Dr Anjum Naeem ( Department of Medicine , Shaukat Khanum Memorial Cancer Hospital and Research Centre , Lahore. )
Hassan Hameed ( Department of Medicine , Shaukat Khanum Memorial Cancer Hospital and Research Centre , Lahore. )
Bashir Ahmad ( Department of Medicine , Shaukat Khanum Memorial Cancer Hospital and Research Centre , Lahore. )
Muhammad Tahir Aziz ( Department of Medicine , Shaukat Khanum Memorial Cancer Hospital and Research Centre , Lahore. )
Yasser Hussain ( Department of Medicine , Shaukat Khanum Memorial Cancer Hospital and Research Centre , Lahore. )
Mansoor Ahmed ( Department of Medicine , Shaukat Khanum Memorial Cancer Hospital and Research Centre , Lahore. )
January 2011, Volume 61, Issue 1
Original Article
Abstract
Objective: To check the effectiveness of ticarcillin clavulanate versus cefepime as monotherapy in febrile neutropenia in lymphoma patients and also to check tolerability profile of both drugs.
Methods: We randomly assigned 107 neutropenic patients to receive either cefepime or ticarcillin/clavulanate. The clinical efficacy and tolerability profile of both drugs were compared using either cefepime or ticarcillin clavulanate (TC) as an empirical treatment for management of febrile neutropenia in lymphoma patients only with same characteristics at time of presentation.
Results: A significant difference in efficacy of the two treatment arms was noted. A successful outcome was reported with 28 (51%) out of 55 in cefepime arm compared to 16(42%) out of 52 patients in ticarcillin/clavulanate group (p=0.35; 95% Confidence). The distribution of time for defervesence was estimated for each treatment group and a trend to a shorter time for defervesence was found in the CEFEPIME group (48.4 hour for cefepime, 58.28 hour for TC group; p= 0.018). For microbiologically documented infections, the successful eradication rate was 49% (6 of 14 patients) for TC group as compared to 83% (10 of 12 patients) for cefepime group. This difference was statistically significant for microbiologically documented infections. Twenty seven (52%) patients of TC group and 19(35%) of cefepime group required modifications of antibiotic regimen. The most frequent modifications consisted of the addition of either an amino glycoside (amikacin) or glycopeptides (vancomycin).
Conclusion: CEFEPIME regimen was more effective than TC regimen, with a consistent trend toward a better outcome associated with cefepime compared to Ticarcillin/clavulanate.
Keywords: Febrile Neutropenic, cefepinie, ticarcillin/clavulanate (JPMA 61:18; 2011).
Introduction
Febrile neutropenia is a common complication of cancer treatment associated with significant morbidity and mortality. Early institution of broad-spectrum antibiotic treatment reduces mortality.1,2 The combination of ß-lactam antibiotics plus an amino glycoside or a glycopeptides was long considered the \'gold standard\' of empirical antibiotic therapy in febrile neutropenic patients.3 This combination therapy had proven effectiveness as documented in various studies and had good antimicrobial activity against both gram positive and negative bacterium and a low incidence of emerging bacterial resistance. However there are disadvantages of combination treatment as high cost, need of drug monitoring with low therapeutic index like amino glycoside or vancomycin and resource intensity involved in preparation and application. Recent evolution of broad spectrum antibiotics like carbepenems, anti pseudomonal penicillin or fourth generation cephalosporin led to a change in this practice during the past decade. Thus, recent studies have evaluated carbepenems and third- or fourth-generation cephalosporin as initial monotherapy in febrile neutropenic cancer patients and found that monotherapy is as effective and safer than the classical combination of a ß-lactam and an amino glycoside.4-7 Current guidelines propose \'cefepime or ceftazidime or imipenem or meropenem\', empirically, as single ß-lactams, with or without vancomycin for management of febrile neutropenia.8,9 The potential of tricarcillin clavulanate is pointed at, stipulating further experience. The aim of this prospective randomized study was to evaluate and to compare the efficacy and safety of cefepime and triacrcillin/clavulanate as empirical monotherapy and as part of a sequential regimen.
