Muhammad Tariq ( Department of Medicine, Aga Khan University Hospital, Karachi, Pakistan. )
Taimur Saleem ( Medical Graduates, Aga Khan University Hospital, Karachi, Pakistan. )
Hamid Ullah ( Medical Graduates, Aga Khan University Hospital, Karachi, Pakistan. )
Vikram Mehraj ( Department of Pathology and Microbiology, Aga Khan University Hospital, Karachi, Pakistan. )
Abdul Jawwad Samdani ( Medical Graduates, Aga Khan University Hospital, Karachi, Pakistan. )
Syeda Hena Kazmi ( Medical Graduates, Aga Khan University Hospital, Karachi, Pakistan. )
Syed Imran Ayaz ( Medical Graduates, Aga Khan University Hospital, Karachi, Pakistan. )
Mehmood Riaz ( Department of Medicine, Aga Khan University Hospital, Karachi, Pakistan. )
Mohammed Junaid Patel ( Department of Medicine, Aga Khan University Hospital, Karachi, Pakistan. )
Jaweed Akhtar ( Department of Medicine, Aga Khan University Hospital, Karachi, Pakistan. )
Mohammed Asim Beg ( Department of Pathology and Microbiology, Aga Khan University Hospital, Karachi, Pakistan. )
January 2011, Volume 61, Issue 1
Original Article
Abstract
Objective: To compare the clinical outcomes of a loading dose regimen of quinine with a uniform dose regimen in patients with severe falciparum malaria.
Methods: A retrospective chart review of 315 patients admitted with severe falciparum malaria and treated with quinine at a tertiary care teaching hospital of Karachi, Pakistan during 1999-2006 was conducted. Group A with 103 patients (32.7%) was given an initial loading dose of quinine while group B with 212 patients (67.3%) did not receive the loading dose. The two groups were compared in terms of reduction of parasite load, resolution of fever, recovery of consciousness and incidence of adverse effects. Outcome parameters were measured on the third day of therapy.
Results: More individuals in group A (62.1%) were afebrile as compared to group B (54.7%) at day 3 of therapy. Patients in group B showed greater reduction in parasitaemia (47.2% at baseline to 4.7% on day 3) as compared to group A (56.3 % at baseline to 9.7% on day 3). Following therapy, fewer patients in group B had altered consciousness (7.1% at baseline to 4.7% on day 3) as compared to patients in group A (7.8% at baseline to 5.8% on day 3). However, these associations were not statistically significant. The incidence of thrombocytopenia was higher in Group A (5.8%) as compared to Group B (0.9%).
Conclusion: Although quinine loading dose may be more effective than uniform dose in rapid fever clearance; it also appears to be associated with higher toxicity. Uniform dose of quinine may be prescribed in severe falciparum malaria in view of its better safety profile.
Keyworks: Severe falciparum malaria; Quinine; Loading Dose; Pakistan (JPMA 61:27; 2011).
Introduction
Falciparum malaria is a severe and a potentially life-threatening parasitic disease with increasing prevalence in most endemic regions of the world. This translates into devastating health and socio-economic consequences; especially in areas where there is increasing drug resistance in the causative organism or where the cases are inappropriately managed.1,2 The disease affects more than 500 million people and causes 2-3 million deaths each year worldwide.2-4 Malaria is also a major health problem in Pakistan, with an estimated number of annual episodes of about 1.6 million. Of these, about 33% are reported as falciparum malaria cases. Most of the disease episodes have been recorded in the post-monsoon period (August-November); the stagnant water pools providing optimal breeding conditions for the mosquitoes.5
Falciparum malaria is associated with significant morbidity and mortality.2,6 In particular, severe falciparum malaria may be complicated by Acute Renal Failure (ARF), Disseminated Intravascular Coagulation (DIC), Cerebral Malaria, Hypoglycaemia, Metabolic Acidosis, Pneumonia, Acute Respiratory Distress Syndrome (ARDS) and Anaemia.4,7
Early and effective treatment of severe falciparum malaria may shorten its duration and prevent development of complications with a reduction in associated mortality. However, there is little consensus with regards to the most optimum modus operandi for the therapeutic management of this condition. Quinine is still being used as a first-line drug in falciparum malaria since resistance to Chloroquine has been widely reported from many parts of the world including Pakistan.2,4,6,8 The World Health Organization (WHO) recommends in its handbook for management of severe malaria, that the course of treatment with Quinine may follow a high initial Loading Dose (qLD) regimen of 20mg/kg of body weight followed by Maintenance Dose (qMD) of 10mg/kg repeated 8 hourly to complete a 7 day course of treatment.4
However, according to a Cochrane Review of 2004,9 the use of an initial loading dose of quinine remains debatable. Randomized controlled trials (RCTs) assessing this subject are few and far between; only 4 trials (n=144) fulfilled the inclusion criteria of this Cochrane Review. It was concluded that while qLD was associated with fewer deaths, this association was not statistically significant. qLD caused a reduction in fever clearance time and parasite clearance time but there was no statistically significant impact on the recovery of consciousness, neurological sequelae, as convulsions.
