: 391-414.
3.Yuen SF, Ng FN, So LY. Evaluation of the accuracy of leukocyte esterase testing to detect pyuria in young febrile children: prospective study. Hong Kong Med J 2001; 7: 5-8.
4.Fang SB, Lee HC, Yeung CY, Tsai JD. Urinary tract infections in young infants with prolonged jaundice. Acta Paediatr Taiwan 2005; 46: 356-60.
5.Cheng YW, Wong SN. Diagnosing symptomatic urinary tract infections in infants by catheter urine culture. J Paediatr Child Health 2005; 41: 437-40.
6.Vacant J, Cukier J. [Study of 100 congenital anomalies of the pyelo-ureteral junction in the child]. J Urol Nephrol (Paris) 1976; 82: 896-904.
7.Hoberman A, Wald ER, Reynolds EA, Penchansky L, Charron M. Is urine culture necessary to rule out urinary tract infection in young febrile children? Pediatr Infect Dis J 1996; 15: 304-9.
8.McGillivray D, Mok E, Mulrooney E, Kramer MS. A head-to-head comparison:"clean-void" bag versus catheter urinalysis in the diagnosis of urinary tract infection in young children. J Pediatr 2005; 147: 451-6.
9.Antwi S BI, Baffoe-Bonnie B. Urine Dipstick as a Screening test for Urinary Tract INfection. Am Trop Paediatr 2008; 6: 117-22.
10.Barton M, Bell Y, Thame M, Nicholson A, Trotman H. Urinary tract infection in neonates with serious bacterial infections admitted to the University Hospital of the West Indies. West Indian Med J 2008; 57: 101-5.
11.Ghaemi S, Fesharaki RJ, Kelishadi R. Late onset jaundice and urinary tract infection in neonates. Indian J Pediatr 2007; 74: 139-41.
12.Chiu CH, Lin TY, Bullard MJ. Identification of febrile neonates unlikely to have bacterial infections. Pediatr Infect Dis J 1997; 16: 59-63.
13.Hoberman A, Wald ER, Reynolds EA, Penchansky L, Charron M. Is urine culture necessary to rule out urinary tract infection in young febrile children? Pediatr Infect Dis J 1996; 15: 304-9.
14.Lin DS, Huang SH, Lin CC, Tung YC, Huang TT, Chiu NC, et al. Urinary tract infection in febrile infants younger than eight weeks of age. Pediat 2000; 105: e20.
15.Gregory J, Hey E. Blood neutrophil response to bacterial infection in the first month of life. Arch Dis Child 1972; 47: 747-53.
16.Di Stefano A, Cingolani M, Podesta E, Di Rovasenda E, Cipriani C. [Glycosaminoglycans as a defense mechanism of the urothelium, in children]. Minerva Pediatr 1989; 41: 167-71.
17.Vachvanichsanong P. Urinary tract infection: one lingering effect of childhood kidney diseases--review of the literature. J Nephrol 2007; 20: 21-8.
18.Freedman SB, Al-Harthy N, Thull-Freedman J. The crying infant: diagnostic testing and frequency of serious underlying disease. Pediatr 2009; 123: 841-8.
19.Joshi SG, Ghole VS, Niphadkar KB. Neonatal gram-negative bacteremia. Indian J Pediatr 2000; 67: 27-32.
20.Goldman M, Lahat E, Strauss S, Reisler G, Livne A, Gordin L, et al. Imaging after urinary tract infection in male neonates. Pediatr 2000; 105: 1232.
21.Brader P, Riccabona M, Schwarz T, Seebacher U, Ring E. Value of comprehensive renal ultrasound in children with acute urinary tract infection for assessment of renal involvement: comparison with DMSA scintigraphy and final diagnosis. Eur Radiol 2008; 18: 2981-9.
22.Hsieh MH, Madden-Fuentes RJ, Roth DR. Urologic diagnoses among infants hospitalized for urinary tract infection. Urology 2009; 74: 100-3.
23.Kanellopoulos TA, Salakos C, Spiliopoulou I, Ellina A, Nikolakopoulou NM, Papanastasiou DA. First urinary tract infection in neonates, infants and young children: a comparative study. Pediatr Nephrol 2006; 21: 1131-7.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