Materials and Methods
The study was a prospective, open, randomized controlled trial. Included were all febrile neutropenic patients who had been treated with conventional chemotherapy for a primary, refractory or relapsed lymphoma at the department of medical oncology, Shaukat Khanum Memorial Cancer Hospital and Research centre. Eligible were patients with1 temperature of 38ºC over 4 hours apart within 24 hours or a single temperature spike of greater than 38.5ºC.2 Absolute Neutrophil Count of less than 500 or more than 500 with predicted decline to less than 500 in next 24 hours3 adult patients (age 18 to 65 years)4 patients falling in low to moderate risk category according to Infectious Disease Society of America (IDSA) guidelines. Patients who had repeated febrile neutropenic episodes during consecutive cytostatic treatment periods, could be entered more than once. Patients were excluded from the study if they had1 met initial criteria for vancomycin or getting dual therapy with or without vancomycin on day 12 severe hepatic or renal impairment (total bilirubin more than 2 or creatinine more than 2 or creatinine clearance less than 40 ml/min)3 if they had a history of allergy or other incompatibility to one of the study drugs4 patient with systolic blood pressure less than 905 with indwelling catheter or known MRSA colonization. After informed consent patients were randomly assigned to receive either cefepime (2 gram i/v Q8 hourly) or ticarcillin/clavulanate (3.2 gm i/v Q6 hourly) for empirical monotherapy in febrile neutropenia. Prior to commencement of therapy, history, physical examination, complete blood count, serum chemistry, chest X-ray, blood culture and urine analysis were done to evaluate febrile neutropenia. During a febrile episode complete blood count was done daily and serum chemistry on alternate days. Anti microbial susceptibility testing of bacterial isolates were performed by disc diffusion method. Patients were examined daily for clinical symptoms and signs of side effect.
Febrile episodes were classified as1 microbiologically documented infection (both site of infection and organism identified).2 Clinically documented infection (site of infection identified and no organism isolated)3 possible infection where infection was suspected but no site and organism was isolated). Patient was evaluated at 72 hours and on 7th day after start of treatment. The response to treatment was defined as resolution/ disappearance of fever within 72 hours of starting treatment, along with overall clinical improvement, eradication of infection without modification of antibiotic therapy and no recurrence of infection one week after discontinuation of antibiotics.
The treatment was considered a failure if persistence of fever of more than 38ºC at 72 hours or persistence of infecting organism requiring modification of antibiotic therapy at 72 hours, death within first 48 hours (primarily related to infection) and relapse defined as reappearance of infection within seven days of discontinuation of antibiotics.
For statistical analysis, two tailed Fisher\'s exact test and chi-square\'s test were used to compare difference in proportion between groups and response rate. Differences between medians were analyzed by the Mann-Whitney U test. Confidence interval (CI) of 95% for difference between two study groups was determined where appropriate.
For the sample size calculation, it was hypothesized that the use of ticarcillin/ clavulanate had the response rate equal to or more than cefepime. For a study to detect such an effect 50 patients in each treatment groups were required.
Age and gender distribution of the patients were calculated and for age mean, median, mode, standard deviation and range were determined. For each febrile neutropenia episode, afebrile status following three days of therapy was categorized into YES/NO. Accordingly, Z test for two proportions along with 95% confidence limit around the point estimate was used to evaluate difference between the two treatment groups. For studying episodes of febrile neutropenia and to evaluate difference of neutropenic interval between cefepime and ticarcillin/clavulanate and to compare duration of therapy in each arm, the Mann-Whitney U test was used.
Results
This was a prospective, randomized controlled clinical trial with a total of 123 neutropenic patients with persistent fever. Sixty one patients were randomly assigned in cefepime group and 59 in the Ticarcillin/clavulanate group, of whom 16 patients were not eligible for the following reasons: error in definition of treatment regimen (11 patients) and administration of parental antibiotics other than study regimens (5 patients). So a total of 107 patients were assessed for the study drugs assessment, 55 in the cefepime group and 52 in the ticarcillin/clavulanate group.