Thus on one hand, there are studies that have illustrated the use of a high initial IV Quinine Loading Dose as being beneficial;2,10-12 on the other hand, there are studies that negate the advantages of the Loading Dose Regime over standard treatment of maintenance dose for severe/complicated malaria.6,13,14
Thus, it is important to collect more data on the subject in order to delineate the direction of the therapeutic trend with regards to use of loading dose of quinine. Current data are insufficient to directly demonstrate the benefits or drawbacks of this form of treatment. Therefore, the current study was undertaken to assess the efficacy and safety of Quinine Loading Dose in the treatment of severe falciparum malaria in a malaria endemic geographical region of the world in order to evaluate its effect on various parameters such as (1) reduction of parasite load, (2) resolution of fever, (3) recovery of consciousness and (4) adverse effects. The results of this study may, therefore, help improve current prescribing practices in the treatment of severe falciparum malaria.
Methods
This retrospective review of patient charts was conducted in the Department of Medicine, Aga Khan University Hospital (AKUH) Karachi, Pakistan. A computerized data base search for anonymous patient information was conducted under the guidelines of Ethical Review Committee (ERC) of the Aga Khan University Hospital. No patient was individually identified and the data was obtained in an anonymous fashion without ever alluding to the identity of the patient. The data of hospitalized adult patients was extracted meeting the following inclusion criteria: a) age 15 or above, b) admitted with the diagnosis of 'Severe Falciparum Malaria' c) admitted during an 8 year period from January 1, 1999 to December 31, 2006 d) treated with intravenous (IV) or oral Quinine. All hospitalized patients were considered to have severe malaria based on the categorization as per WHO classification for severe falciparum malaria.4 We excluded those patients who were negative for Malarial Parasite (MP) on admission or who were diagnosed on the basis of Immuno-chromatographic (ICT) test. Quantitative assessment of parasitemia through MP test was categorized as 'heavy' when there were more than 1000 parasites/100 high power microscopic field, 'moderate' when there were 11-1000 parasites/100 high power microscopic fields and "scanty" when there were 1-10 parasites/100 high power microscopic fields. Two comparison groups were made based on the administration of quinine regime. The loading dose (qLD) regime consisted of 20mg/kg of body weight given IV followed by maintenance dose of 10 mg/kg body weight given either IV or oral repeated 8 hourly for 5-7 days. The "no loading" dose regime comprised of quinine maintenance dose (qMD) only. The primary comparison between the two groups in terms of different parameters was done on the 3rd day after therapy was administered. Data was entered and analyzed using Windows Statistical Package for Social Sciences (SPSS) version 14.15 Data were described calculating means and standard deviations for continuous variables and frequencies for categorical variables. Univariate analysis was conducted defining dependent variables on the basis of first dose of quinine therapy and it was categorized in two groups, i.e., quinine loading dose (qLD) group and no loading dose group (qMD). Student's t-test was used to compare means among qLD and qMD groups. Chi-square test or Fisher's exact test, as appropriate, were used to compare categorical independent variables among the two groups. A p-value of < 0.05 was considered statistically significant; unless otherwise specified.