At study inclusion there was no significant difference between the 2 treatment groups in gender and age and also patients in both arms were well balanced with respect to underlying disease. Eighty one patients out of 107 had Non Hodgkin lymphoma and 20 had Hodgkin\'s disease as shown in Table-1.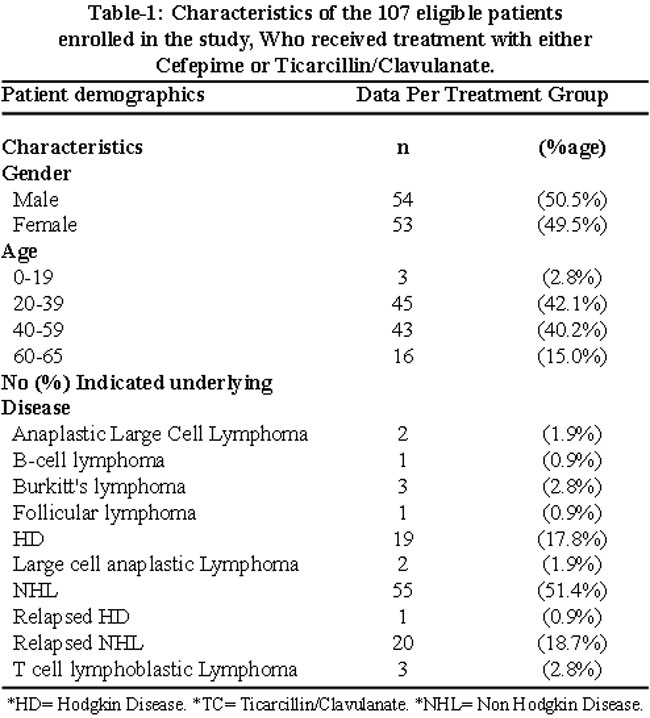
The two groups had similar use of growth factors, previous oral antibiotic prophylaxis, presence and severity of mucositis and other concomitant medical conditions. Also at the start of study drugs therapy, all patients had an absolute neutrophil count of more than 100 micro /liter. Patients with absolute neutrophil count between 100 and 500 were 71% and 63% respectively while those with ANC between 500 and 1000 were 28.8% and 36.4 % in ticarcillin/clavulanate and cefepime group respectively
During therapy, the number of patients with persistent febrile neutropenia in both groups were; ANC < 100/mm3 were 30.8% and 20.0%, ANC 100-500/mm3 were 21.2% and 54.5% and patients with ANC 500-1000/mm3 were 48.1% and 25.5% respectively in ticarcillin/clavulanate and cefepime group.
Overall, no difference was found in both groups either in vital signs, duration and intensity of neutropenia and laboratory parameters scrutinized during therapy. Mean duration of therapy in both groups was 7 days.
Patients were assessed for low risk and moderate risk as per patient risk constraint of IDSA before starting therapy. As per mentioned parameters, low risk patients were 27% and moderate risk patients were 73% in ticarcillin arm and in cefepime arm 33% were low risk and 67% were moderate risk patients.
While assessing clinical response, a total of 62 episodes were classified as unexplained fever between two treatment groups. Breakdown between two groups was 27/33 (63%) in ticarcillin group and 35/39 (71%) in cefepime group.
Response rate for microbiologically documented infections in comparison to possible infections was not significantly different in both groups with P -value (95% confidence interval) .086 and .095 respectively.