Results
A total of 315 patients were included in this study. The mean age was 38.5 ± 16.8 years with 239 (75.9%) males and 76 (24.1%) females. Most patients (87.9%) were admitted through emergency room (ER) while the remaining were admitted through consulting clinics. The majority (69.8%) of the patients were admitted during a four month post-monsoon period from August to November. The initial Loading dose of quinine (qLD) was given to Group A with 103 (32.7%) patients. Group B with 212 (67.3%) patients did not receive qLD. All patients received maintenance dose, either IV (33%) or oral (67%).
Thrombocytopenia, cerebral malaria, anaemia, pneumonia, Acute Respiratory Distress Syndrome (ARDS), metabolic acidosis, hypoglycaemia, volume depletion and jaundice were complications equally distributed (p>0.05) in the two groups at the time of presentation. Acute renal failure (p=0.008) and disseminated intravascular coagulation (DIC) (p=0.008) were complications more common in patients in Group A. A comparison of other clinical parameters between the two groups including altered level of consciousness (GCS 14 or less), documented mean temperature and quantitative MP on microscopy is shown in Table-1.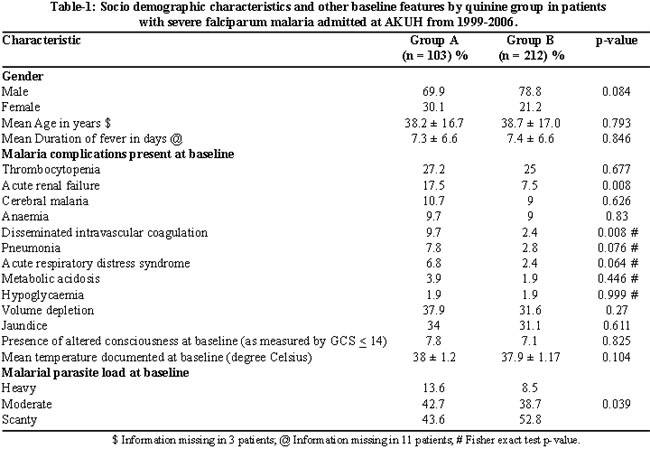
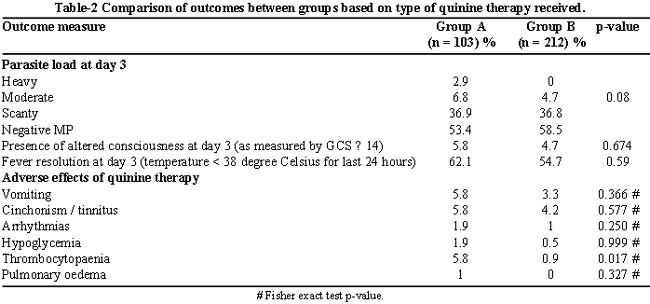
Table-2 shows a comparison of the outcomes between the two groups. These were measured on the third day of the administration of therapy. It was seen that by third day of therapy, the difference in quantitative parasitaemia between the two groups was insignificant (p=0.08). In terms of recovery of consciousness at day 3, the two groups were again comparable (p=0.674). There was no statistically significant difference between the two groups in terms of fever resolution at day 3 (p=0.59). However, more individuals in Group A were afebrile as compared to Group B (62.1 % vs. 54.7%).
Group A and Group B were also comparable with regards to the majority of the in-hospital quinine-specific major side effects including cinchonism/tinnitus (headache, dizziness, hearing loss, and ringing in the ears) nausea/vomiting, hypoglycaemia, pulmonary oedema and cardiac arrhythmia (prolongation of QT interval). The groups, however, showed a statistically significant difference with regards to the incidence of thrombocytopenia (platelet count < 150,000 cells per microliter); more patients who had received qLD showed this adverse effect (p=0.017).