Response rate to bacteraemia in cefepime group was significantly different than the response rate of ticarcillin/clavulanate (Table-2).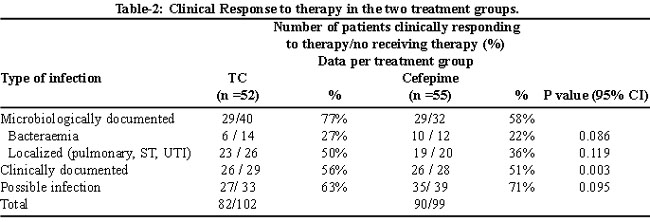
The overall response rate to therapy was compared in the two groups on day 7 after start of therapy. A successful outcome was reported in 16 (42%) out of 52 patients in ticarcillin/clavulanate group compared to 28 out of 55 patients in cefepime group (p = 0.35; CI 95%). Similarly on second visit 72 hours after inclusion in study, fever resolution was experienced in 42 patients ( 76%) of ticarcillin group as compared to 51 patients in cefepime group (93%). A shorter time to defervescence was also experienced in cefepime group as compared to ticarcillin group (48.4 hours for cefepime as compared to 58.28 hours for ticarcillin/clavulante group). Persistence of fever was statistically insignificant in both groups, with 19% in ticaricillin group and 7 % in cefepime group having no resolution of fever.
The low number of patients with microbiologically documented infection in ticarcillin group precluded further destratification of analysis. Relapse rate of favourable responders within 14 days of study inclusion were similar for both cefepime (5%) and ticarcillin clavulanate (11%) group (P = 0.51). Overall good response rate was statistically significant in both groups with 31% in cefepime and 51% in ticarcillin/clavulanate group ( p=0.035) (Table-3).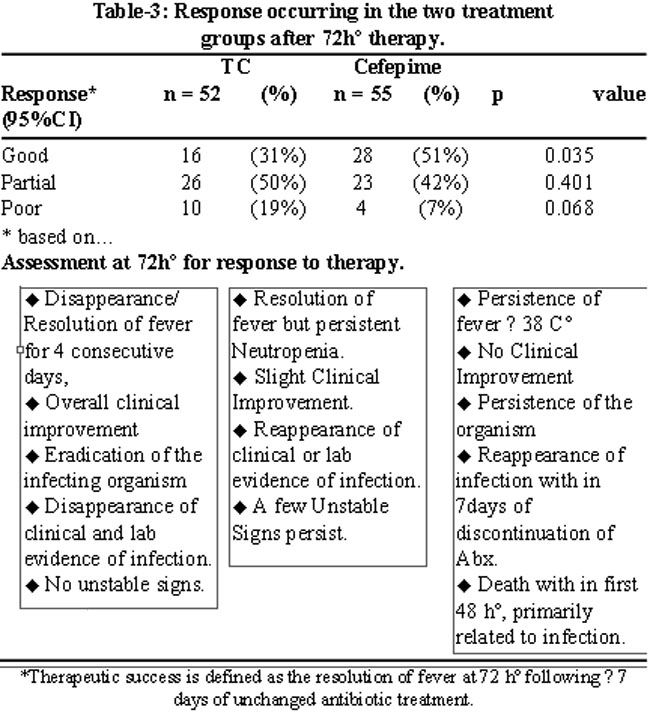
Poor response was 19% in Ticarcillin group and 7% in cefepime group.
Antibiotic modification within 7 days of start of therapy was required in 26 (50%) patients in Ticarcillin group and 19 (35%) in cefepime group.
Within the complete follow up period established in the protocol (14 days) and considering all available data, none of the 55 patients in cefepime group and 1 out of the 53 eligible patients enrolled in ticarcillin group died due to infection and super infection. There was no statistically significant difference in overall mortality rates between the two treatment groups. Mortality due to presenting infection and further infection was relatively uncommon, most deaths being attributed to underlying disease, fatal haemorrhage, and other causes. No death was related to toxicity to study antibiotics therapy.