Discussion
This study assessed the effectiveness and beneficial aspects of incorporating the rationale of a high initial IV quinine loading dose in the treatment of adult patients with moderate to severe and/or complicated falciparum malaria. Demographic details in this study showed that majority of patients were males possibly as a result of more exposure to the vectors of malaria although no evidence was found to substantiate this internationally.
According to literature, quinine has evolved to be the first-line drug for severe / complicated falciparum malaria.2,4,6,9,16,17 As per standard recommendations of WHO,4 the globally advised rationale comprises of high initial loading dose of quinine followed by a maintenance dose.
Our analysis showed that only one third of the patients received qLD, which in addition to the attending physician\'s choice may be attributed to either lack of ascertainment of prior drug administration, or use of quinine and its derivatives in past 12 hours.4,6 Results of our study show success in early achievement of fever clearance in patients who received qLD, but parasite clearance (negative MP) was not significant.
Assessment of scientific literature and discussions over the efficacy and advantages of quinine loading dose regime revealed that qLD may contribute a beneficial effect in terms of Fever Clearance Time (FCT) as well as Parasite Clearance Time (PCT).2,9,10,14,18-20
Patients given qLD showed an improvement in the level of consciousness, accounting for early recovery of mental stability. Fargier et al19 commented on significant reduction of coma recovery time in patients on qLD therapy. However, the Cochrane Review9 advocates insufficient evidence to determine association of qLD with shorter recovery of consciousness, with many other citations suggesting no beneficial effects on the coma recovery.6,10,13,18
Other authors have suggested IV quinine dosage regimen may be associated with multiple adverse effects, which lead to poor compliance and side effects including, cinchonism, cardiac arrythmias, hypotension, vomiting and abdominal pain, thrombocytopenia, anaemia, partial hearing loss/tinnitus and hypoglycaemia.2,4,6,17,18,21-23 Many authors have cited that qLD was affiliated more with the development of these side-effects rather than the no qLD regimen.9,13,14,16,18,20,24,25 We observed no difference in terms of adverse effects except thrombocytopenia which was higher in Group A as compared to Group B.
A few limitations of this study include its retrospective design and patient sample from a single hospital. Also, while studying malaria cases for a period of eight years, we encountered deficient data in the compiled records, leading to elimination of such cases. Attention in these areas is advised for future research to collate significant results. Paucity of data from South Asian countries should be addressed with well designed prospective clinical trials which we are planning to conduct with the baseline information from the current study.
Conclusion
This study showed that quinine loading dose may be more effective than uniform dose in rapid fever clearance but it is also associated with higher toxicity. Maintenance dose of quinine maybe prescribed in severe falciparum malaria because of its better safety profile. Further well-designed, prospective studies are needed to directly demonstrate the impact of loading dose on parasitemia reduction and consciousness recovery in severe falciparum malaria.
Acknowledgement
We would like to acknowledge Dr. Mohammad Wasay (Section of Neurology, Department of Medicine, Aga Khan University) for his valuable suggestions for improving the manuscript.
References
1.Nosten F, Ashley E. The detection and treatment of Plasmodium falciparum malaria: time for change. J Postgrad Med 2004; 50: 35-9.
2.Pasvol G. The treatment of complicated and severe malaria. Br Med Bull 2005; 75-76: 29-47.
3.Gogtay NJ. Malaria-hope on the horizon. J Postgrad Med 2004; 50: 5-6.
4.WHO. Management of severe malaria: A practical handbook. 2nd ed. Geneva: World Health Organization; 2000.
5.WHO-EMRO. Epidemiological situation. WHO-Eastern Mediterranean Regional Office, 2005. (Online) 2010 (Cited 2010 Sep 14). Available from URL: http://www.emro.who.int/rbm/countryprofiles-pak.htm.
6.Mishra SK, Mohanty S, Mohanty A, Das BS. Management of severe and complicated malaria. J Postgrad Med 2006; 52: 281-7.
7.Beg MA, Sani N, Mehraj V, Jafri W, Khan MA, Malik A, et al. Comparative features and outcomes of malaria at a tertiary care hospital in Karachi, Pakistan. Int J Infect Dis 2008; 12: 37-42.
8.Khan MA, Smego RA, Razi ST, Beg MA. Emerging drug-resistance and Gzidelines for treatment of malaria. J Coll Physicians Surg Pak 2004; 14: 319-24.
9.Lesi A, Meremikwu M. High first dose quinine regimen for treating severe malaria. Cochrane Database Syst Rev 2004; CD003341.
10.Mehta SR, Lazar AI, Kasthuri AS. Experience on loading dose--quinine therapy in cerebral malaria. J Assoc Physicians India 1994; 42: 376-8.
11.White NJ, Looareesuwan S, Warrell DA, Warrell MJ, Chanthavanich P, Bunnag D, et al. Quinine loading dose in cerebral malaria. Am J Trop Med Hyg 1983; 32: 1-5.
12.Winstanley PA, Mberu EK, Watkins WM, Murphy SA, Lowe B, Marsh K. Towards optimal regimens of parenteral quinine for young African children with cerebral malaria: unbound quinine concentrations following a simple loading dose regimen. Trans R Soc Trop Med Hyg 1994; 88: 577-80.
13.Pukrittayakamee S, Supanaranond W, Looareesuwan S, Vanijanonta S, White NJ. Quinine in severe falciparum malaria: evidence of declining efficacy in Thailand. Trans R Soc Trop Med Hyg 1994; 88: 324-7.
14.Tombe M, Bhatt KM, Obel AO. Quinine loading dose in severe Falciparum malaria at Kenyatta National Hospital, Kenya. East Afr Med J 1992; 69: 670-4.
15.SPSS: Statistical Package for the Social Sciences. In: SPSS-Inc, editor. 14.0.2 ed 2006.
16.Davis TM, Supanaranond W, Pukrittayakamee S, Karbwang J, Molunto P, Mekthon S, et al. A safe and effective consecutive-infusion regimen for rapid quinine loading in severe falciparum malaria. J Infect Dis 1990; 161: 1305-8.
17.Njuguna P, Newton C. Management of severe falciparum malaria. J Postgrad Med 2004; 50: 45-50.
18.Assimadi JK, Gbadoe AD, Agbodjan-Djossou O, Ayewada K, Goeh-Akue E, Kusiaku K, et al. Treatment of cerebral malaria in African children by intravenous quinine: comparison of a loading dose regimen to a regimen without a loading dose. Arch Pediatr 2002; 9: 587-94.
19.Fargier JJ, Louis FJ, Cot M, Maubert B, Hounsinou C, Louis JP, et al. Reduction of coma by quinine loading dose in falciparum cerebral malaria. Lancet 1991; 338: 896-7.
20.Pasvol G, Newton CR, Winstanley PA, Watkins WM, Peshu NM, Were JB, et al. Quinine treatment of severe falciparum malaria in African children: a randomized comparison of three regimens. Am J Trop Med Hyg 1991; 45: 702-13.
21.Farver DK, Lavin MN. Quinine-induced hepatotoxicity. Ann Pharmacother 1999; 33: 32-4.
22.Mishra SK, Mohanty S, Das BS, Patnaik JK, Satpathy SK, Mohanty D. Hepatic changes in P. falciparum malaria. Indian J Malariol 1992; 29: 167-71.
23.Bonington A, Davidson RN, Winstanley PA, Pasvol G. Fatal quinine cardiotoxicity in the treatment of falciparum malaria. Trans R Soc Trop Med Hyg 1996; 90: 305-7.
24.Dondorp A, Nosten F, Stepniewska K, Day N, White N; South East Asian Quinine Artesunate Malaria Trial (Seaquamat Group). Artesunate versus quinine for treatment of severe falciparum malaria: a randomised trial. Lancet 2005; 366: 717-25.
25.Jacqz-Aigrain E, Bennasr S, Desplanques L, Peralma A, Beaufils F. (Severe poisoning risk linked to intravenous administration of quinine). Arch Pediatr 1994; 1: 14-9.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