Safety:
All 107 patients enrolled in the study were evaluated for adverse experience on day 7 after initiation of therapy. 17 patients in TC group and 23 in cefepime group (p < 0.265 CI 95%) faced adverse reaction considered possibly, probably or definitely related to study drugs. When only moderate and severe adverse reactions were considered, no statistically significant difference was experienced between the two treatment groups. Nephrotoxicity deemed probable or definitely related to study drugs developed in 1 patient in cefepime group and 3 patients of ticarcillin group. Thrombocytopenia related to antibiotics regimen was reported for 2 patients in cefepime arm and 3 in ticaricillin arm. Nausea and vomiting experienced in 3 patients of cefepime group and 2 of Ticarcillin group. Also 6 patients of ticarcillin arm and 11 patients of cefepime arm developed gastrointestinal intolerance mainly diarrhoea. A mild skin rash was seen in 1 patient of Ticarcillin group and none in cefepime group. In none of patients in both study groups, the drugs were discontinued because of toxicity.
Discussion
Despite the advent of recombinant colony stimulating factor, febrile neutropenia remains a major concern among patients experiencing myelotoxicity from standard chemotherapy.1 These issues are of paramount importance in patients who received high dose chemotherapies with or without radiation. The treatment of febrile neutropenia with a combination of beta lactam and aminglycoside remains the gold standard. But the advent of broad spectrum, bactericidal antibiotics such as carbepenems or extended spectrum cephalosporin\'s such as cefepime has raised questions whether combination therapy can be safely replaced with monotherapy in management of granulocytopenic febrile neutropenia.3,10-12 Recent data as in one study conducted at EORTC reported similar success rate when cefepime and ceftazidime were compared with amikacin in empiric treatment of febrile neutropenia.6 Use of cefepime in combination or alone in febrile neutropenia is FDA approved. Anti-pseudomonal penicillin has a spectrum similar to cefepime but its efficacy and safety as monotherapy in febrile neutropenia is not yet confirmed in adults.13
The presented study established that cefepime and ticarcillin/clavulanate are not equally effective in management of febrile neutropenia in lymphoma patients with low to moderate neutropenia. Interestingly our study observed that response rate in both treatment arms are lower than expected (response rate in randomized trials range from 68 to 82%). The lower response may be explained by the theory how efficacy is compared to effectiveness in general: while efficacy measures drug performance in ideal setting and under strict protocol procedures that determine how the drug must be used and monitored. On the other hand, effectiveness quantifies benefit expected from therapeutic strategy and in a real setting. Due to a small number of patients with microbiologically documented infection in each treatment group, it was difficult to interpret the difference in responses among various clinical sites of infections and different infective organisms.
Safety data in both treatment arms was comparable. Although two regimens were comparable in term of clinical effectiveness, in clinically documented infections. However, when regarding response to bactaeremia, overall response to therapy, distribution of time to defervesence suggests that ticarcillin /clavulanate regimen is not as effective as cefepime in treatment of early episodes of febrile neutropenia in haematological malignancies after chemotherapy.
Moreover there has only been one trial done in the past that compared cefepime versus ticaricillin vlavulanate and azetronam in febrile neutropenia with high dose chemotherapy.14 They found good efficacy of cefepime and low cost, as compared to Ticarcillin clavulanate and azetronam. Although we cannot compare our trial with the previously mentioned due to the fact that the population they targeted was different and the drug combination was different with addition of azetronam to ticaricillin clavulanate.14 Although the above mentioned trial did target a different population and high risk patients, it still showed similar results with cefepime as a better choice when compared to Ticarcillin clavulanate. Out trial is the first that compared ticarcillin/clavlanate with cefepime as monotherapy. Data surely favours cefepime, but further confirmation with bigger multicentre trials is needed.
Conclusion
In conclusion cefepime seems to be more efficacious than ticarcillin/clavulanate and may be associated with lesser requirement of amino glycoside, glycopeptides or both. Safety profile of cefepime is comparable to ticarcillin/clavulanate with slightly more side effects in cefepime arm. But this cannot be said that cefepime is a very good option or addition of either amino glycoside or glycopeptides may not be necessary to treat febrile neutorpenia.
With emerging and expanding resistance of Extended spectrum beta lactamse producing bacterium resistance varies from country to country and even city to city as well. It is thus recommended to follow the local antibiogram and guidelines before prescribing cefepime or ticarcillin clavulanate in patients with febrile neutropenia.
References
1.Pizzo PA. Empirical therapy and prevention of infection in the immunocompromised host. In: Mandell GL, Douglas RG Jr, Bennett JE, eds. Principles and practice of infectious diseases. 3rd ed. New York: Churchill Livingstone, 1990; pp 2303-12.
2.Rubin M, Walsh TJ, Pizzo PA. Clinical approach to infections in the compromised host. In: Hoffman R, Benz EJ Jr, Shattil SJ, Furie B, Cohen HJ, eds. Hematology: basic principles and practice. New York: Churchill Livingstone, 1991; pp 1063-114.
3.Cometta A, Zinner S, de Bock R, Calandra T, Gaya H, Klastersky J, et al. Piperacillin-tazobactam plus amikacin versus ceftazidime plus amikacin as empiric therapy for fever in granulocytopenic patients with cancer. The International Antimicrobial Therapy Cooperative Group of the European Orgnaization for Research and Treatment of Cancer. Antimicrob Agents Chemother 1995; 39: 445-52.
4.Bodey GP, Elting L, Kassamali H, Lim BP. Escherichia coli bacteremia in cancer patients. Am J Med 1986; 81: 85-95.
5.Ramphal R, Gucalp R, Rotstein C, Cimino M, Oblon D. Clinical experience with single agent and combination regimens in the management of infection in the febrile neutropenic patient. Am J Med 1996; 100: 83S-89S.
6.Paul M, Soares-Weiser K, Leibovici L. Beta-Lactam monotherapy versus Beta-lactam-aminoglycoside combination therapy for fever with neutropenia: systematic review and meta-analysis. BMJ 2003; 326: 1111.
7.Rolston KV, Berkey P, Bodey GR, Anaissie EJ, Khardori NM, Joshi JH, et al. A comparison of imipenem to ceftazidime with or without amikacin as empiric therapy in neutropenic patients. Arch Intern Med 1992; 152: 283-91.
8.Hughes WT, Armstrong D, Bodey GP, Bow EL, Brown AE, Calandra T, et al. Guidelines for the use of antimicrobialagents in neutropenic patients with cancer. Clin Infect Dis 2002; 34: 730-51.
9.Montalar J, Segura A, Bosch C, Galan A, Juan O, Molins C, Giner V, Aparicio J.Cefepime monotherapy as an empirical initial treatment of patients with febrile neutropenia. Med Oncol 2002; 19: 161-6.
10.Jaresko GS, Zarotsky V, Holtom PD. Cefepime use in febrile neutropenic patients (FNP): a meta-analysis. Interscience Conference on Antimicrobial Agents and Chemotherapy 2000; 40: 474.
11.Tamura K, Matsuoka H, Tsukada J, Masuda M, Ikeda S, Matsuishi E, et al. Cefepime or carbapenem treatment for febrile neutropenia as a single agent is as effective as a combination of 4th-generation cephalosporin+aminoglycosides: comparative study. Am J Hematol 2002; 71: 248-55.
12.Pereira CA, Petrilli AS, Carlesse FA, Luisi FA, da Silva KV, de Martino Lee ML. Cefepime monotherapy is as effective as ceftriaxone plus amikacin in pediatric patients with cancer and high-risk febrile enutropenia in a randomized comparison. J Microbiol Immunol Infect 2009; 42: 141-7.
13.Uygun V, Karasu GT, Ogune D, Yesilipek A, Hazar V. Piperacillin/taobactam versus cefepime for the empirical treatment of pediatric cancer patients with neutropenia and fever: a randomized and open-label study. Pediatr Blood Cancer 2009; 53: 610-4.
14.Fleming DR, Ziegler C, Baize T, Mudd L, Goldsmith GH, Herzig RH. Cefepime versus ticarcillin and clavulanate potassium and aztreonam for febrile neutropenia therapy in high-dose chemotherapy patients. Am J Clin Oncol 2003; 26: 285-8.
Related Articles
Journal of the Pakistan Medical Association has agreed to receive and publish manuscripts in accordance with the principles of the following committees:




